Abstract
Crit Care Sci. 2023;35(3):243-255
DOI 10.5935/2965-2774.20230136-pt
To update the recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
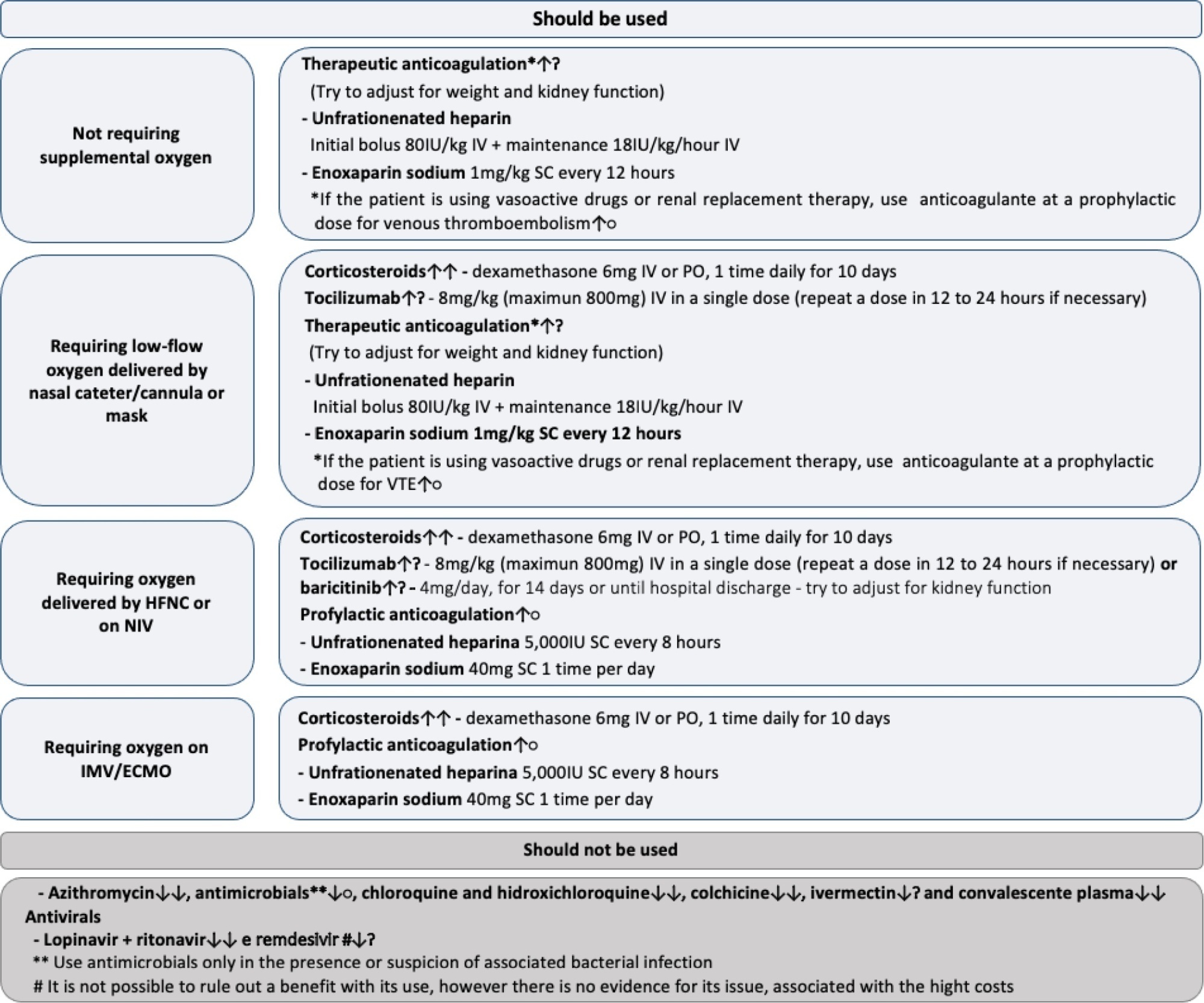
Twenty-one recommendations were generated, including strong recommendations for the use of corticosteroids in patients using supplemental oxygen and conditional recommendations for the use of tocilizumab and baricitinib for patients on supplemental oxygen or on noninvasive ventilation and anticoagulants to prevent thromboembolism. Due to suspension of use authorization, it was not possible to make recommendations regarding the use of casirivimab + imdevimab. Strong recommendations against the use of azithromycin in patients without suspected bacterial infection, hydroxychloroquine, convalescent plasma, colchicine, and lopinavir + ritonavir and conditional recommendations against the use of ivermectin and remdesivir were made.
New recommendations for the treatment of hospitalized patients with COVID-19 were generated, such as those for tocilizumab and baricitinib. Corticosteroids and prophylaxis for thromboembolism are still recommended, the latter with conditional recommendation. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and to promote resource economy.
Abstract
Rev Bras Ter Intensiva. 2022;34(1):87-95
DOI 10.5935/0103-507x.20220003-en
The TELE-critical Care verSus usual Care On ICU PErformance (TELESCOPE) trial aims to assess whether a complex telemedicine intervention in intensive care units, which focuses on daily multidisciplinary rounds performed by remote intensivists, will reduce intensive care unit length of stay compared to usual care.
The TELESCOPE trial is a national, multicenter, controlled, open label, cluster randomized trial. The study tests the effectiveness of daily multidisciplinary rounds conducted by an intensivist through telemedicine in Brazilian intensive care units. The protocol was approved by the local Research Ethics Committee of the coordinating study center and by the local Research Ethics Committee from each of the 30 intensive care units, following Brazilian legislation. The trial is registered with ClinicalTrials. gov (NCT03920501). The primary outcome is intensive care unit length of stay, which will be analyzed accounting for the baseline period and cluster structure of the data and adjusted by prespecified covariates. Secondary exploratory outcomes included intensive care unit performance classification, in-hospital mortality, incidence of nosocomial infections, ventilator-free days at 28 days, rate of patients receiving oral or enteral feeding, rate of patients under light sedation or alert and calm, and rate of patients under normoxemia.
According to the trial’s best practice, we report our statistical analysis prior to locking the database and beginning analyses. We anticipate that this reporting practice will prevent analysis bias and improve the interpretation of the reported results.
Abstract
Rev Bras Ter Intensiva. 2022;34(1):107-115
DOI 10.5935/0103-507X.20220005-en
To evaluate clinical practices and hospital resource organization during the early COVID-19 pandemic in Brazil.
This was a multicenter, cross-sectional survey. An electronic questionnaire was provided to emergency department and intensive care unit physicians attending COVID-19 patients. The survey comprised four domains: characteristics of the participants, clinical practices, COVID-19 treatment protocols and hospital resource organization.
Between May and June 2020, 284 participants [median (interquartile ranges) age 39 (33 - 47) years, 56.3% men] responded to the survey; 33% were intensivists, and 9% were emergency medicine specialists. Half of the respondents worked in public hospitals. Noninvasive ventilation (89% versus 73%; p = 0.001) and highflow nasal cannula (49% versus 32%; p = 0.005) were reported to be more commonly available in private hospitals than in public hospitals. Mechanical ventilation was more commonly used in public hospitals than private hospitals (70% versus 50%; p = 0,024). In the Emergency Departments, positive endexpiratory pressure was most commonly adjusted according to SpO2, while in the intensive care units, positive end-expiratory pressure was adjusted according to the best lung compliance. In the Emergency Departments, 25% of the respondents did not know how to set positive end-expiratory pressure. Compared to private hospitals, public hospitals had a lower availability of protocols for personal protection equipment during tracheal intubation (82% versus 94%; p = 0.005), managing mechanical ventilation [64% versus 75%; p = 0.006] and weaning patients from mechanical ventilation [34% versus 54%; p = 0.002]. Finally, patients spent less time in the emergency department before being transferred to the intensive care unit in private hospitals than in public hospitals [2 (1 - 3) versus 5 (2 - 24) hours; p < 0.001].
This survey revealed significant heterogeneity in the organization of hospital resources, clinical practices and treatments among physicians during the early COVID-19 pandemic in Brazil.
Abstract
Rev Bras Ter Intensiva. 2022;34(1):1-12
DOI 10.5935/0103-507X.20220001-en
Several therapies are being used or proposed for COVID-19, and many lack appropriate evaluations of their effectiveness and safety. The purpose of this document is to develop recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
A group of 27 experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Sixteen recommendations were generated. They include strong recommendations for the use of corticosteroids in patients using supplemental oxygen, the use of anticoagulants at prophylactic doses to prevent thromboembolism and the nonuse of antibiotics in patients without suspected bacterial infection. It was not possible to make a recommendation regarding the use of tocilizumab in patients hospitalized with COVID-19 using oxygen due to uncertainties regarding the availability of and access to the drug. Strong recommendations against the use of hydroxychloroquine, convalescent plasma, colchicine, lopinavir + ritonavir and antibiotics in patients without suspected bacterial infection and also conditional recommendations against the use of casirivimab + imdevimab, ivermectin and rendesivir were made.
To date, few therapies have proven effective in the treatment of hospitalized patients with COVID-19, and only corticosteroids and prophylaxis for thromboembolism are recommended. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and promote economical resource use.

Abstract
Rev Bras Ter Intensiva. 2021;33(4):624-634
DOI 10.5935/0103-507X.20210088
To identify the clinical and epidemiological profile of adult intensive care units in Brazil.
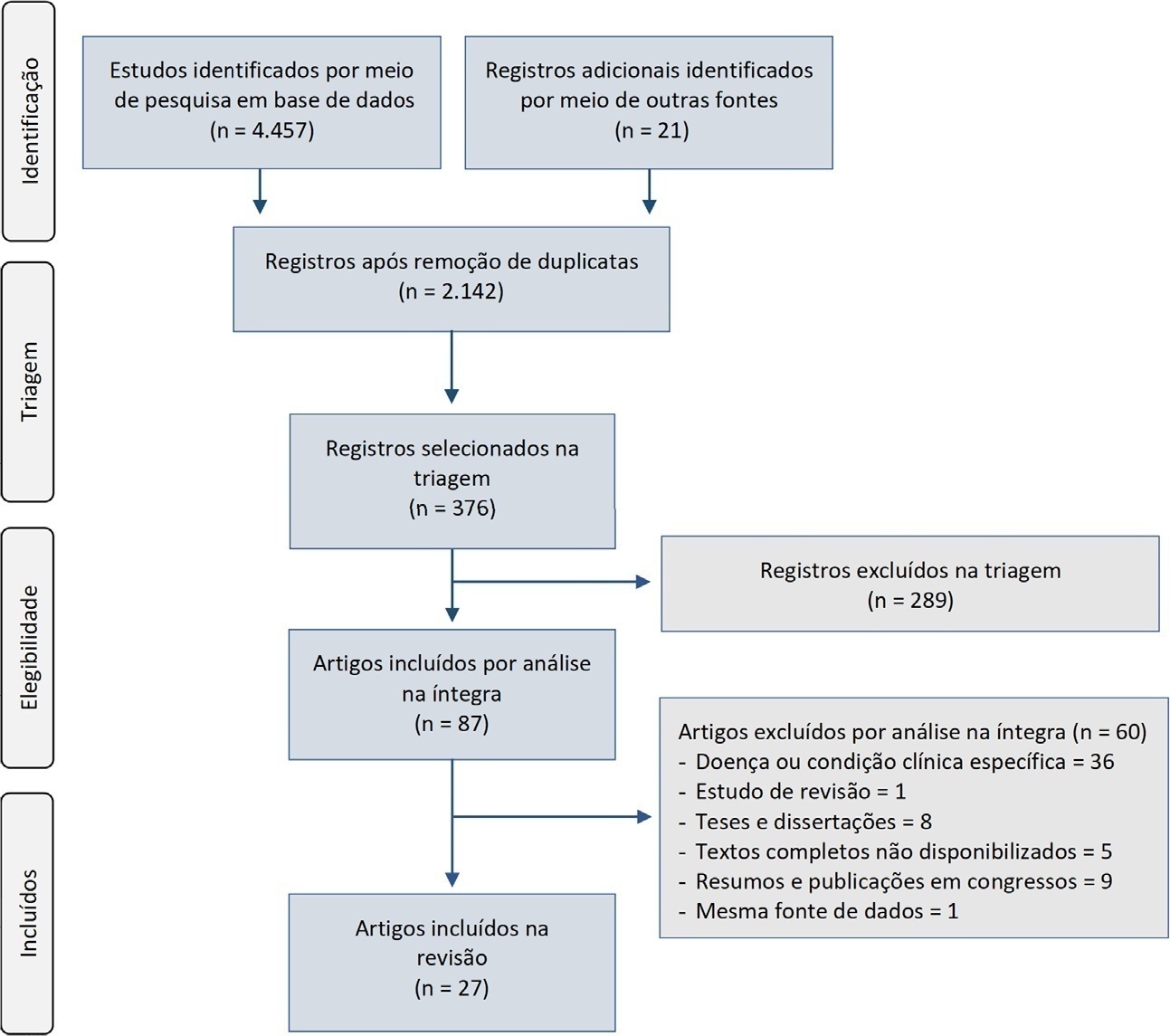
A systematic review was performed using a comprehensive strategy to search PubMed®, Embase, SciELO, and the Biblioteca Virtual em Saúde. The eligibility criteria for this review were observational studies that described the epidemiological and/or clinical profile of critically ill patients admitted to Brazilian intensive care units and were published between 2007 and 2020.
From the 4,457 identified studies, 27 were eligible for this review, constituting an analysis of 113 intensive care units and a final sample of 75,280 individuals. There was a predominance of male and elderly patients. Cardiovascular diseases were the main cause of admission to the intensive care unit. The Acute Physiology and Chronic Health Evaluation II score was the most widely used disease severity assessment system. The length of stay and mortality in the intensive care unit varied widely between institutions.
These results can help guide the planning and organization of intensive care units, providing support for decision-making and the implementation of interventions that ensure better quality patient care.
Abstract
Rev Bras Ter Intensiva. 2021;33(3):384-393
DOI 10.5935/0103-507X.20210055
To describe current clinical practices related to the use of high-flow nasal cannula therapy by Brazilian pediatric intensivists and compare them with those in other countries.
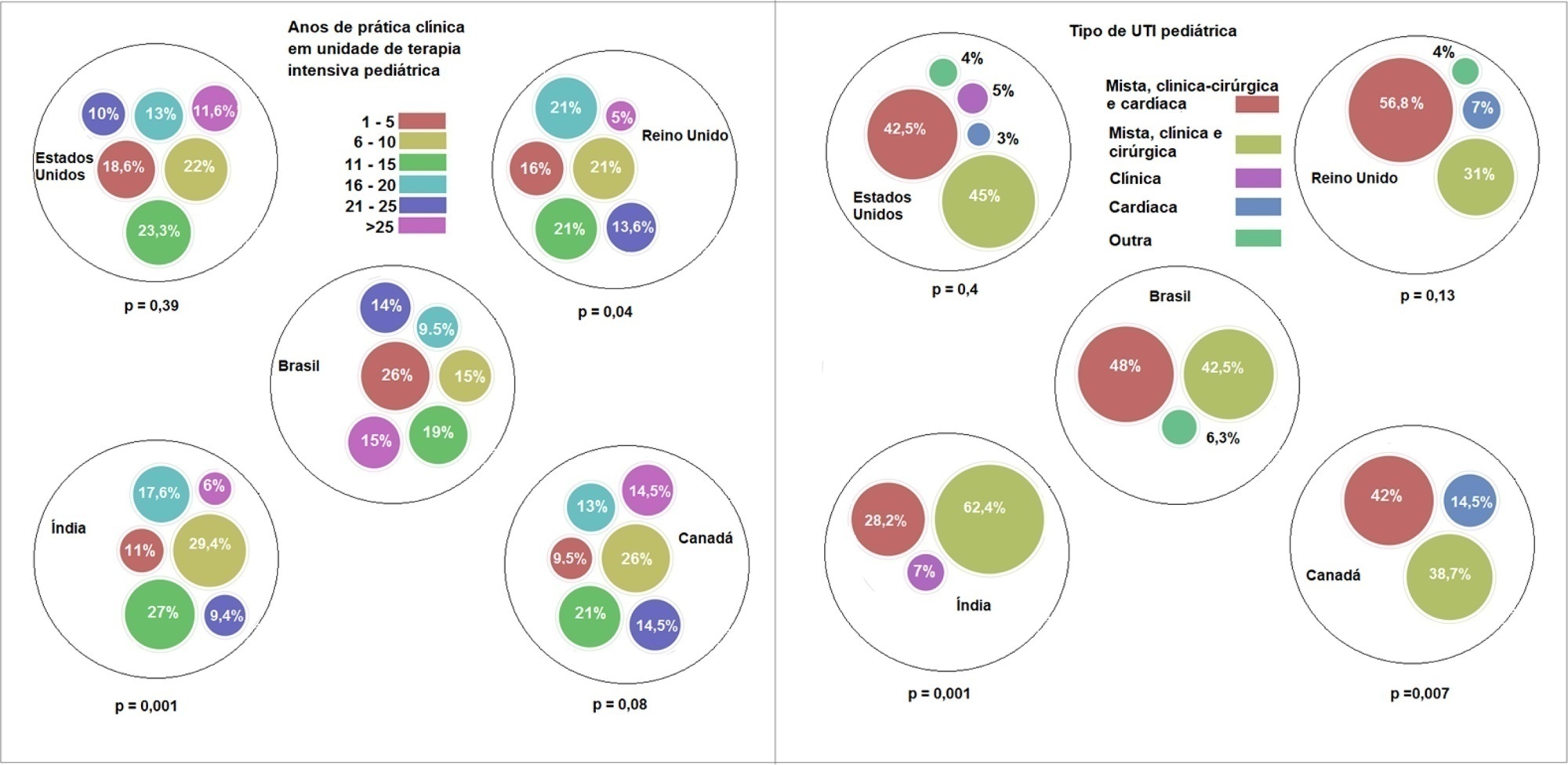
A questionnaire was administered to pediatric intensivists in North and South America, Asia, Europe, and Australia/New Zealand for the main study. We compared the Brazilian cohort with cohorts in the United States of America, Canada, the United Kingdom, and India
Overall, 501 physicians responded, 127 of which were in Brazil. Only 63.8% of respondents in Brazil had a high-flow nasal cannula available, in contrast to 100% of respondents in the United Kingdom, Canada, and the United States. The attending physician was responsible for the decision to start a high-flow nasal cannula according to 61.2% respondents in Brazil, 95.5% in the United Kingdom, 96.6% in the United States, 96.8% in Canada, and 84.7% in India. A total of 62% of respondents in Brazil, 96.3% in the United Kingdom, 96.6% in the United States, 96.8% in Canada, and 84.7% in India reported that the attending physician was responsible for the decision to wean or modify the high-flow nasal cannula settings. When high-flow nasal cannula therapy failed due to respiratory distress/failure, 82% of respondents in Brazil would consider a trial of noninvasive ventilation before endotracheal intubation, compared to 93% in the United Kingdom, 88% in the United States, 91.5% in Canada, and 76.8% in India. More Brazilian intensivists (6.5%) than intensivists in the United Kingdom, United States, and India (1.6% for all) affirmed using sedatives frequently with high-flow nasal cannulas.
The availability of high-flow nasal cannulas in Brazil is still not widespread. There are some divergences in clinical practices between Brazilian intensivists and their colleagues abroad, mainly in processes and decision-making about starting and weaning high-flow nasal cannula therapy.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):433-438
DOI 10.5935/0103-507X.20200073
To describe the use of neuromuscular blockade as well as other practices among Brazilian physicians in adult intensive care units.
An online national survey was designed and administered to Brazilian intensivists. Questions were selected using the Delphi method and assessed physicians’ demographic data, intensive care unit characteristics, practices regarding airway management, use of neuromuscular blockade and sedation during endotracheal intubation in the intensive care unit. As a secondary outcome, we applied a multivariate analysis to evaluate factors associated with the use of neuromuscular blockade.
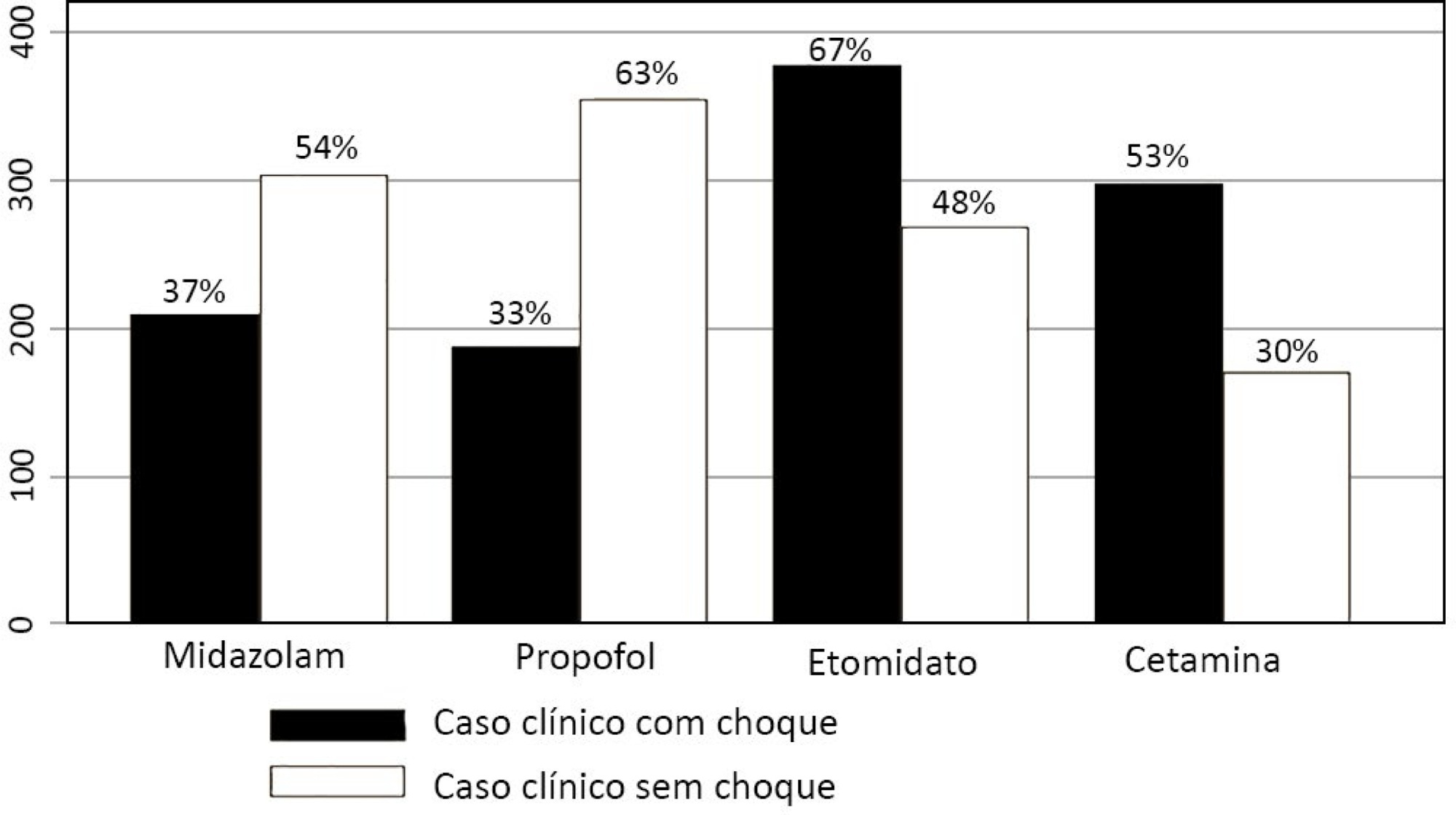
Five hundred sixty-five intensivists from all Brazilian regions responded to the questionnaire. The majority of respondents were male (65%), with a mean age of 38 ( 8.4 years, and 58.5% had a board certification in critical care. Only 40.7% of the intensivists reported the use of neuromuscular blockade during all or in more than 75% of endotracheal intubations. In the multivariate analysis, the number of intubations performed monthly and physician specialization in anesthesiology were directly associated with frequent use of neuromuscular blockade. Etomidate and ketamine were more commonly used in the clinical situation of hypotension and shock, while propofol and midazolam were more commonly prescribed in the situation of clinical stability.
The reported use of neuromuscular blockade was low among intensivists, and sedative drugs were chosen in accordance with patient hemodynamic stability. These results may help the design of future studies regarding airway management in Brazil.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária - ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Search
Search in:
Case reports (56) Child (53) Coronavirus infections (33) COVID-19 (45) Critical care (115) Critical illness (54) ICU (25) Infant, newborn (27) Intensive care (72) Intensive care units (254) Intensive care units, pediatric (31) mechanical ventilation (38) Mortality (75) Physical therapy modalities (28) Prognosis (61) Respiration, artificial (117) Respiratory insufficiency (26) risk factors (34) SARS-CoV-2 (28) Sepsis (98)