You searched for:"Erica Aranha Suzumura"
We found (5) results for your search.-
Review Article
Challenges for the development of alternative low-cost ventilators during COVID-19 pandemic in Brazil
Rev Bras Ter Intensiva. 2020;32(3):444-457
Abstract
Review ArticleChallenges for the development of alternative low-cost ventilators during COVID-19 pandemic in Brazil
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
Views0ABSTRACT
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária – ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Keywords:Biomedical engineeringBrazilCOVID-19Positive pressure respirationRespiration, artificialSevere acute respiratory syndromeVentilators, mechanicalSee more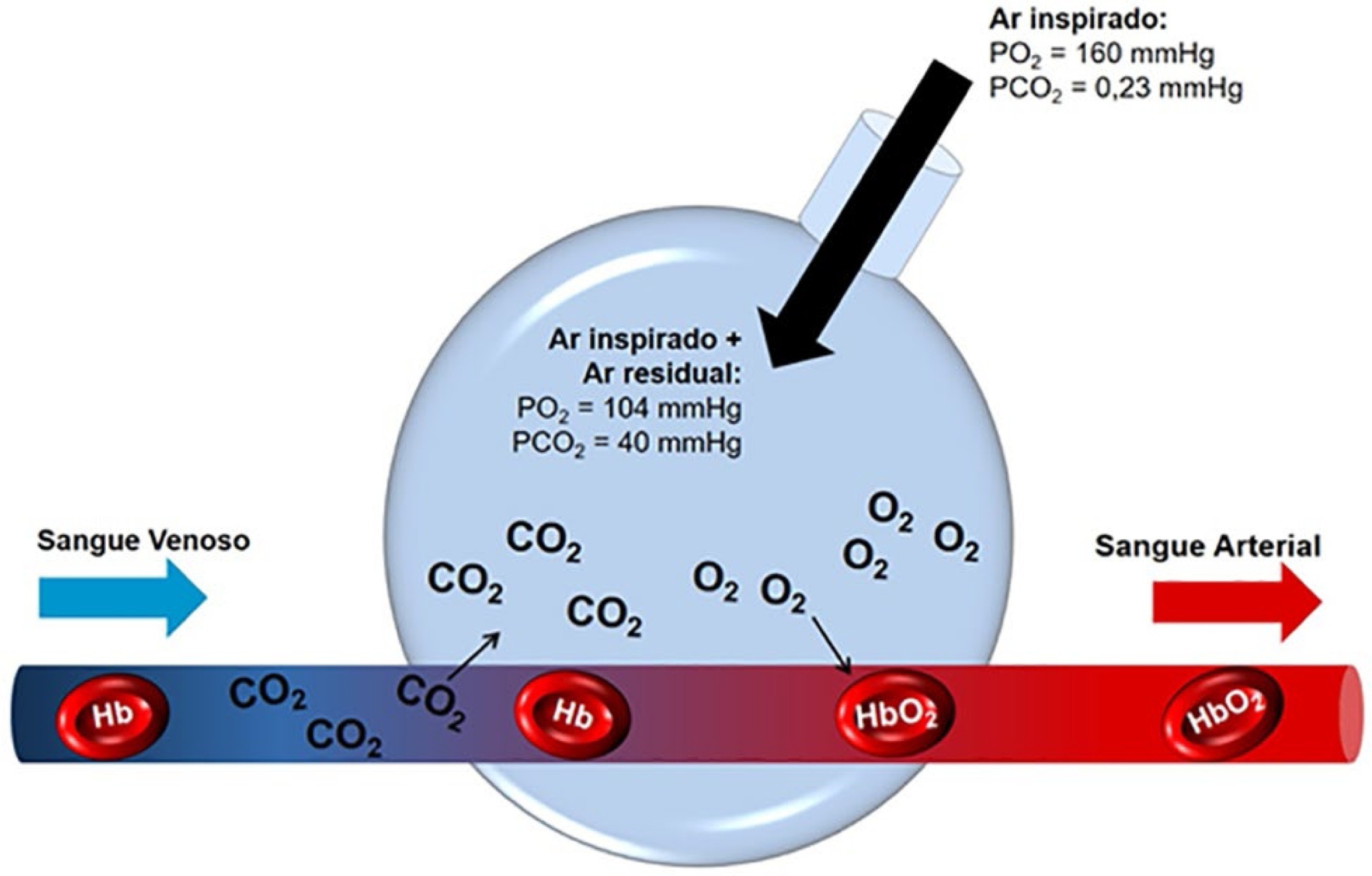
-
Original Articles
Statistical analysis plan for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART). A randomized controlled trial
Rev Bras Ter Intensiva. 2017;29(2):142-153
Abstract
Original ArticlesStatistical analysis plan for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART). A randomized controlled trial
Rev Bras Ter Intensiva. 2017;29(2):142-153
DOI 10.5935/0103-507X.20170024
Views0See moreABSTRACT
Background:
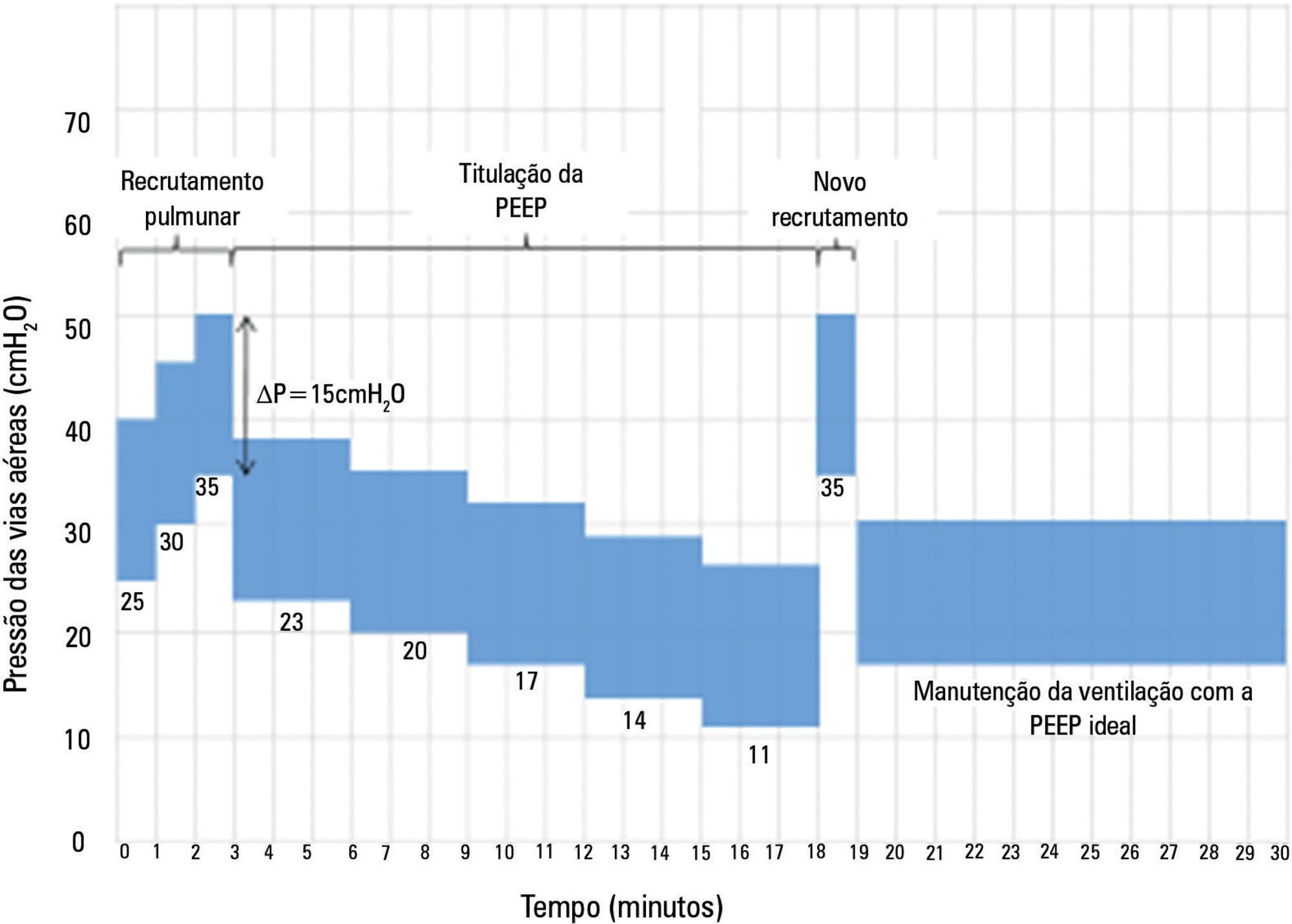
The Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) is an international multicenter randomized pragmatic controlled trial with allocation concealment involving 120 intensive care units in Brazil, Argentina, Colombia, Italy, Poland, Portugal, Malaysia, Spain, and Uruguay. The primary objective of ART is to determine whether maximum stepwise alveolar recruitment associated with PEEP titration, adjusted according to the static compliance of the respiratory system (ART strategy), is able to increase 28-day survival in patients with acute respiratory distress syndrome compared to conventional treatment (ARDSNet strategy).
Objective:
To describe the data management process and statistical analysis plan.
Methods:
The statistical analysis plan was designed by the trial executive committee and reviewed and approved by the trial steering committee. We provide an overview of the trial design with a special focus on describing the primary (28-day survival) and secondary outcomes. We describe our data management process, data monitoring committee, interim analyses, and sample size calculation. We describe our planned statistical analyses for primary and secondary outcomes as well as pre-specified subgroup analyses. We also provide details for presenting results, including mock tables for baseline characteristics, adherence to the protocol and effect on clinical outcomes.
Conclusion:
According to best trial practice, we report our statistical analysis plan and data management plan prior to locking the database and beginning analyses. We anticipate that this document will prevent analysis bias and enhance the utility of the reported results.
Trial registration:
ClinicalTrials.gov number, NCT01374022.
-
How to assess intensive care randomized trials
Rev Bras Ter Intensiva. 2009;21(2):219-225
Abstract
How to assess intensive care randomized trials
Rev Bras Ter Intensiva. 2009;21(2):219-225
DOI 10.1590/S0103-507X2009000200016
Views0See moreRandomized controlled trials are scientific investigations considered as the gold-standard to evaluate therapeutic interventions. Randomized controlled trials may examine the safety and efficacy of new drugs and therapeutic procedures or compare the effects of two or more drugs or any other intervention. In this article, we present the essential features of these studies, as well as, factors which may bias randomized controlled trials. We also present criteria to critically appraise articles reporting randomized controlled trials, explain how to interpret the results and how to apply them to clinical practice.

-
How to critically assess intensive care cohort studies?
Rev Bras Ter Intensiva. 2008;20(1):93-98
Abstract
How to critically assess intensive care cohort studies?
Rev Bras Ter Intensiva. 2008;20(1):93-98
DOI 10.1590/S0103-507X2008000100015
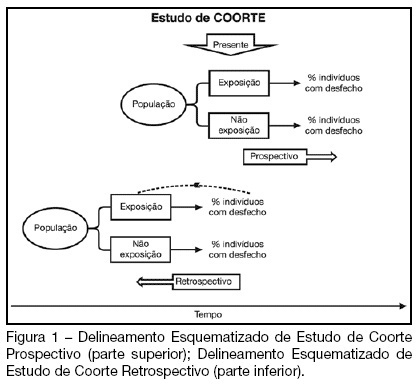
Views0See moreBACKGROUND AND OBJECTIVES: Cohort studies are useful to identify risk and prognostic factors, assess disease natural history and verify the impact of diagnostic or therapeutic interventions. This article aims to guide readers on how to critically assess papers using a cohort research design. CONTENTS: For a critical appraisal of cohort studies, one must observe: the existence of selection and information bias, the continuity of the follow-up, the control of confounding variables, the significance of the results and its applicability in clinical practice. CONCLUSIONS: Knowledge of factors which affect the quality of cohort studies allows intensive care professionals to select the best available evidence to guide the decision making process.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis





