Abstract
Crit Care Sci. 2024;36:e20240210en
DOI 10.62675/2965-2774.20240210-en
Driving pressure has been suggested to be the main driver of ventilator-induced lung injury and mortality in observational studies of acute respiratory distress syndrome. Whether a driving pressure-limiting strategy can improve clinical outcomes is unclear.
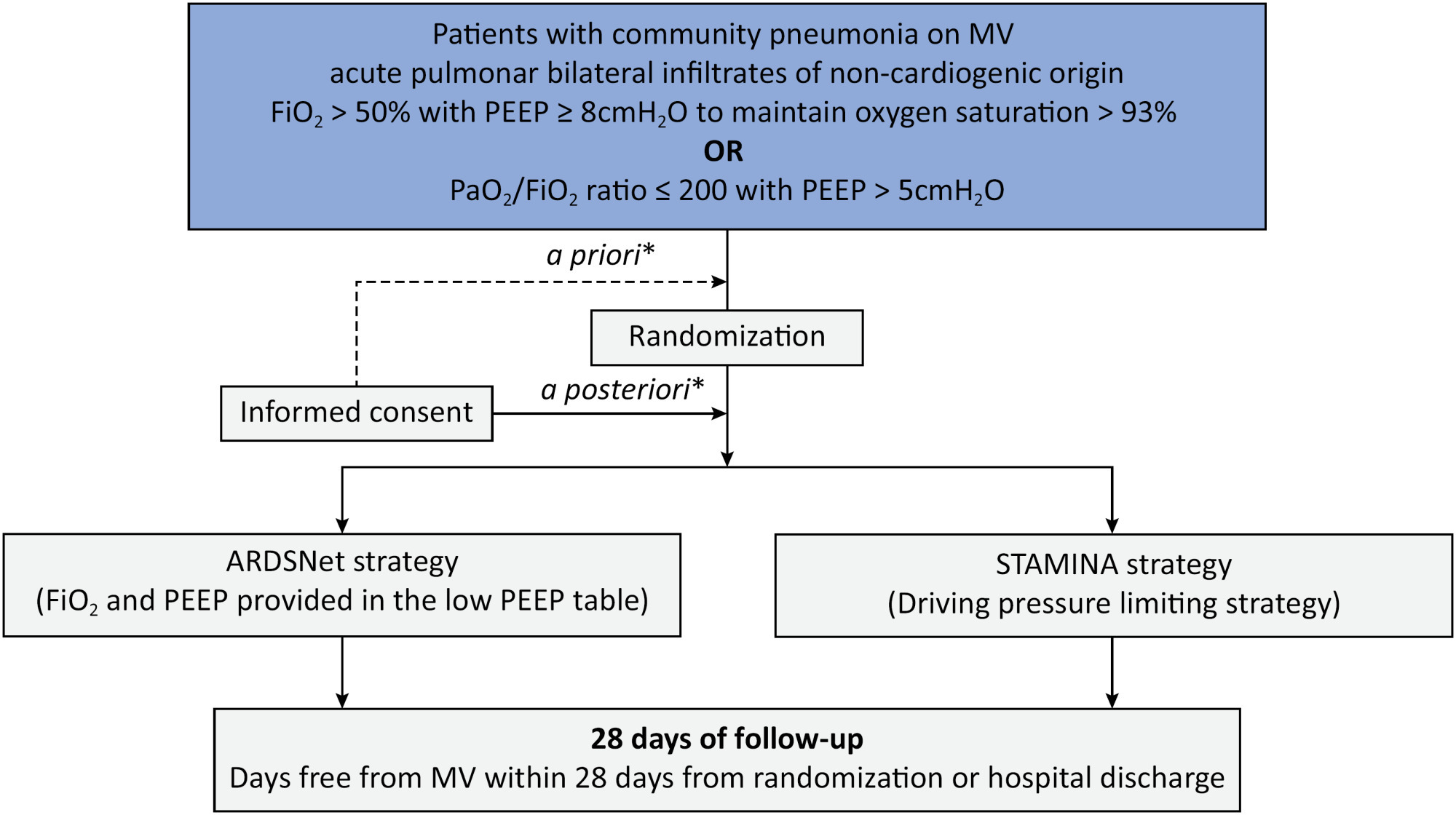
To describe the protocol and statistical analysis plan that will be used to test whether a driving pressure-limiting strategy including positive end-expiratory pressure titration according to the best respiratory compliance and reduction in tidal volume is superior to a standard strategy involving the use of the ARDSNet low-positive end-expiratory pressure table in terms of increasing the number of ventilator-free days in patients with acute respiratory distress syndrome due to community-acquired pneumonia.
The ventilator STrAtegy for coMmunIty acquired pNeumoniA (STAMINA) study is a randomized, multicenter, open-label trial that compares a driving pressure-limiting strategy to the ARDSnet low-positive end-expiratory pressure table in patients with moderate-to-severe acute respiratory distress syndrome due to community-acquired pneumonia admitted to intensive care units. We expect to recruit 500 patients from 20 Brazilian and 2 Colombian intensive care units. They will be randomized to a driving pressure-limiting strategy group or to a standard strategy using the ARDSNet low-positive end-expiratory pressure table. In the driving pressure-limiting strategy group, positive end-expiratory pressure will be titrated according to the best respiratory system compliance.
The primary outcome is the number of ventilator-free days within 28 days. The secondary outcomes are in-hospital and intensive care unit mortality and the need for rescue therapies such as extracorporeal life support, recruitment maneuvers and inhaled nitric oxide.
STAMINA is designed to provide evidence on whether a driving pressure-limiting strategy is superior to the ARDSNet low-positive end-expiratory pressure table strategy for increasing the number of ventilator-free days within 28 days in patients with moderate-to-severe acute respiratory distress syndrome. Here, we describe the rationale, design and status of the trial.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária - ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Abstract
Rev Bras Ter Intensiva. 2019;31(3):289-295
DOI 10.5935/0103-507X.20190052
To evaluate the effects of ventilator hyperinflation on respiratory mechanics.
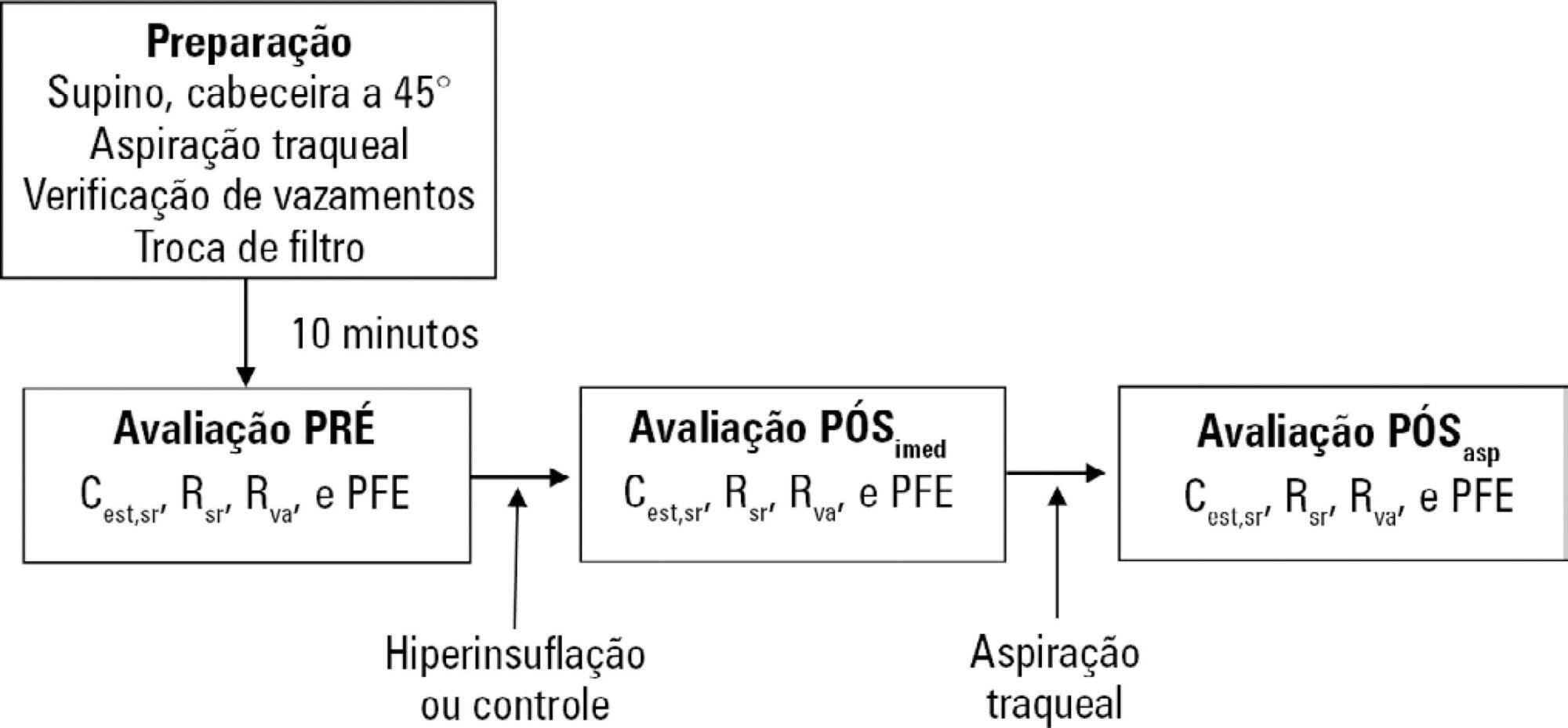
A randomized crossover clinical trial was conducted with 38 mechanically ventilated patients with pulmonary infection. The order of the hyperinflation and control (without changes in the parameters) conditions was randomized. Hyperinflation was performed for 5 minutes in pressure-controlled ventilation mode, with progressive increases of 5cmH2O until a maximum pressure of 35cmH2O was reached, maintaining positive end expiratory pressure. After 35cmH2O was reached, the inspiratory time and respiratory rate were adjusted so that the inspiratory and expiratory flows reached baseline levels. Measurements of static compliance, total resistance and airway resistance, and peak expiratory flow were evaluated before the technique, immediately after the technique and after aspiration. Two-way analysis of variance for repeated measures was used with Tukey's post hoc test, and p < 0.05 was considered significant.
Ventilator hyperinflation increased static compliance, which remained at the same level after aspiration (46.2 ± 14.8 versus 52.0 ± 14.9 versus 52.3 ± 16.0mL/cmH2O; p < 0.001). There was a transient increase in airway resistance (6.6 ± 3.6 versus 8.0 ± 5.5 versus 6.6 ± 3.5cmH2O/Ls-1; p < 0.001) and a transient reduction in peak expiratory flow (32.0 ± 16.0 versus 29.8 ± 14.8 versus 32.1 ± 15.3Lpm; p <0.05) immediately after the technique; these values returned to pretechnique levels after tracheal aspiration. There were no changes in the control condition, nor were hemodynamic alterations observed.
Ventilator hyperinflation promoted increased compliance associated with a transient increase in airway resistance and peak expiratory flow, with reduction after aspiration.
Abstract
Rev Bras Ter Intensiva. 2017;29(2):142-153
DOI 10.5935/0103-507X.20170024
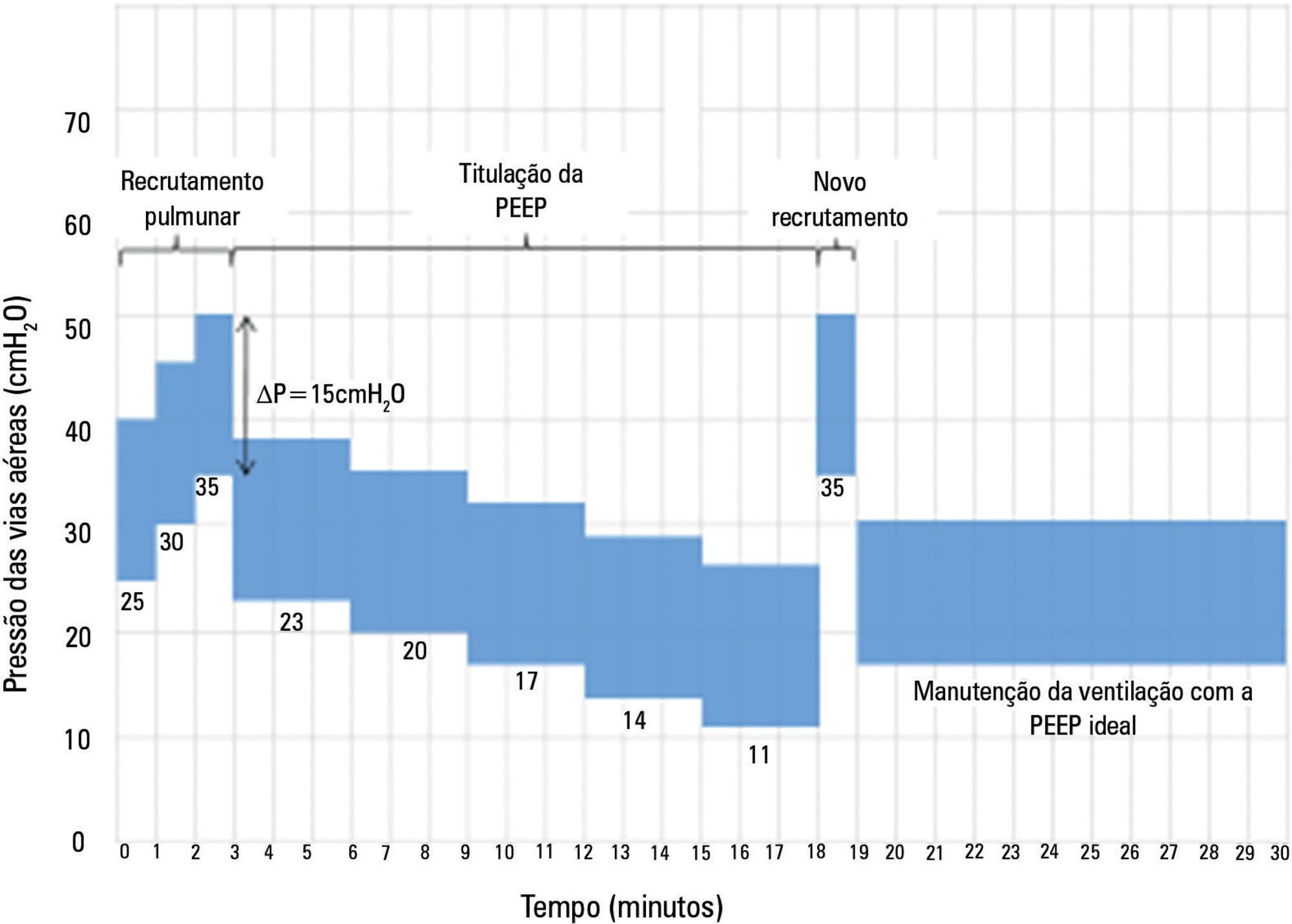
The Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) is an international multicenter randomized pragmatic controlled trial with allocation concealment involving 120 intensive care units in Brazil, Argentina, Colombia, Italy, Poland, Portugal, Malaysia, Spain, and Uruguay. The primary objective of ART is to determine whether maximum stepwise alveolar recruitment associated with PEEP titration, adjusted according to the static compliance of the respiratory system (ART strategy), is able to increase 28-day survival in patients with acute respiratory distress syndrome compared to conventional treatment (ARDSNet strategy).
To describe the data management process and statistical analysis plan.
The statistical analysis plan was designed by the trial executive committee and reviewed and approved by the trial steering committee. We provide an overview of the trial design with a special focus on describing the primary (28-day survival) and secondary outcomes. We describe our data management process, data monitoring committee, interim analyses, and sample size calculation. We describe our planned statistical analyses for primary and secondary outcomes as well as pre-specified subgroup analyses. We also provide details for presenting results, including mock tables for baseline characteristics, adherence to the protocol and effect on clinical outcomes.
According to best trial practice, we report our statistical analysis plan and data management plan prior to locking the database and beginning analyses. We anticipate that this document will prevent analysis bias and enhance the utility of the reported results.
ClinicalTrials.gov number, NCT01374022.
Abstract
Rev Bras Ter Intensiva. 2016;28(3):341-347
DOI 10.5935/0103-507X.20160058
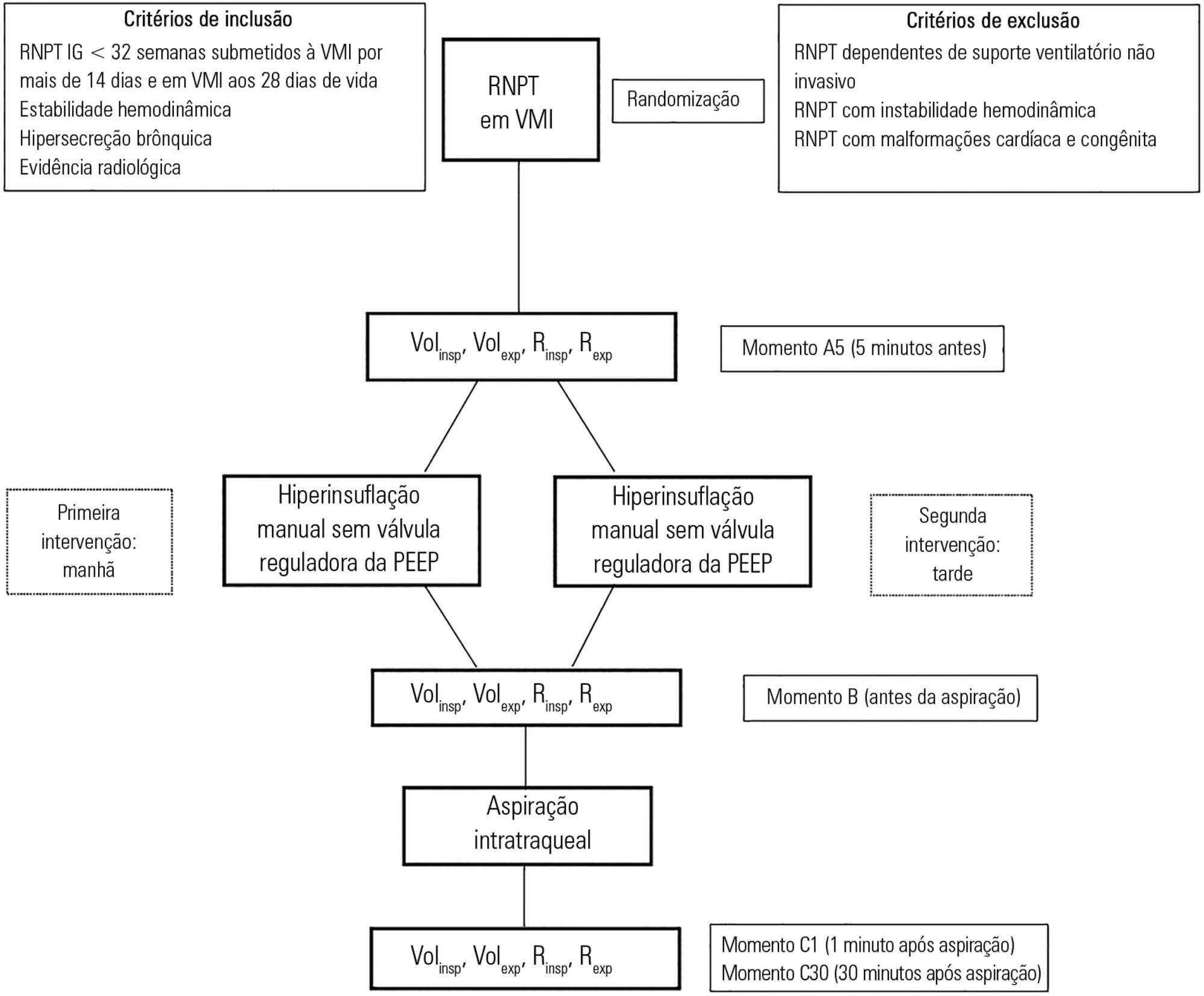
To assess the effects of manual hyperinflation, performed with a manual resuscitator with and without the positive end-expiratory pressure valve, on the respiratory function of preterm newborns under mechanical ventilation.
Cross-sectional study of hemodynamically stable preterm newborns with gestational age of less than 32 weeks, under mechanical ventilation and dependent on it at 28 days of life. Manual hyperinflation was applied randomly, alternating the use or not of the positive end-expiratory pressure valve, followed by tracheal aspiration for ending the maneuver. For nominal data, the two-tailed Wilcoxon test was applied at the 5% significance level and 80% power.
Twenty-eight preterm newborns, with an average birth weight of 1,005.71 ± 372.16g, an average gestational age of 28.90 ± 1.79 weeks, an average corrected age of 33.26 ± 1.78 weeks, and an average mechanical ventilation time of 29.5 (15 - 53) days, were studied. Increases in inspiratory and expiratory volumes occurred between time-points A5 (before the maneuver) and C1 (immediately after tracheal aspiration) in both the maneuver with the valve (p = 0.001 and p = 0.009) and without the valve (p = 0.026 and p = 0.001), respectively. There was also an increase in expiratory resistance between time-points A5 and C1 (p = 0.044).
Lung volumes increased when performing the maneuver with and without the valve, with a significant difference in the first minute after aspiration. There was a significant difference in expiratory resistance between the time-points A5 (before the maneuver) and C1 (immediately after tracheal aspiration) in the first minute after aspiration within each maneuver.
Abstract
Rev Bras Ter Intensiva. 2012;24(1):58-63
DOI 10.1590/S0103-507X2012000100009
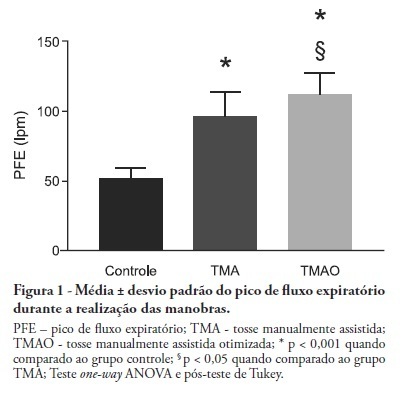
OBJECTIVE: Mechanical ventilation is associated with retained airway secretions. Manually assisted cough contributes to the displacement of bronchial mucus, whereas positive end-expiratory pressure increases collateral ventilation and maintains airway patency. This study aimed to assess the effects of manually assisted cough, either alone or added to increased positive end-expiratory pressure and inspiratory time (optimized manually assisted cough), on the expiratory peak flow and respiratory system mechanics in mechanically ventilated patients. METHODS: In this controlled and randomized clinical trial, respiratory mechanics and expiratory peak flow were assessed in male and female patients undergoing either tracheal suctioning alone, manually assisted cough followed by tracheal suctioning or optimized manually assisted cough followed by tracheal suctioning. RESULTS: Thirty-five patients completed the trial. Respiratory system resistance was significantly reduced after optimized manually assisted cough (16.0 ± 3.6 versus 12.4 ± 3.1 cmH2O/L/s; p = 0.04). The expiratory peak flow during optimized manually assisted cough was significantly higher in comparison with the values observed during manually assisted cough (112.3 ± 15.6 versus 95.8 ± 18.3 Lpm; p < 0.05). Both values were significantly higher than the values observed in the group undergoing tracheal suctioning alone (52.0 ± 7.6 Lpm; p < 0.001). CONCLUSION: Optimized manually assisted cough increases the expiratory peak flow in comparison with manually assisted cough; in addition, this procedure reduces respiratory system resistance.
Abstract
Rev Bras Ter Intensiva. 2011;23(3):291-296
DOI 10.1590/S0103-507X2011000300006
Positive intrathoracic pressure may cause hemodynamic changes, which can be transmitted to the cranial compartment, changing intracranial pressure and cerebral perfusion pressure. This can be increased when high positive end-expiratory pressure values are used. OBJECTIVE: To measure the impact of different positive end-expiratory pressure levels on intracranial pressure, cerebral perfusion pressure and mean blood pressure. METHOD: This study was conducted in a neurological intensive care unit and included 25 adult hemorrhagic stroke patients who were mechanically ventilated on airway pressure control mode. Patients were subjected to various positive end-expiratory values ranging between 0 and 14 cmH2O. The order of these values were randomized, and the variables were assessed five minutes after each new positive end-expiratory pressure level was initiated. RESULTS: Incremental positive end-expiratory pressures led to increased intracranial pressure (p < 0.001), however, no statistically significant changes were observed in mean blood pressure or cerebral perfusion pressure. CONCLUSION: In this population of patients with hemorrhagic stroke, positive end-expiratory pressure values up to 14 cmH2O did not alter cerebral perfusion pressure or mean blood pressure. Increased intracranial pressures were noted, although these elevations were not clinically significant

Abstract
Rev Bras Ter Intensiva. 2010;22(1):40-46
DOI 10.1590/S0103-507X2010000100008
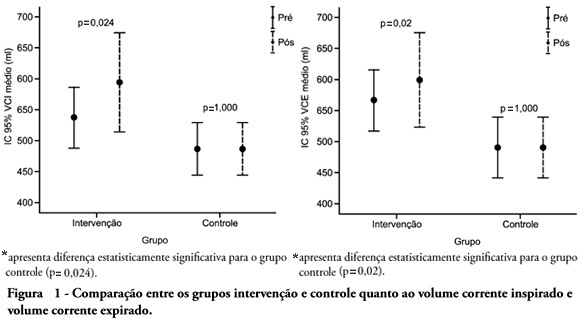
OBJECTIVE: To verify the effects of manual hyperinflation maneuver associated with positive end expiratory pressure in coronary artery bypass grafting patients. METHODS: This was a randomized trial, conducted from August 2007 to July 2008 in the intensive care unit of the Hospital Luterano (ULBRA). The patients were divided in the groups intervention - with manual hyperinflation plus positive end expiratory pressure - and controlThe ventilatory variables were measured before and after the manual hyperinflation. The t Student's test was used for independent and paired samples as well as Fisher's exact test and McNemar's Chi-square test with 0.05 significance level. RESULTS: Eighteen patients were included. The mean age was 64± 11 years and 55.6% were female. The inspired tidal volume was 594± 112ml in the intervention group and 487± 51ml in the control group (p=0.024) and the expired tidal volume was 598± 105ml in the intervention group and 490± 58ml in the control group (p=0.02). The mean pre-maneuver static pulmonary compliance in the intervention group was 41.6± 12.1 ml/cmH2O and post maneuver it was 47.4± 16.6 ml/cmH2O (p=0.03). There was no significant between groups difference in the following variables: oxygen peripheral saturation, oxygen arterial pressure, extubation time and radiological changes. CONCLUSION: The results show that the manual hyperinflation associated with positive end expiratory pressure maneuver trends to promote increased lung volumes and static compliance, however these findings require further confirmation.
Search
Search in:
Case reports (56) Child (53) Coronavirus infections (34) COVID-19 (46) Critical care (115) Critical illness (54) Extracorporeal membrane oxygenation (26) Infant, newborn (27) Intensive care (72) Intensive care units (256) Intensive care units, pediatric (31) mechanical ventilation (38) Mortality (76) Physical therapy modalities (28) Prognosis (61) Respiration, artificial (119) Respiratory insufficiency (26) risk factors (34) SARS-CoV-2 (28) Sepsis (98)