You searched for:"Fernando Godinho Zampieri"
We found (27) results for your search.-
Special Article
Dapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
Abstract
Special ArticleDapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
DOI 10.5935/2965-2774.20230129-pt
Views9ABSTRACT
Background:
Critical illness is a major ongoing health care burden worldwide and is associated with high mortality rates. Sodium-glucose cotransporter-2 inhibitors have consistently shown benefits in cardiovascular and renal outcomes. The effects of sodium-glucose cotransporter-2 inhibitors in acute illness have not been properly investigated.
Methods:
DEFENDER is an investigator-initiated, multicenter, randomized, open-label trial designed to evaluate the efficacy and safety of dapagliflozin in 500 adult participants with acute organ dysfunction who are hospitalized in the intensive care unit. Eligible participants will be randomized 1:1 to receive dapagliflozin 10mg plus standard of care for up to 14 days or standard of care alone. The primary outcome is a hierarchical composite of hospital mortality, initiation of kidney replacement therapy, and intensive care unit length of stay, up to 28 days. Safety will be strictly monitored throughout the study.
Conclusion:
DEFENDER is the first study designed to investigate the use of a sodium-glucose cotransporter-2 inhibitor in general intensive care unit patients with acute organ dysfunction. It will provide relevant information on the use of drugs of this promising class in critically ill patients.
ClinicalTrials.gov registry:
NCT05558098
Keywords:Critical care outcomesCritical illnessOrgan dysfunctionSodium-glucose transporter 2 inhibitorsSee more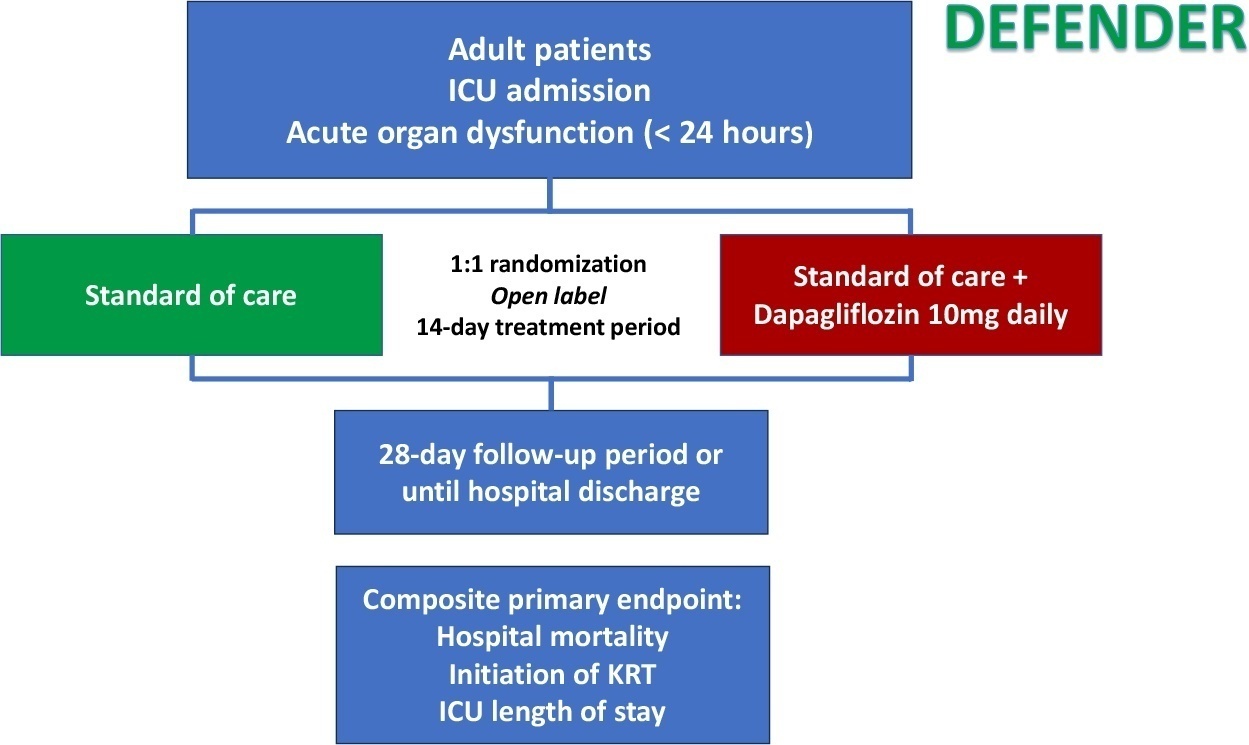
-
Letter to the Editor
Acute kidney injury in hospitalized patients with COVID-19: a retrospective cohort
Crit Care Sci. 2023;35(2):236-238
Abstract
Letter to the EditorAcute kidney injury in hospitalized patients with COVID-19: a retrospective cohort
Crit Care Sci. 2023;35(2):236-238
DOI 10.5935/2965-2774.20230428-pt
Views4TO THE EDITORCoronavirus disease 2019 (COVID-19) has been reported to cause acute kidney injury (AKI).(-) Although severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) may directly harm the kidneys through endothelial and coagulation dysfunction,() AKI in COVID-19 may also be related to additional organ dysfunctions and other host factors, including mechanical ventilation. The incidence of AKI […]See more
-
Original Article
Effects of balanced solution on short-term outcomes in traumatic brain injury patients: a secondary analysis of the BaSICS randomized trial
Rev Bras Ter Intensiva. 2022;34(4):410-417
Abstract
Original ArticleEffects of balanced solution on short-term outcomes in traumatic brain injury patients: a secondary analysis of the BaSICS randomized trial
Rev Bras Ter Intensiva. 2022;34(4):410-417
DOI 10.5935/0103-507X.20220261-en
Views2ABSTRACT
Objective:
To describe the effects of balanced solution use on the short-term outcomes of patients with traumatic brain injury enrolled in BaSICS trial.
Methods:
Patients were randomized to receive either 0.9% saline or balanced solution during their intensive care unit stay. The primary endpoint was 90-day mortality, and the secondary outcomes were days alive and free of intensive care unit stay at 28 days. The primary endpoint was assessed using Bayesian logistic regression. The secondary endpoint was assessed using a Bayesian zero-inflated beta binomial regression.
Results:
We included 483 patients (236 in the 0.9% saline arm and 247 in the balanced solution arm). A total of 338 patients (70%) with a Glasgow coma scale score ≤ 12 were enrolled. The overall probability that balanced solutions were associated with higher 90-day mortality was 0.98 (OR 1.48; 95%CrI 1.04 – 2.09); this mortality increment was particularly noticeable in patients with a Glasgow coma scale score below 6 at enrollment (probability of harm of 0.99). Balanced solutions were associated with -1.64 days alive and free of intensive care unit at 28 days (95%CrI -3.32 – 0.00) with a probability of harm of 0.97.
Conclusion:
There was a high probability that balanced solutions were associated with high 90-day mortality and fewer days alive and free of intensive care units at 28 days.
Keywords:Balanced solutionsBrain injuriesCritical careHospital mortalityMortalitySaline solutiontraumaticSee more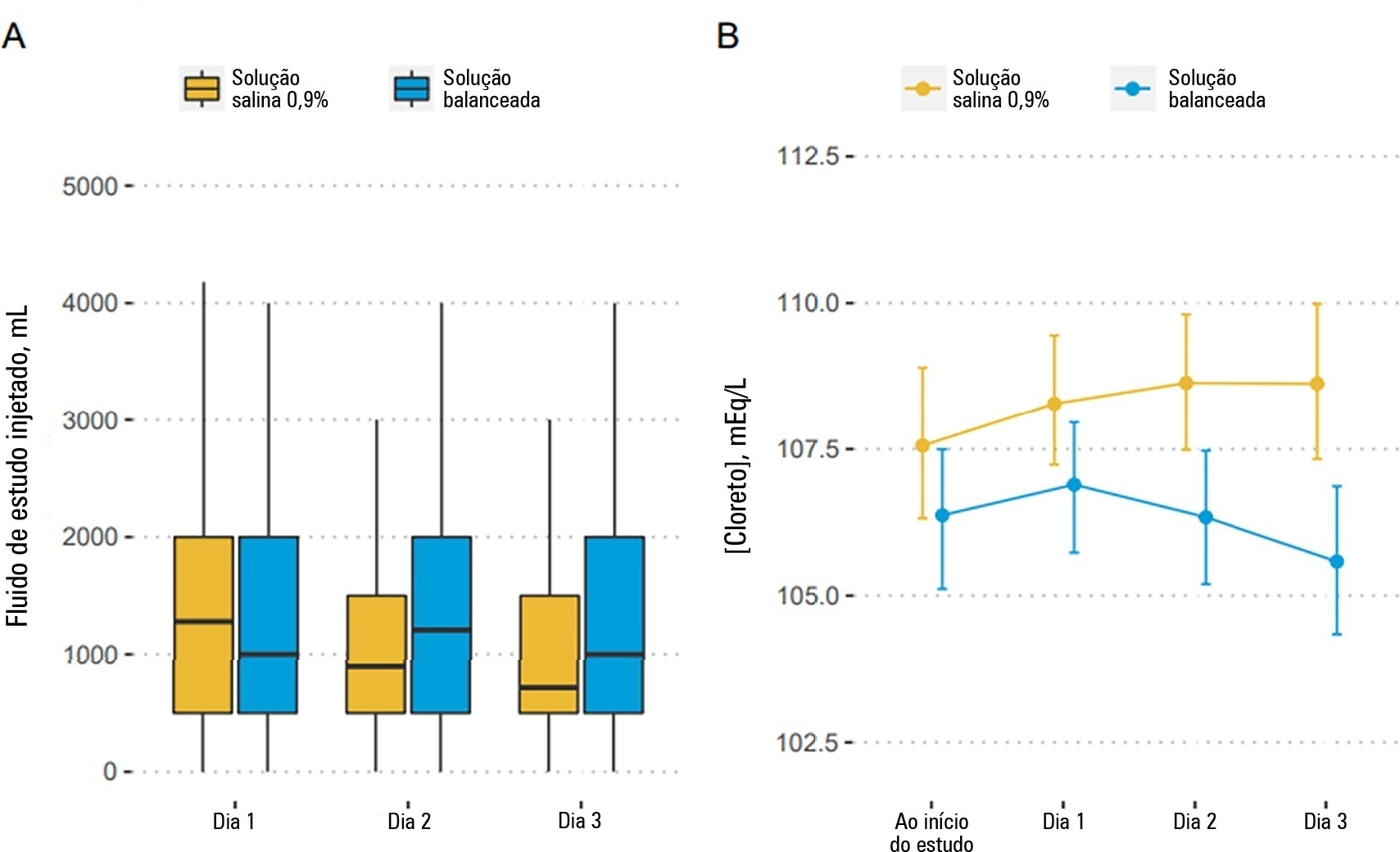
-
Original Article
Clinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
Abstract
Original ArticleClinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
DOI 10.5935/0103-507X.20220040-en
Views2ABSTRACT
Objective:
To compare the lung mechanics and outcomes between COVID-19-associated acute respiratory distress syndrome and non-COVID-19-associated acute respiratory distress syndrome.
Methods:
We combined data from two randomized trials in acute respiratory distress syndrome, one including only COVID-19 patients and the other including only patients without COVID-19, to determine whether COVID-19-associated acute respiratory distress syndrome is associated with higher 28-day mortality than non-COVID-19 acute respiratory distress syndrome and to examine the differences in lung mechanics between these two types of acute respiratory distress syndrome.
Results:
A total of 299 patients with COVID-19-associated acute respiratory distress syndrome and 1,010 patients with non-COVID-19-associated acute respiratory distress syndrome were included in the main analysis. The results showed that non-COVID-19 patients used higher positive end-expiratory pressure (12.5cmH2O; SD 3.2 versus 11.7cmH2O SD 2.8; p < 0.001), were ventilated with lower tidal volumes (5.8mL/kg; SD 1.0 versus 6.5mL/kg; SD 1.2; p < 0.001) and had lower static respiratory compliance adjusted for ideal body weight (0.5mL/cmH2O/kg; SD 0.3 versus 0.6mL/cmH2O/kg; SD 0.3; p = 0.01). There was no difference between groups in 28-day mortality (52.3% versus 58.9%; p = 0.52) or mechanical ventilation duration in the first 28 days among survivors (13 [IQR 5 – 22] versus 12 [IQR 6 – 26], p = 0.46).
Conclusion:
This analysis showed that patients with non-COVID-19-associated acute respiratory distress syndrome have different lung mechanics but similar outcomes to COVID-19-associated acute respiratory distress syndrome patients. After propensity score matching, there was no difference in lung mechanics or outcomes between groups.
Keywords:Coronavirus infectionsCOVID-19Critical careCritical care outcomesRespiratory distress syndromeRespiratory mechanicsSee more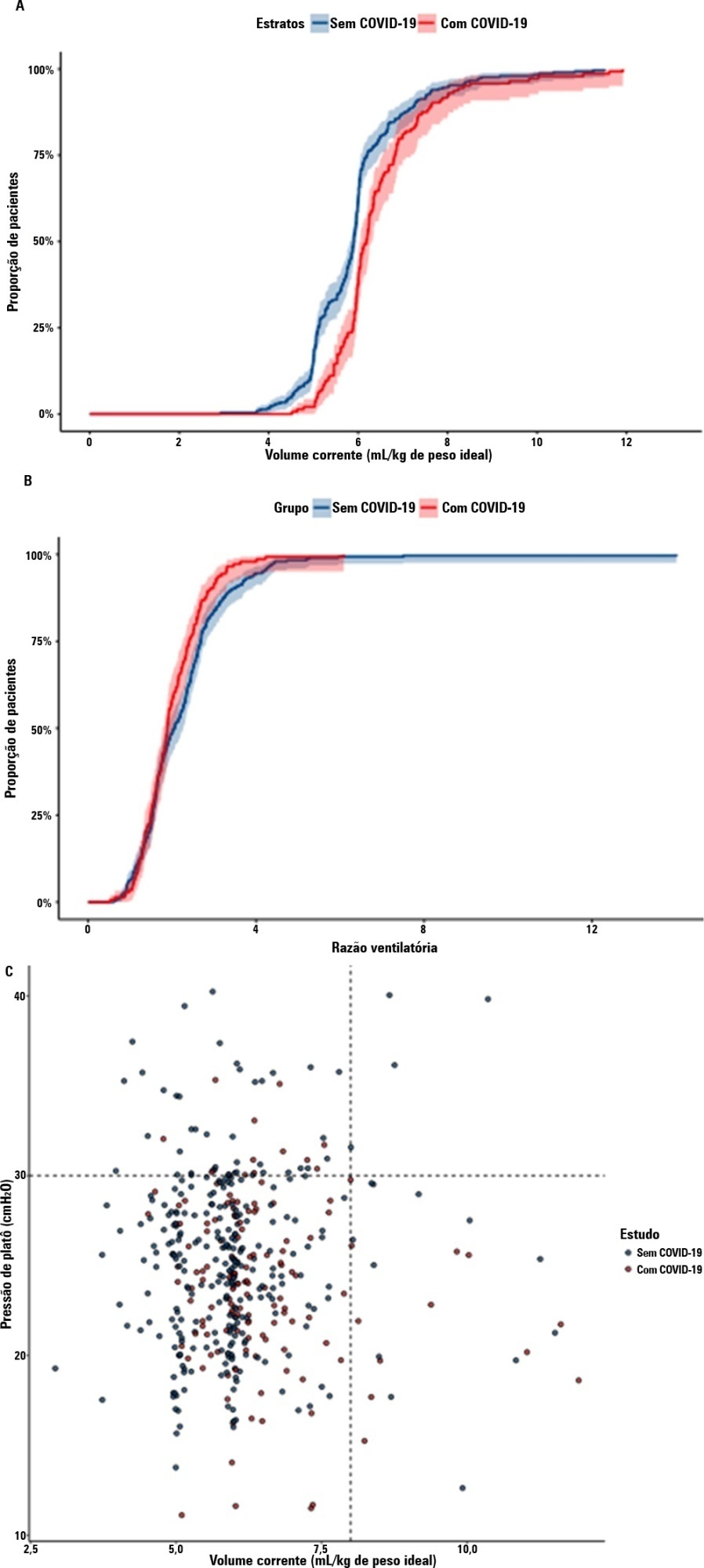
-
Original Article
Performance status and acute organ dysfunction influence hospital mortality in critically ill patients with cancer and suspected infection: a retrospective cohort analysis
Rev Bras Ter Intensiva. 2021;33(2):298-303
Abstract
Original ArticlePerformance status and acute organ dysfunction influence hospital mortality in critically ill patients with cancer and suspected infection: a retrospective cohort analysis
Rev Bras Ter Intensiva. 2021;33(2):298-303
DOI 10.5935/0103-507X.20210038
Views1See moreABSTRACT
Objective:
To evaluate how performance status impairment and acute organ dysfunction influence hospital mortality in critically ill patients with cancer who were admitted with suspected sepsis.
Methods:
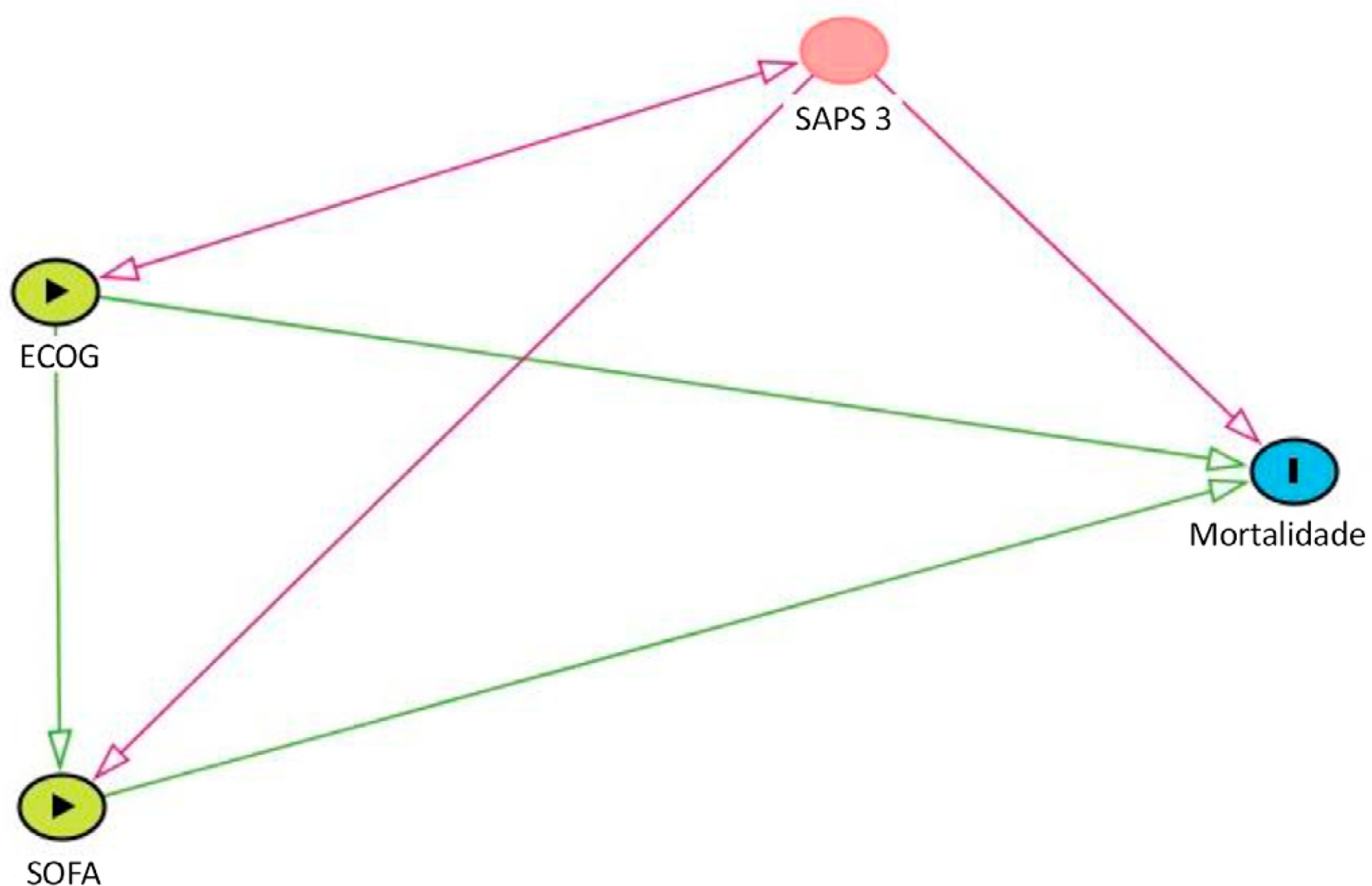
Data were obtained from a retrospective cohort of patients, admitted to an intensive care unit, with cancer and with a suspected infection who received parenteral antibiotics and underwent the collection of bodily fluid samples. We used logistic regression with hospital mortality as the outcome and the Sequential Organ Failure Assessment score, Eastern Cooperative Oncology Group status, and their interactions as predictors.
Results:
Of 450 patients included, 265 (58.9%) died in the hospital. For patients admitted to the intensive care unit with lower Sequential Organ Failure Assessment (≤ 6), performance status impairment influenced the in-hospital mortality, which was 32% among those with no and minor performance status impairment and 52% among those with moderate and severe performance status impairment, p < 0.01. However, for those with higher Sequential Organ Failure Assessment (> 6), performance status impairment did not influence the in-hospital mortality (73% among those with no and minor impairment and 84% among those with moderate and severe impairment; p = 0.1).
Conclusion:
Performance status impairment seems to influence hospital mortality in critically ill cancer patients with suspected sepsis when they have less severe acute organ dysfunction at the time of intensive care unit admission.
-
Special Article
Quality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
Abstract
Special ArticleQuality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
Views0See moreAbstract
Introduction:
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
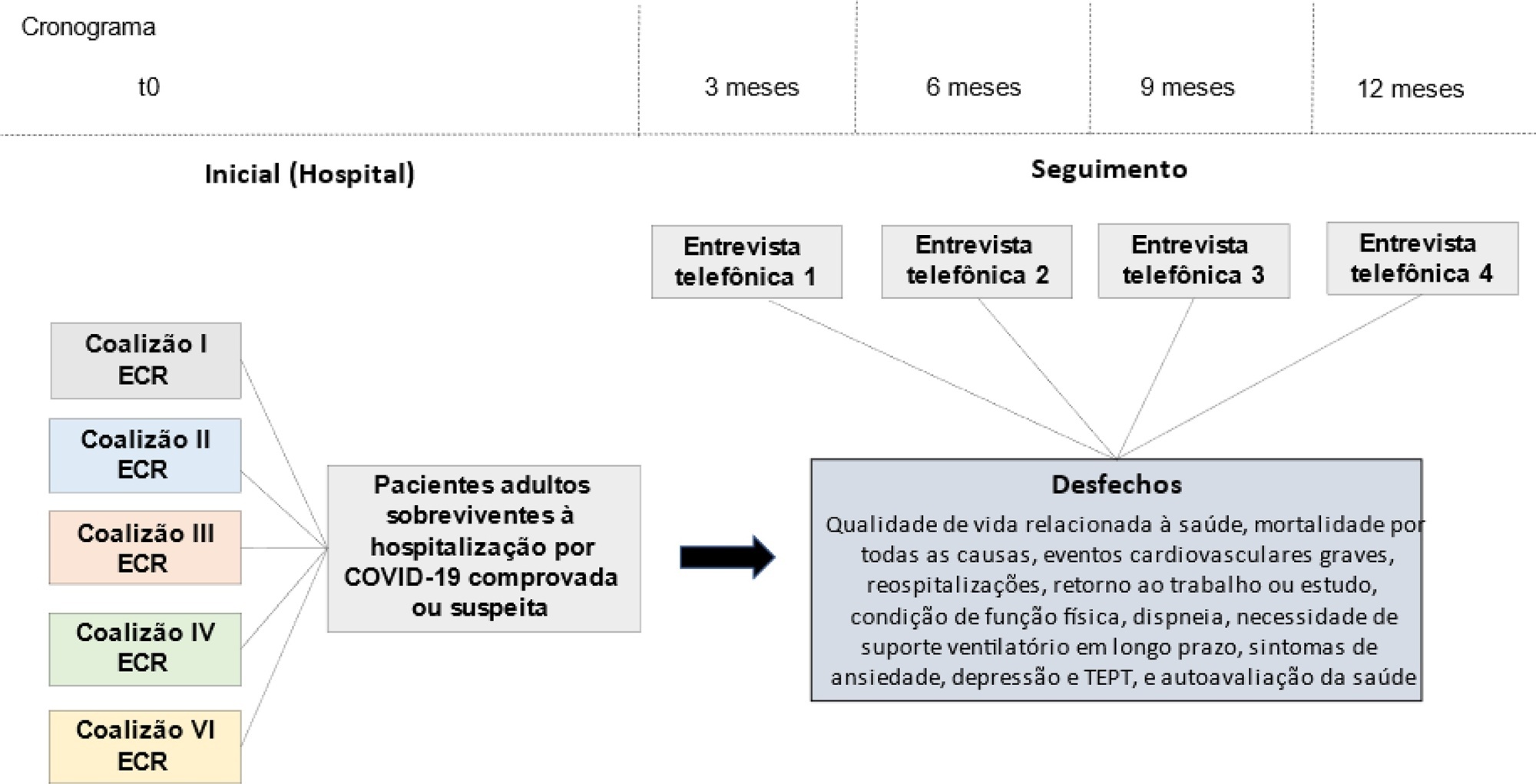
Methods:
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
Ethics and dissemination:
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
-
Original Article
Statistical analysis plan for the Balanced Solution versus Saline in Intensive Care Study (BaSICS)
Rev Bras Ter Intensiva. 2020;32(4):493-505
Abstract
Original ArticleStatistical analysis plan for the Balanced Solution versus Saline in Intensive Care Study (BaSICS)
Rev Bras Ter Intensiva. 2020;32(4):493-505
DOI 10.5935/0103-507X.20200081
Views1See moreAbstract
Objective:
To report the statistical analysis plan (first version) for the Balanced Solutions versus Saline in Intensive Care Study (BaSICS).
Methods:
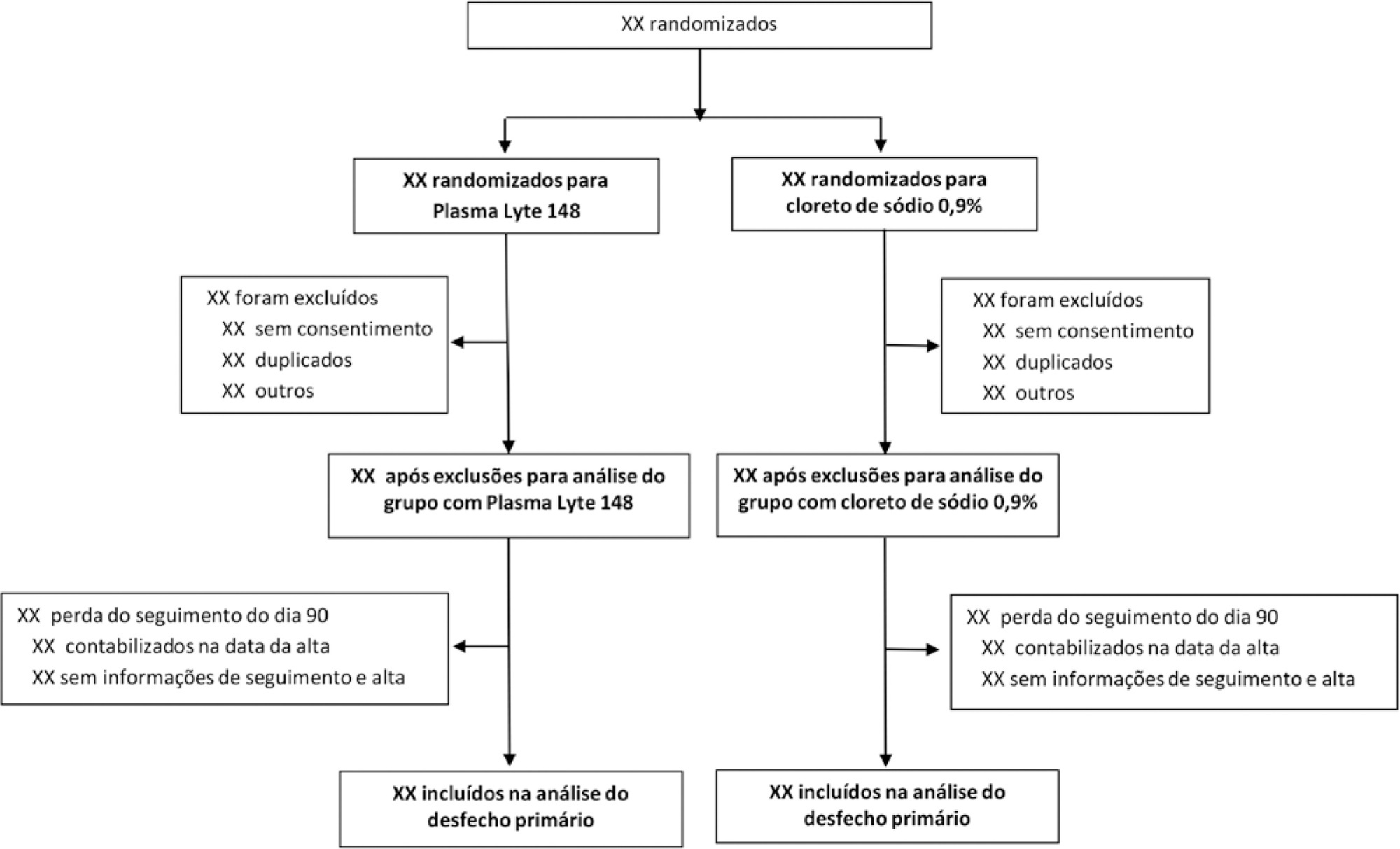
BaSICS is a multicenter factorial randomized controlled trial that will assess the effects of Plasma-Lyte 148 versus 0.9% saline as the fluid of choice in critically ill patients, as well as the effects of a slow (333mL/h) versus rapid (999mL/h) infusion speed during fluid challenges, on important patient outcomes. The fluid type will be blinded for investigators, patients and the analyses. No blinding will be possible for the infusion speed for the investigators, but all analyses will be kept blinded during the analysis procedure.
Results:
BaSICS will have 90-day mortality as its primary endpoint, which will be tested using mixed-effects Cox proportional hazard models, considering sites as a random variable (frailty models) adjusted for age, organ dysfunction and admission type. Important secondary endpoints include renal replacement therapy up to 90 days, acute renal failure, organ dysfunction at days 3 and 7, and mechanical ventilation-free days within 28 days.
Conclusion:
This manuscript provides details on the first version of the statistical analysis plan for the BaSICS trial and will guide the study’s analysis when follow-up is finished.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness ICU Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis