You searched for:"Glauco Adrieno Westphal"
We found (27) results for your search.-
Clinical Report
Prospective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
Abstract
Clinical ReportProspective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
DOI 10.62675/2965-2774.20240210-en
Views72ABSTRACT
Background:
Driving pressure has been suggested to be the main driver of ventilator-induced lung injury and mortality in observational studies of acute respiratory distress syndrome. Whether a driving pressure-limiting strategy can improve clinical outcomes is unclear.
Objective:
To describe the protocol and statistical analysis plan that will be used to test whether a driving pressure-limiting strategy including positive end-expiratory pressure titration according to the best respiratory compliance and reduction in tidal volume is superior to a standard strategy involving the use of the ARDSNet low-positive end-expiratory pressure table in terms of increasing the number of ventilator-free days in patients with acute respiratory distress syndrome due to community-acquired pneumonia.
Methods:
The ventilator STrAtegy for coMmunIty acquired pNeumoniA (STAMINA) study is a randomized, multicenter, open-label trial that compares a driving pressure-limiting strategy to the ARDSnet low-positive end-expiratory pressure table in patients with moderate-to-severe acute respiratory distress syndrome due to community-acquired pneumonia admitted to intensive care units. We expect to recruit 500 patients from 20 Brazilian and 2 Colombian intensive care units. They will be randomized to a driving pressure-limiting strategy group or to a standard strategy using the ARDSNet low-positive end-expiratory pressure table. In the driving pressure-limiting strategy group, positive end-expiratory pressure will be titrated according to the best respiratory system compliance.
Outcomes:
The primary outcome is the number of ventilator-free days within 28 days. The secondary outcomes are in-hospital and intensive care unit mortality and the need for rescue therapies such as extracorporeal life support, recruitment maneuvers and inhaled nitric oxide.
Conclusion:
STAMINA is designed to provide evidence on whether a driving pressure-limiting strategy is superior to the ARDSNet low-positive end-expiratory pressure table strategy for increasing the number of ventilator-free days within 28 days in patients with moderate-to-severe acute respiratory distress syndrome. Here, we describe the rationale, design and status of the trial.
Keywords:Extracorporeal membrane oxygenationPneumoniaPositive pressure respirationRespiration, artificialRespiratory distress syndromeVentilator-induced lung injurySee more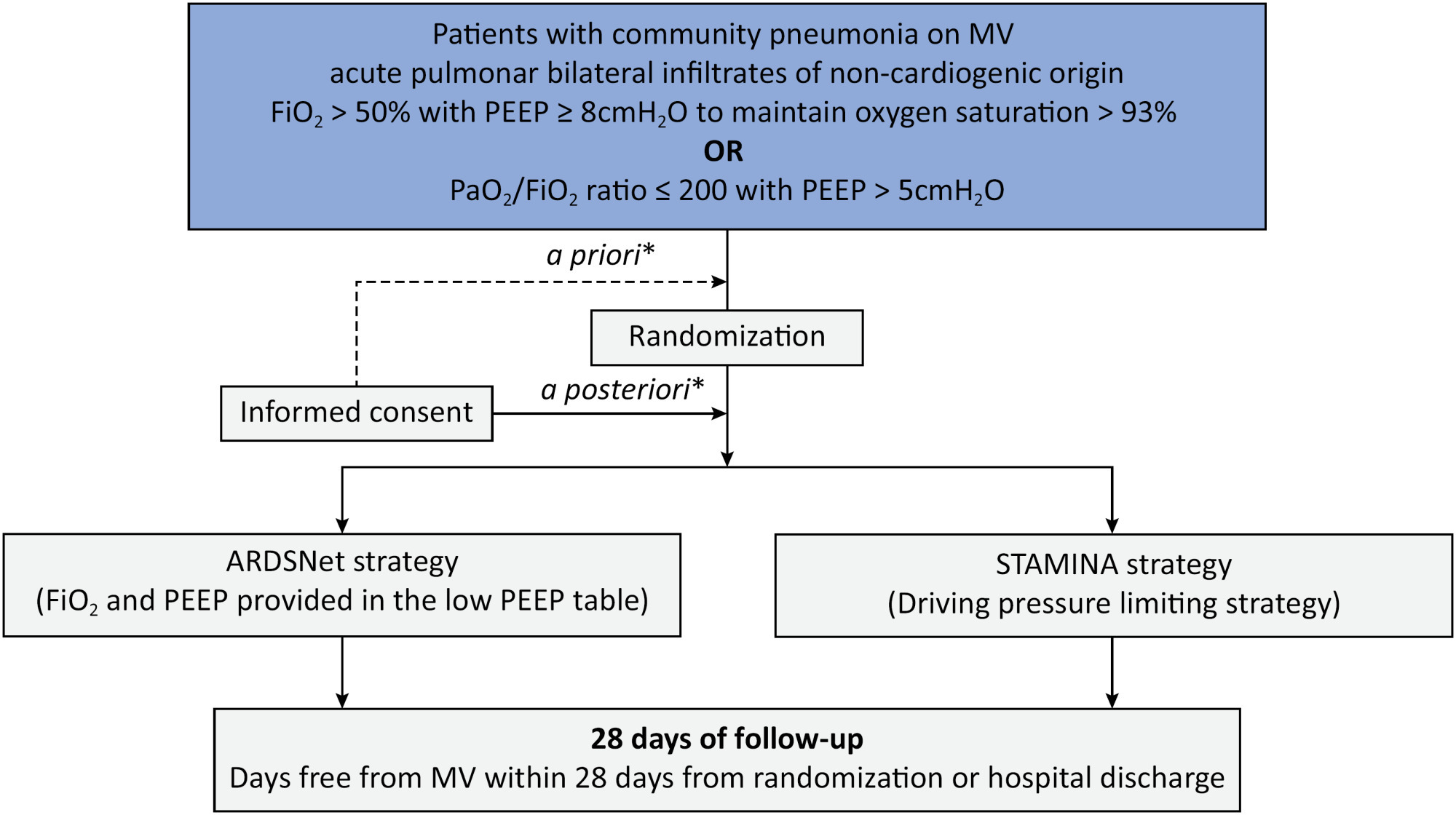
-
Special Article
Dapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
Abstract
Special ArticleDapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
DOI 10.5935/2965-2774.20230129-pt
Views9ABSTRACT
Background:
Critical illness is a major ongoing health care burden worldwide and is associated with high mortality rates. Sodium-glucose cotransporter-2 inhibitors have consistently shown benefits in cardiovascular and renal outcomes. The effects of sodium-glucose cotransporter-2 inhibitors in acute illness have not been properly investigated.
Methods:
DEFENDER is an investigator-initiated, multicenter, randomized, open-label trial designed to evaluate the efficacy and safety of dapagliflozin in 500 adult participants with acute organ dysfunction who are hospitalized in the intensive care unit. Eligible participants will be randomized 1:1 to receive dapagliflozin 10mg plus standard of care for up to 14 days or standard of care alone. The primary outcome is a hierarchical composite of hospital mortality, initiation of kidney replacement therapy, and intensive care unit length of stay, up to 28 days. Safety will be strictly monitored throughout the study.
Conclusion:
DEFENDER is the first study designed to investigate the use of a sodium-glucose cotransporter-2 inhibitor in general intensive care unit patients with acute organ dysfunction. It will provide relevant information on the use of drugs of this promising class in critically ill patients.
ClinicalTrials.gov registry:
NCT05558098
Keywords:Critical care outcomesCritical illnessOrgan dysfunctionSodium-glucose transporter 2 inhibitorsSee more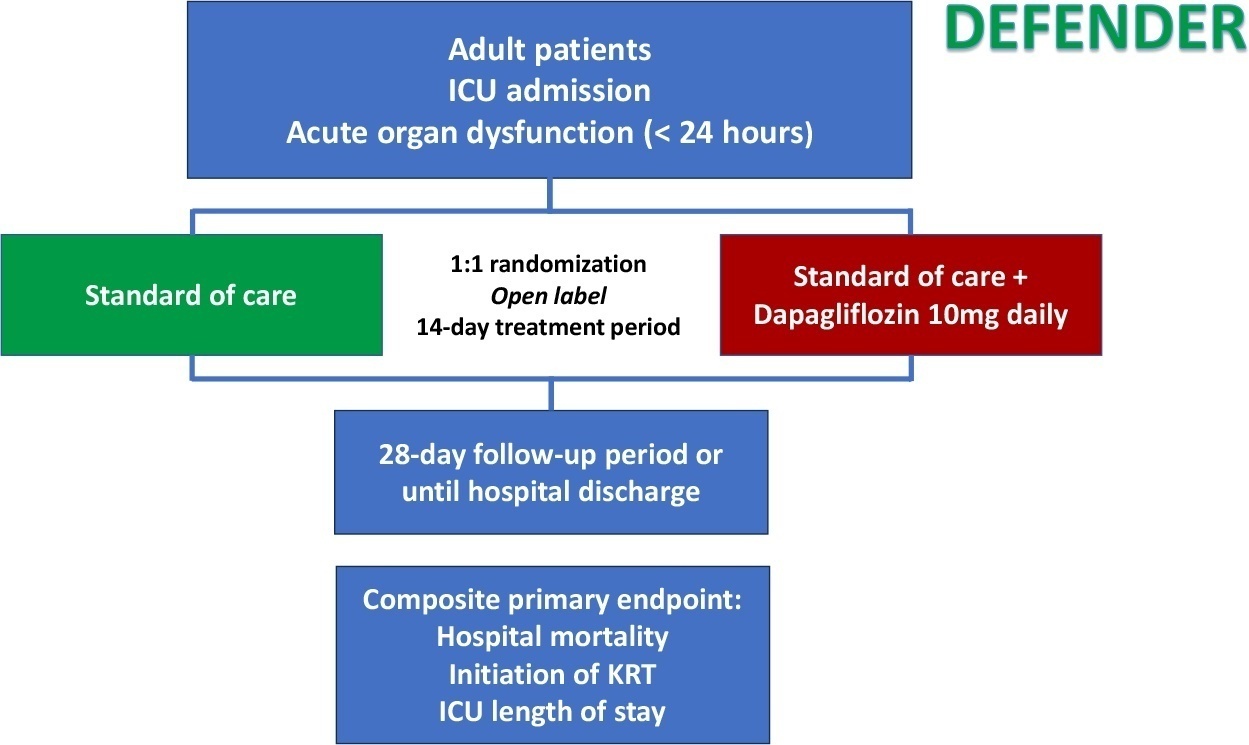
-
Original Article
Resuscitation fluid practices in Brazilian intensive care units: a secondary analysis of Fluid-TRIPS
Rev Bras Ter Intensiva. 2021;33(2):206-218
Abstract
Original ArticleResuscitation fluid practices in Brazilian intensive care units: a secondary analysis of Fluid-TRIPS
Rev Bras Ter Intensiva. 2021;33(2):206-218
DOI 10.5935/0103-507X.20210028
Views1See moreAbstract
Objective:
To describe fluid resuscitation practices in Brazilian intensive care units and to compare them with those of other countries participating in the Fluid-TRIPS.
Methods:
This was a prospective, international, cross-sectional, observational study in a convenience sample of intensive care units in 27 countries (including Brazil) using the Fluid-TRIPS database compiled in 2014. We described the patterns of fluid resuscitation use in Brazil compared with those in other countries and identified the factors associated with fluid choice.
Results:
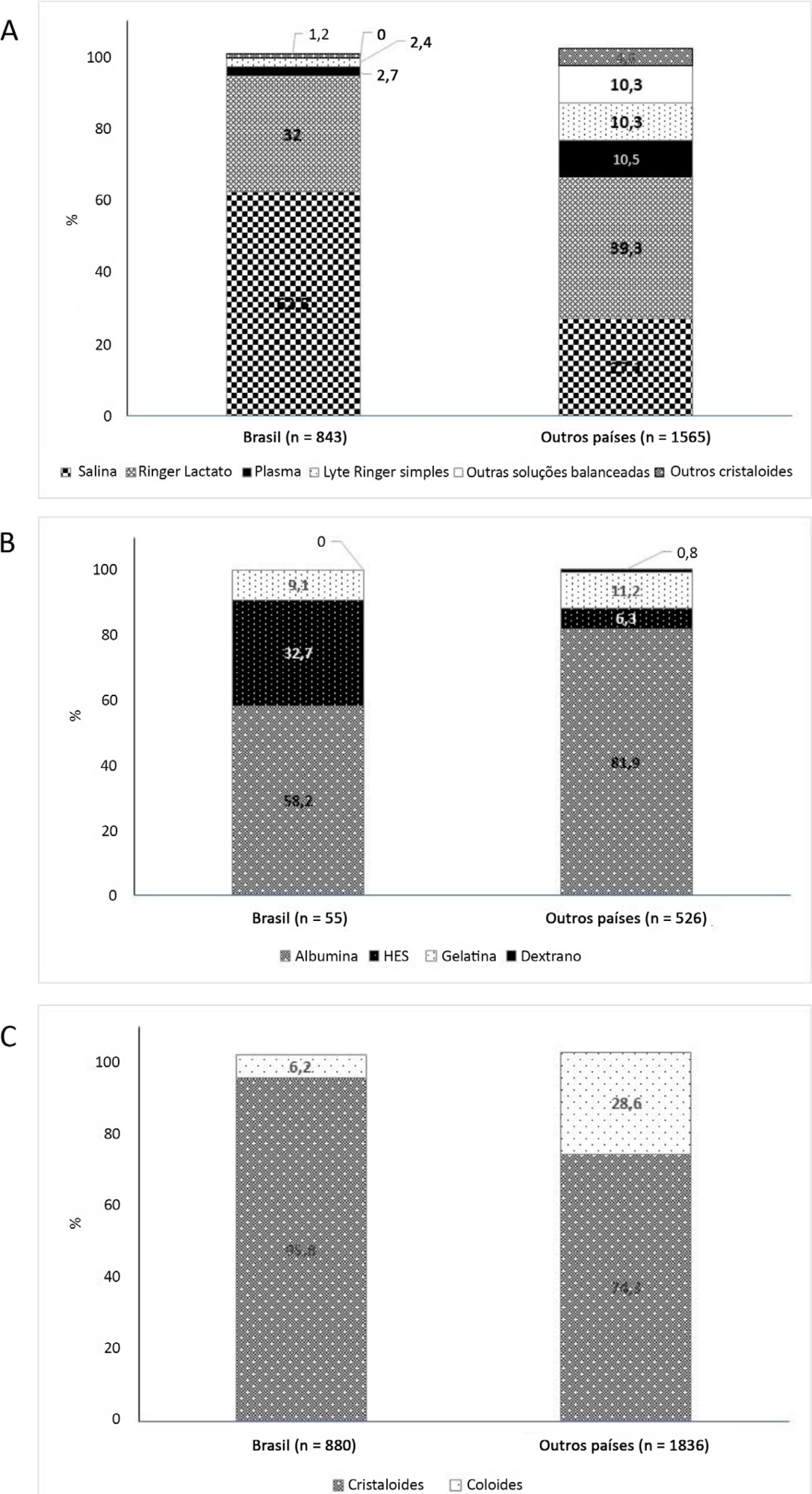
On the study day, 3,214 patients in Brazil and 3,493 patients in other countries were included, of whom 16.1% and 26.8% (p < 0.001) received fluids, respectively. The main indication for fluid resuscitation was impaired perfusion and/or low cardiac output (Brazil: 71.7% versus other countries: 56.4%, p < 0.001). In Brazil, the percentage of patients receiving crystalloid solutions was higher (97.7% versus 76.8%, p < 0.001), and 0.9% sodium chloride was the most commonly used crystalloid (62.5% versus 27.1%, p < 0.001). The multivariable analysis suggested that the albumin levels were associated with the use of both crystalloids and colloids, whereas the type of fluid prescriber was associated with crystalloid use only.
Conclusion:
Our results suggest that crystalloids are more frequently used than colloids for fluid resuscitation in Brazil, and this discrepancy in frequencies is higher than that in other countries. Sodium chloride (0.9%) was the crystalloid most commonly prescribed. Serum albumin levels and the type of fluid prescriber were the factors associated with the choice of crystalloids or colloids for fluid resuscitation.
-
Special Article
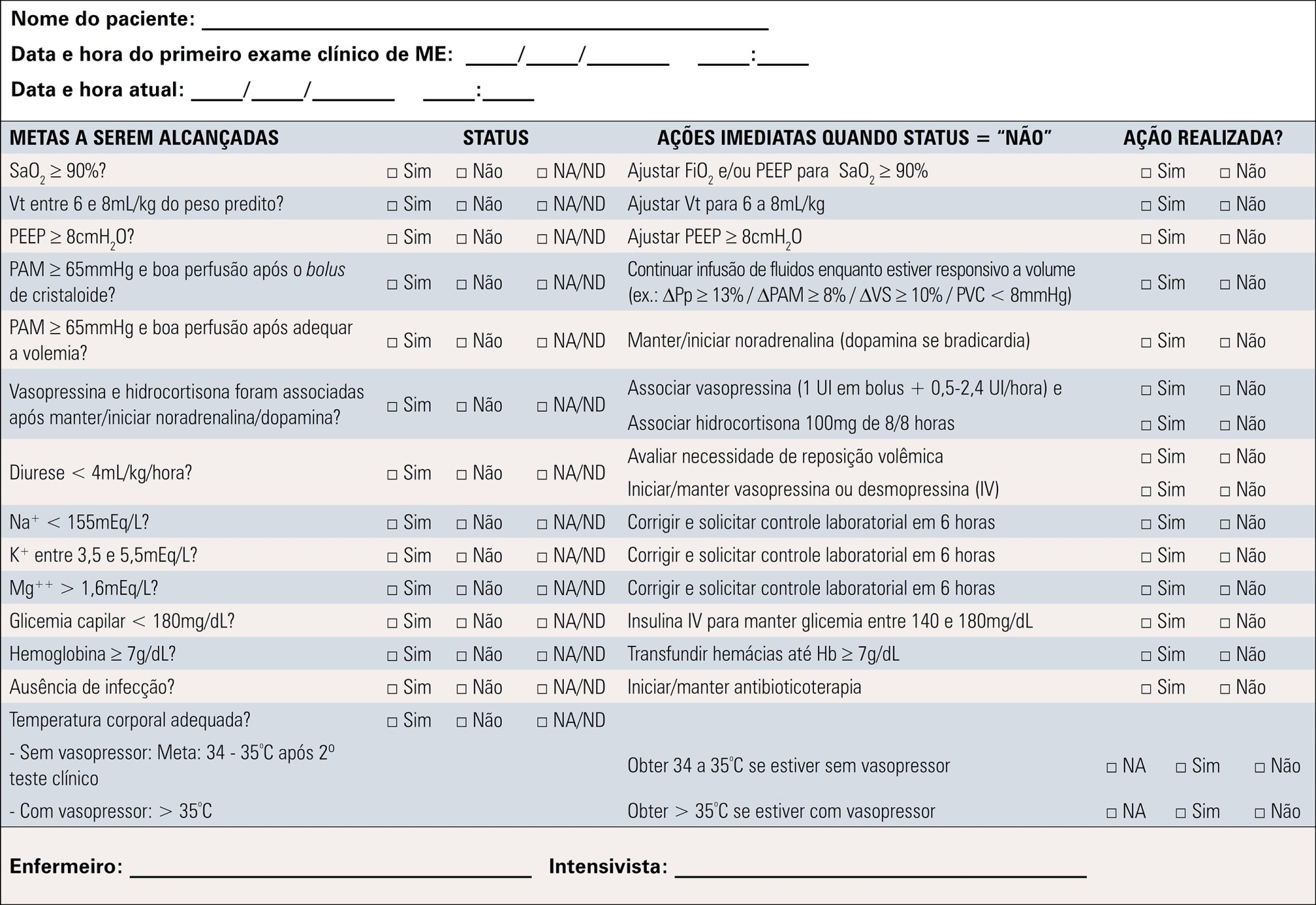
Brazilian guidelines for the management of brain-dead potential organ donors. The task force of the Associação de Medicina Intensiva Brasileira, Associação Brasileira de Transplantes de Órgãos, Brazilian Research in Critical Care Network, and the General Coordination of the National Transplant System
Rev Bras Ter Intensiva. 2021;33(1):1-11
Abstract
Special ArticleBrazilian guidelines for the management of brain-dead potential organ donors. The task force of the Associação de Medicina Intensiva Brasileira, Associação Brasileira de Transplantes de Órgãos, Brazilian Research in Critical Care Network, and the General Coordination of the National Transplant System
Rev Bras Ter Intensiva. 2021;33(1):1-11
DOI 10.5935/0103-507X.20210001
Views5See moreAbstract
Objective:
To contribute to updating the recommendations for brain-dead potential organ donor management.
Methods:
A group of 27 experts, including intensivists, transplant coordinators, transplant surgeons, and epidemiologists, answered questions related to the following topics were divided into mechanical ventilation, hemodynamics, endocrine-metabolic management, infection, body temperature, blood transfusion, and checklists use. The outcomes considered were cardiac arrests, number of organs removed or transplanted as well as function / survival of transplanted organs. The quality of evidence of the recommendations was assessed using the Grading of Recommendations Assessment, Development, and Evaluation system to classify the recommendations.
Results:
A total of 19 recommendations were drawn from the expert panel. Of these, 7 were classified as strong, 11 as weak and 1 was considered a good clinical practice.
Conclusion:
Despite the agreement among panel members on most recommendations, the grade of recommendation was mostly weak.
-
Original Article
Can the behavior of blood pressure after elevation of the positive end-expiratory pressure help to determine the fluid responsiveness status in patients with septic shock?
Rev Bras Ter Intensiva. 2020;32(3):374-380
Abstract
Original ArticleCan the behavior of blood pressure after elevation of the positive end-expiratory pressure help to determine the fluid responsiveness status in patients with septic shock?
Rev Bras Ter Intensiva. 2020;32(3):374-380
DOI 10.5935/0103-507X.20200065
Views0Abstract
Objective:
To evaluate whether the decrease in blood pressure caused by the increase in the positive end-expiratory pressure corresponds to the pulse pressure variation as an indicator of fluid responsiveness.
Methods:
This exploratory study prospectively included 24 patients with septic shock who were mechanically ventilated and subjected to three stages of elevation of the positive end-expiratory pressure: from 5 to 10cmH2O (positive end-expiratory pressure level 1), from 10 to 15cmH2O (positive end-expiratory pressure level 2), and from 15 to 20cmH2O (positive end-expiratory pressure level 3). Changes in systolic blood pressure, mean arterial pressure, and pulse pressure variation were evaluated during the three maneuvers. The patients were classified as responsive (pulse pressure variation ≥ 12%) or unresponsive to volume replacement (pulse pressure variation < 12%).
Results:
The best performance at identifying patients with pulse pressure variation ≥ 12% was observed at the positive end-expiratory pressure level 2: -9% systolic blood pressure variation (area under the curve 0.73; 95%CI: 0.49 – 0.79; p = 0.04), with a sensitivity of 63% and specificity of 80%. Concordance was low between the variable with the best performance (variation in systolic blood pressure) and pulse pressure variation ≥ 12% (kappa = 0.42; 95%CI: 0.19 – 0.56). The systolic blood pressure was < 90mmHg at positive end-expiratory pressure level 2 in 29.2% of cases and at positive end-expiratory pressure level 3 in 41.63% of cases.
Conclusion:
Variations in blood pressure in response to the increase in positive end-expiratory pressure do not reliably reflect the behavior of the pulse pressure as a measure to identify the fluid responsiveness status.
Keywords:Arterial pressure/physiologyFluid responsivenessIntensive care unitsPositive end-expiratory pressurePulse pressure respiratory variationShock, septicSee more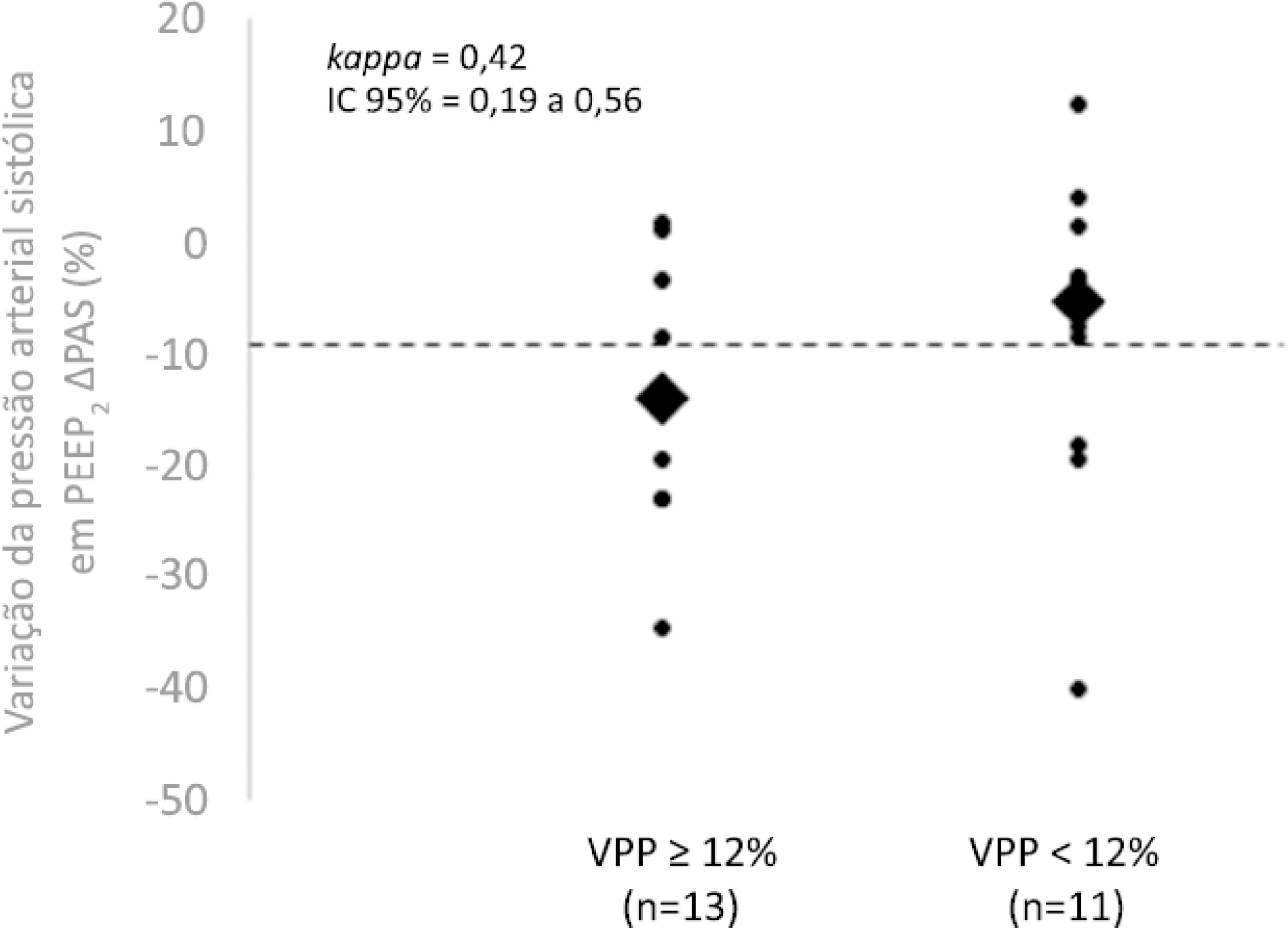
-
Commentary
Shared decision-making in the context of COVID-19
Rev Bras Ter Intensiva. 2020;32(2):200-202
Abstract
CommentaryShared decision-making in the context of COVID-19
Rev Bras Ter Intensiva. 2020;32(2):200-202
DOI 10.5935/0103-507X.20200034
Views0Shared decision-makingShared decision-making finds support in the ethical principle of beneficence and nonmaleficence. It aims to involve patients and/or family members in decisions related to clinical care and should be part of clinical practice. Shared decision-making means respecting the autonomy of patients and ensuring care that is consistent with their values and preferences. Therefore, the […]See more -
Case Report
Use of CPAP as an alternative to the apnea test during the determination of brain death in hypoxemic patients. Report of two cases
Rev Bras Ter Intensiva. 2020;32(2):319-325
Abstract
Case ReportUse of CPAP as an alternative to the apnea test during the determination of brain death in hypoxemic patients. Report of two cases
Rev Bras Ter Intensiva. 2020;32(2):319-325
DOI 10.5935/0103-507X.20200032
Views0ABSTRACT
The apnea test, which involves disconnection from the mechanical ventilator, presents risks during the determination of brain death, especially in hypoxemic patients. We describe the performance of the apnea test without disconnection from the mechanical ventilator in two patients. The first case involved an 8-year-old boy admitted with severe hypoxemia due to pneumonia. He presented with cardiorespiratory arrest, followed by unresponsive coma due to hypoxic-ischemic encephalopathy. Two clinical exams revealed the absence of brainstem reflexes, and transcranial Doppler ultrasound revealed brain circulatory arrest. Three attempts were made to perform the apnea test, which were interrupted by hypoxemia; therefore, the apnea test was performed without disconnection from the mechanical ventilator, adjusting the continuous airway pressure to 10cmH2O and the inspired fraction of oxygen to 100%. The oxygen saturation was maintained at 100% for 10 minutes. Posttest blood gas analysis results were as follows: pH, 6.90; partial pressure of oxygen, 284.0mmHg; partial pressure of carbon dioxide, 94.0mmHg; and oxygen saturation, 100%. The second case involved a 43-year-old woman admitted with subarachnoid hemorrhage (Hunt-Hess V and Fisher IV). Two clinical exams revealed unresponsive coma and absence of all brainstem reflexes. Brain scintigraphy showed no radioisotope uptake into the brain parenchyma. The first attempt at the apnea test was stopped after 5 minutes due to hypothermia (34.9°C). After rewarming, the apnea test was repeated without disconnection from the mechanical ventilator, showing maintenance of the functional residual volume with electrical bioimpedance. Posttest blood gas analysis results were as follows: pH, 7.01; partial pressure of oxygen, 232.0mmHg; partial pressure of carbon dioxide, 66.9mmHg; and oxygen saturation, 99.0%. The apnea test without disconnection from the mechanical ventilator allowed the preservation of oxygenation in both cases. The use of continuous airway pressure during the apnea test seems to be a safe alternative in order to maintain alveolar recruitment and oxygenation during brain death determination.
Keywords:ApneaApnea testingbrain deathContinuous positive airway pressureElectrical impedanceHypoxiaRespiratory insufficiencySleep apnea syndromesTomography/methodsSee more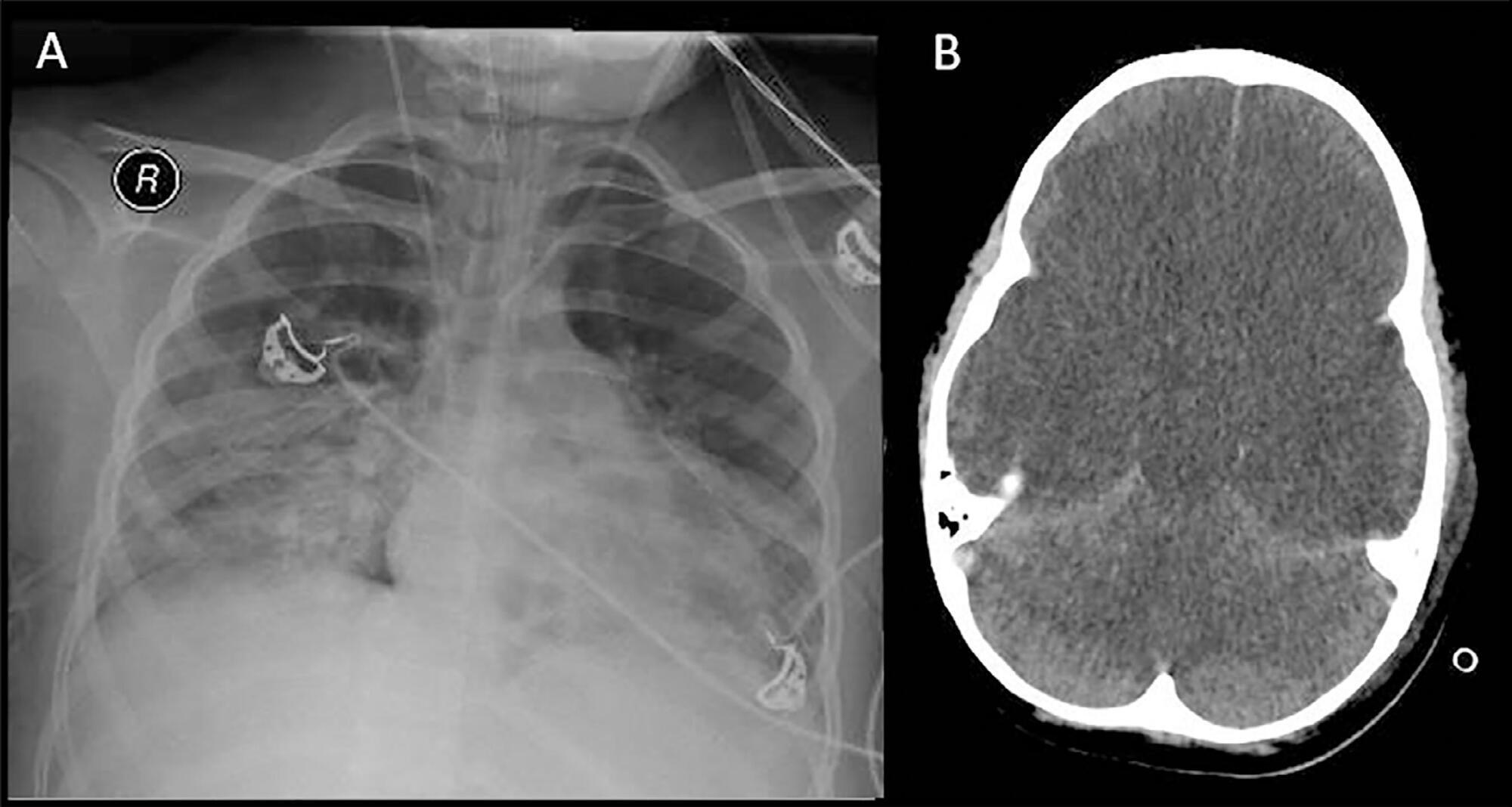
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis