Abstract
Rev Bras Ter Intensiva. 2021;33(1):1-11
DOI 10.5935/0103-507X.20210001
To contribute to updating the recommendations for brain-dead potential organ donor management.
A group of 27 experts, including intensivists, transplant coordinators, transplant surgeons, and epidemiologists, answered questions related to the following topics were divided into mechanical ventilation, hemodynamics, endocrine-metabolic management, infection, body temperature, blood transfusion, and checklists use. The outcomes considered were cardiac arrests, number of organs removed or transplanted as well as function / survival of transplanted organs. The quality of evidence of the recommendations was assessed using the Grading of Recommendations Assessment, Development, and Evaluation system to classify the recommendations.
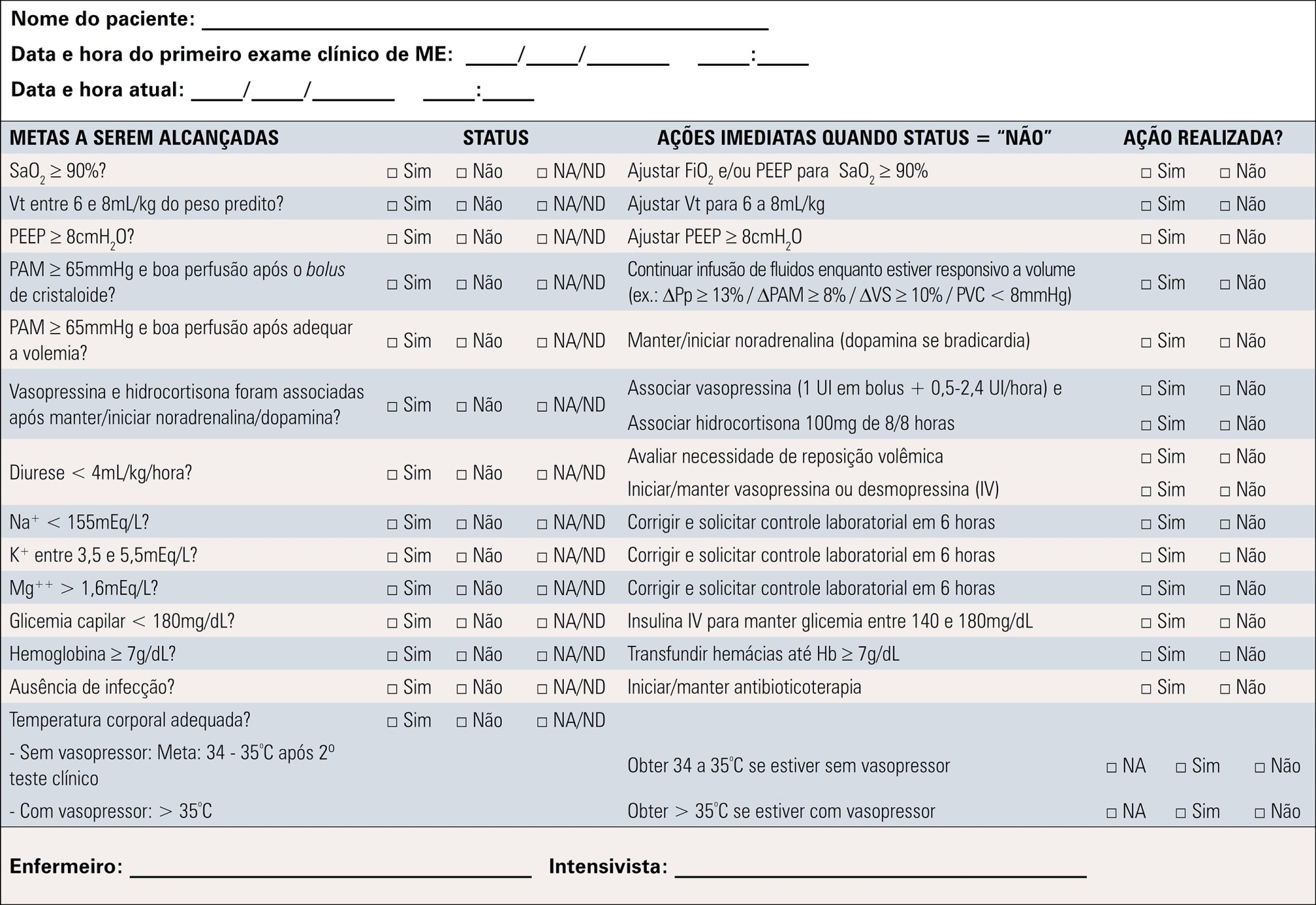
A total of 19 recommendations were drawn from the expert panel. Of these, 7 were classified as strong, 11 as weak and 1 was considered a good clinical practice.
Despite the agreement among panel members on most recommendations, the grade of recommendation was mostly weak.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):166-196
DOI 10.5935/0103-507X.20200039
Different therapies are currently used, considered, or proposed for the treatment of COVID-19; for many of those therapies, no appropriate assessment of effectiveness and safety was performed. This document aims to provide scientifically available evidence-based information in a transparent interpretation, to subsidize decisions related to the pharmacological therapy of COVID-19 in Brazil.
A group of 27 experts and methodologists integrated a task-force formed by professionals from the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB), the Brazilian Society of Infectious Diseases (Sociedad Brasileira de Infectologia - SBI) and the Brazilian Society of Pulmonology and Tisiology (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT). Rapid systematic reviews, updated on April 28, 2020, were conducted. The assessment of the quality of evidence and the development of recommendations followed the GRADE system. The recommendations were written on May 5, 8, and 13, 2020.
Eleven recommendations were issued based on low or very-low level evidence. We do not recommend the routine use of hydroxychloroquine, chloroquine, azithromycin, lopinavir/ritonavir, corticosteroids, or tocilizumab for the treatment of COVID-19. Prophylactic heparin should be used in hospitalized patients, however, no anticoagulation should be provided for patients without a specific clinical indication. Antibiotics and oseltamivir should only be considered for patients with suspected bacterial or influenza coinfection, respectively.
So far no pharmacological intervention was proven effective and safe to warrant its use in the routine treatment of COVID-19 patients; therefore such patients should ideally be treated in the context of clinical trials. The recommendations herein provided will be revised continuously aiming to capture newly generated evidence.
Search
Search in:
Case reports (56) Child (53) Coronavirus infections (34) COVID-19 (46) Critical care (115) Critical illness (54) Extracorporeal membrane oxygenation (26) Infant, newborn (27) Intensive care (72) Intensive care units (256) Intensive care units, pediatric (31) mechanical ventilation (38) Mortality (76) Physical therapy modalities (28) Prognosis (61) Respiration, artificial (119) Respiratory insufficiency (26) risk factors (34) SARS-CoV-2 (28) Sepsis (98)