You searched for:"Viviane Cordeiro Veiga"
We found (25) results for your search.-
Clinical Report
Prospective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
Abstract
Clinical ReportProspective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
DOI 10.62675/2965-2774.20240210-en
Views76ABSTRACT
Background:
Driving pressure has been suggested to be the main driver of ventilator-induced lung injury and mortality in observational studies of acute respiratory distress syndrome. Whether a driving pressure-limiting strategy can improve clinical outcomes is unclear.
Objective:
To describe the protocol and statistical analysis plan that will be used to test whether a driving pressure-limiting strategy including positive end-expiratory pressure titration according to the best respiratory compliance and reduction in tidal volume is superior to a standard strategy involving the use of the ARDSNet low-positive end-expiratory pressure table in terms of increasing the number of ventilator-free days in patients with acute respiratory distress syndrome due to community-acquired pneumonia.
Methods:
The ventilator STrAtegy for coMmunIty acquired pNeumoniA (STAMINA) study is a randomized, multicenter, open-label trial that compares a driving pressure-limiting strategy to the ARDSnet low-positive end-expiratory pressure table in patients with moderate-to-severe acute respiratory distress syndrome due to community-acquired pneumonia admitted to intensive care units. We expect to recruit 500 patients from 20 Brazilian and 2 Colombian intensive care units. They will be randomized to a driving pressure-limiting strategy group or to a standard strategy using the ARDSNet low-positive end-expiratory pressure table. In the driving pressure-limiting strategy group, positive end-expiratory pressure will be titrated according to the best respiratory system compliance.
Outcomes:
The primary outcome is the number of ventilator-free days within 28 days. The secondary outcomes are in-hospital and intensive care unit mortality and the need for rescue therapies such as extracorporeal life support, recruitment maneuvers and inhaled nitric oxide.
Conclusion:
STAMINA is designed to provide evidence on whether a driving pressure-limiting strategy is superior to the ARDSNet low-positive end-expiratory pressure table strategy for increasing the number of ventilator-free days within 28 days in patients with moderate-to-severe acute respiratory distress syndrome. Here, we describe the rationale, design and status of the trial.
Keywords:Extracorporeal membrane oxygenationPneumoniaPositive pressure respirationRespiration, artificialRespiratory distress syndromeVentilator-induced lung injurySee more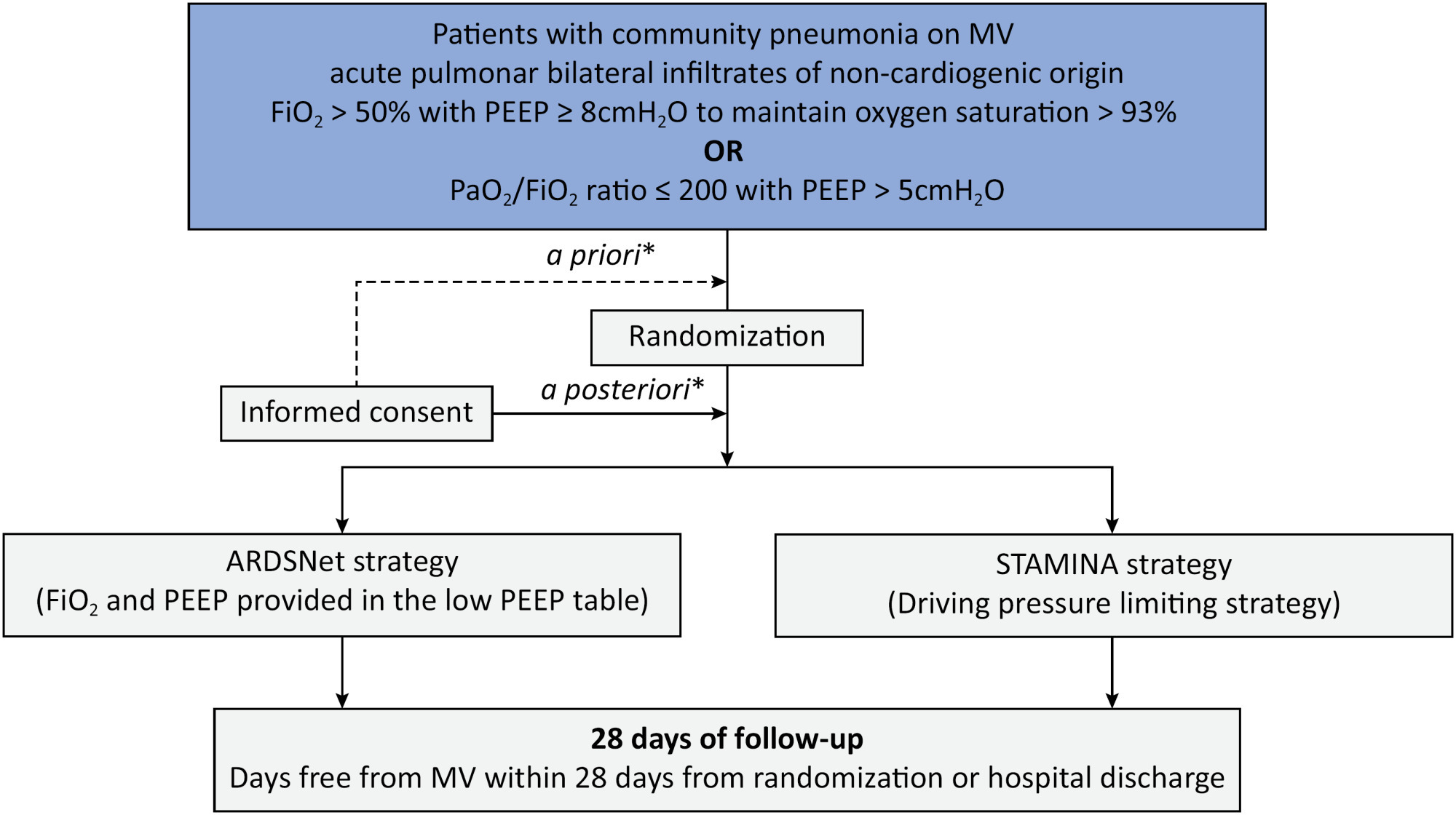
-
Special Article
The II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
Abstract
Special ArticleThe II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
DOI 10.5935/2965-2774.20230136-pt
Views22ABSTRACT
Objective:
To update the recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Methods:
Experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Results:
Twenty-one recommendations were generated, including strong recommendations for the use of corticosteroids in patients using supplemental oxygen and conditional recommendations for the use of tocilizumab and baricitinib for patients on supplemental oxygen or on noninvasive ventilation and anticoagulants to prevent thromboembolism. Due to suspension of use authorization, it was not possible to make recommendations regarding the use of casirivimab + imdevimab. Strong recommendations against the use of azithromycin in patients without suspected bacterial infection, hydroxychloroquine, convalescent plasma, colchicine, and lopinavir + ritonavir and conditional recommendations against the use of ivermectin and remdesivir were made.
Conclusion:
New recommendations for the treatment of hospitalized patients with COVID-19 were generated, such as those for tocilizumab and baricitinib. Corticosteroids and prophylaxis for thromboembolism are still recommended, the latter with conditional recommendation. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and to promote resource economy.
Keywords:BrazilCoronavirus infectionsCOVID-19COVID-19/drug therapyHealth planning guidelinesSARS-CoV-2See more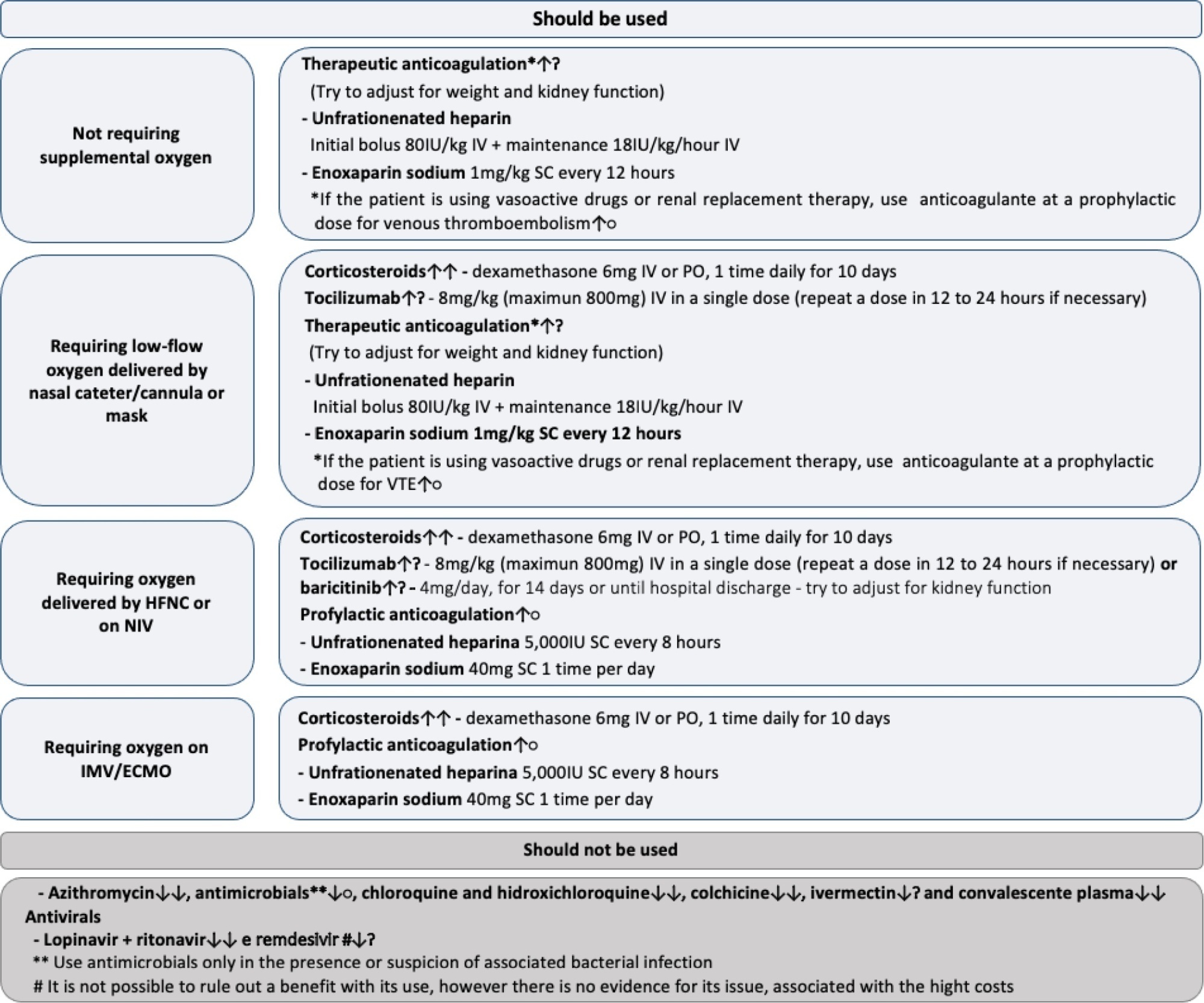
-
Original Article
Effects of balanced solution on short-term outcomes in traumatic brain injury patients: a secondary analysis of the BaSICS randomized trial
Rev Bras Ter Intensiva. 2022;34(4):410-417
Abstract
Original ArticleEffects of balanced solution on short-term outcomes in traumatic brain injury patients: a secondary analysis of the BaSICS randomized trial
Rev Bras Ter Intensiva. 2022;34(4):410-417
DOI 10.5935/0103-507X.20220261-en
Views2ABSTRACT
Objective:
To describe the effects of balanced solution use on the short-term outcomes of patients with traumatic brain injury enrolled in BaSICS trial.
Methods:
Patients were randomized to receive either 0.9% saline or balanced solution during their intensive care unit stay. The primary endpoint was 90-day mortality, and the secondary outcomes were days alive and free of intensive care unit stay at 28 days. The primary endpoint was assessed using Bayesian logistic regression. The secondary endpoint was assessed using a Bayesian zero-inflated beta binomial regression.
Results:
We included 483 patients (236 in the 0.9% saline arm and 247 in the balanced solution arm). A total of 338 patients (70%) with a Glasgow coma scale score ≤ 12 were enrolled. The overall probability that balanced solutions were associated with higher 90-day mortality was 0.98 (OR 1.48; 95%CrI 1.04 – 2.09); this mortality increment was particularly noticeable in patients with a Glasgow coma scale score below 6 at enrollment (probability of harm of 0.99). Balanced solutions were associated with -1.64 days alive and free of intensive care unit at 28 days (95%CrI -3.32 – 0.00) with a probability of harm of 0.97.
Conclusion:
There was a high probability that balanced solutions were associated with high 90-day mortality and fewer days alive and free of intensive care units at 28 days.
Keywords:Balanced solutionsBrain injuriesCritical careHospital mortalityMortalitySaline solutiontraumaticSee more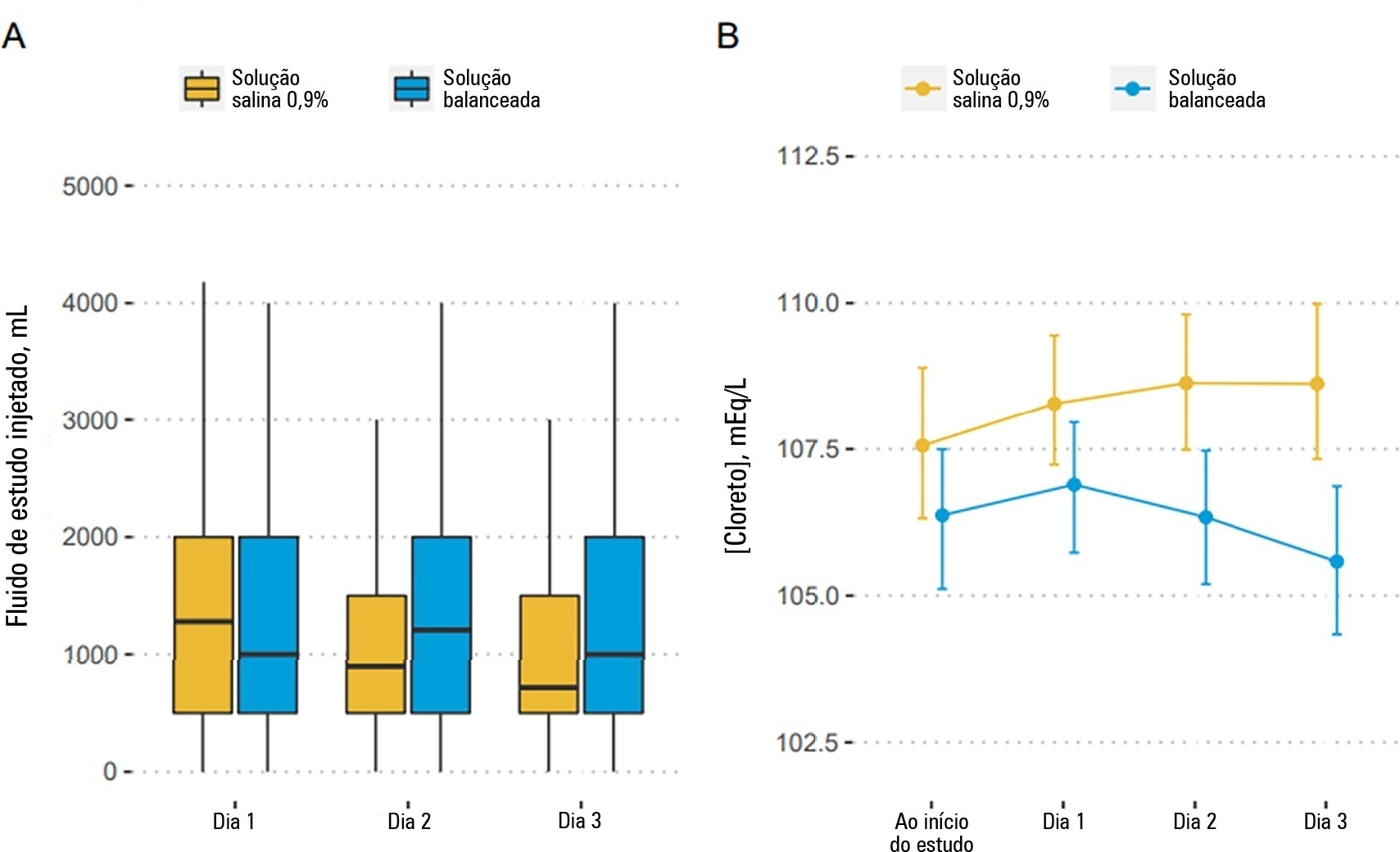
-
Original Article
IMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
Abstract
Original ArticleIMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
Views2ABSTRACT
Objective:
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
Methods:
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
Results:
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
Conclusion:
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.
Keywords:bacterialBacterial infectionsDatabaseDatabase management systemsDrug-resistanceIMPACTO-MRIntensive care unitsSoftwareSee more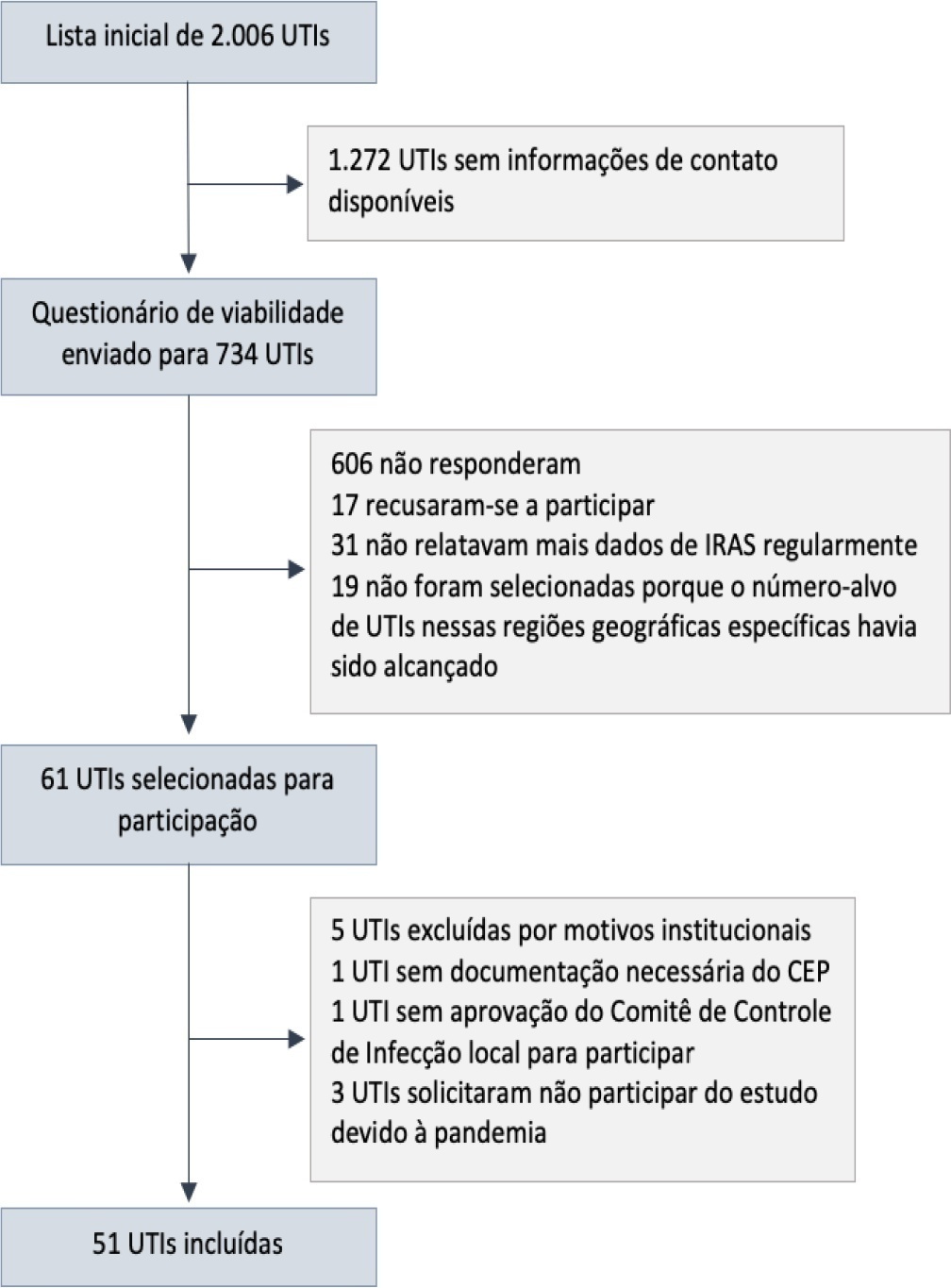
-
Original Article
Clinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
Abstract
Original ArticleClinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
DOI 10.5935/0103-507X.20220040-en
Views3ABSTRACT
Objective:
To compare the lung mechanics and outcomes between COVID-19-associated acute respiratory distress syndrome and non-COVID-19-associated acute respiratory distress syndrome.
Methods:
We combined data from two randomized trials in acute respiratory distress syndrome, one including only COVID-19 patients and the other including only patients without COVID-19, to determine whether COVID-19-associated acute respiratory distress syndrome is associated with higher 28-day mortality than non-COVID-19 acute respiratory distress syndrome and to examine the differences in lung mechanics between these two types of acute respiratory distress syndrome.
Results:
A total of 299 patients with COVID-19-associated acute respiratory distress syndrome and 1,010 patients with non-COVID-19-associated acute respiratory distress syndrome were included in the main analysis. The results showed that non-COVID-19 patients used higher positive end-expiratory pressure (12.5cmH2O; SD 3.2 versus 11.7cmH2O SD 2.8; p < 0.001), were ventilated with lower tidal volumes (5.8mL/kg; SD 1.0 versus 6.5mL/kg; SD 1.2; p < 0.001) and had lower static respiratory compliance adjusted for ideal body weight (0.5mL/cmH2O/kg; SD 0.3 versus 0.6mL/cmH2O/kg; SD 0.3; p = 0.01). There was no difference between groups in 28-day mortality (52.3% versus 58.9%; p = 0.52) or mechanical ventilation duration in the first 28 days among survivors (13 [IQR 5 – 22] versus 12 [IQR 6 – 26], p = 0.46).
Conclusion:
This analysis showed that patients with non-COVID-19-associated acute respiratory distress syndrome have different lung mechanics but similar outcomes to COVID-19-associated acute respiratory distress syndrome patients. After propensity score matching, there was no difference in lung mechanics or outcomes between groups.
Keywords:Coronavirus infectionsCOVID-19Critical careCritical care outcomesRespiratory distress syndromeRespiratory mechanicsSee more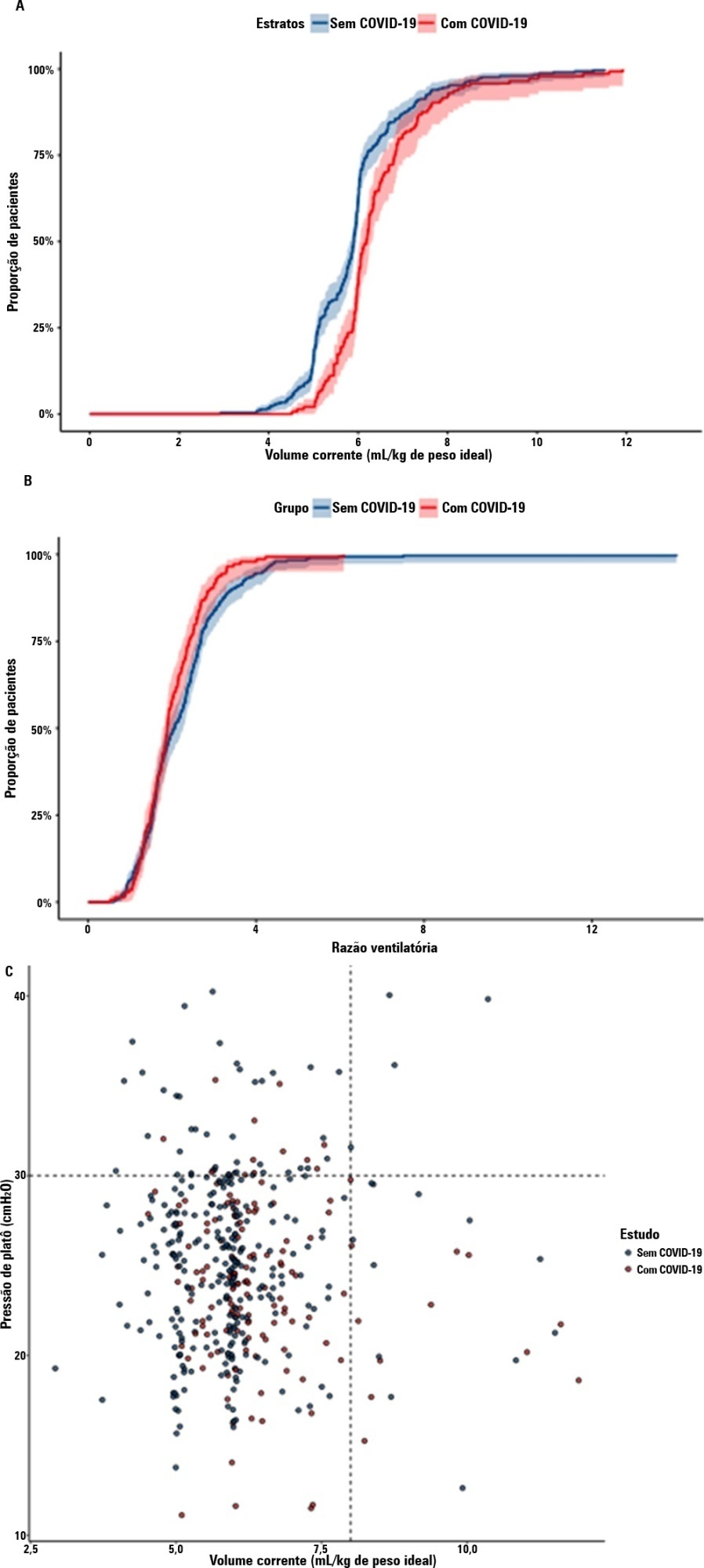
-
Special Article
Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19: Joint guideline of Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia, Sociedade Brasileira de Reumatologia
Rev Bras Ter Intensiva. 2022;34(1):1-12
Abstract
Special ArticleBrazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19: Joint guideline of Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia, Sociedade Brasileira de Reumatologia
Rev Bras Ter Intensiva. 2022;34(1):1-12
DOI 10.5935/0103-507X.20220001-en
Views7See moreABSTRACT
Objective:
Several therapies are being used or proposed for COVID-19, and many lack appropriate evaluations of their effectiveness and safety. The purpose of this document is to develop recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Methods:
A group of 27 experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Results:
Sixteen recommendations were generated. They include strong recommendations for the use of corticosteroids in patients using supplemental oxygen, the use of anticoagulants at prophylactic doses to prevent thromboembolism and the nonuse of antibiotics in patients without suspected bacterial infection. It was not possible to make a recommendation regarding the use of tocilizumab in patients hospitalized with COVID-19 using oxygen due to uncertainties regarding the availability of and access to the drug. Strong recommendations against the use of hydroxychloroquine, convalescent plasma, colchicine, lopinavir + ritonavir and antibiotics in patients without suspected bacterial infection and also conditional recommendations against the use of casirivimab + imdevimab, ivermectin and rendesivir were made.
Conclusion:
To date, few therapies have proven effective in the treatment of hospitalized patients with COVID-19, and only corticosteroids and prophylaxis for thromboembolism are recommended. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and promote economical resource use.

Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis