Abstract
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
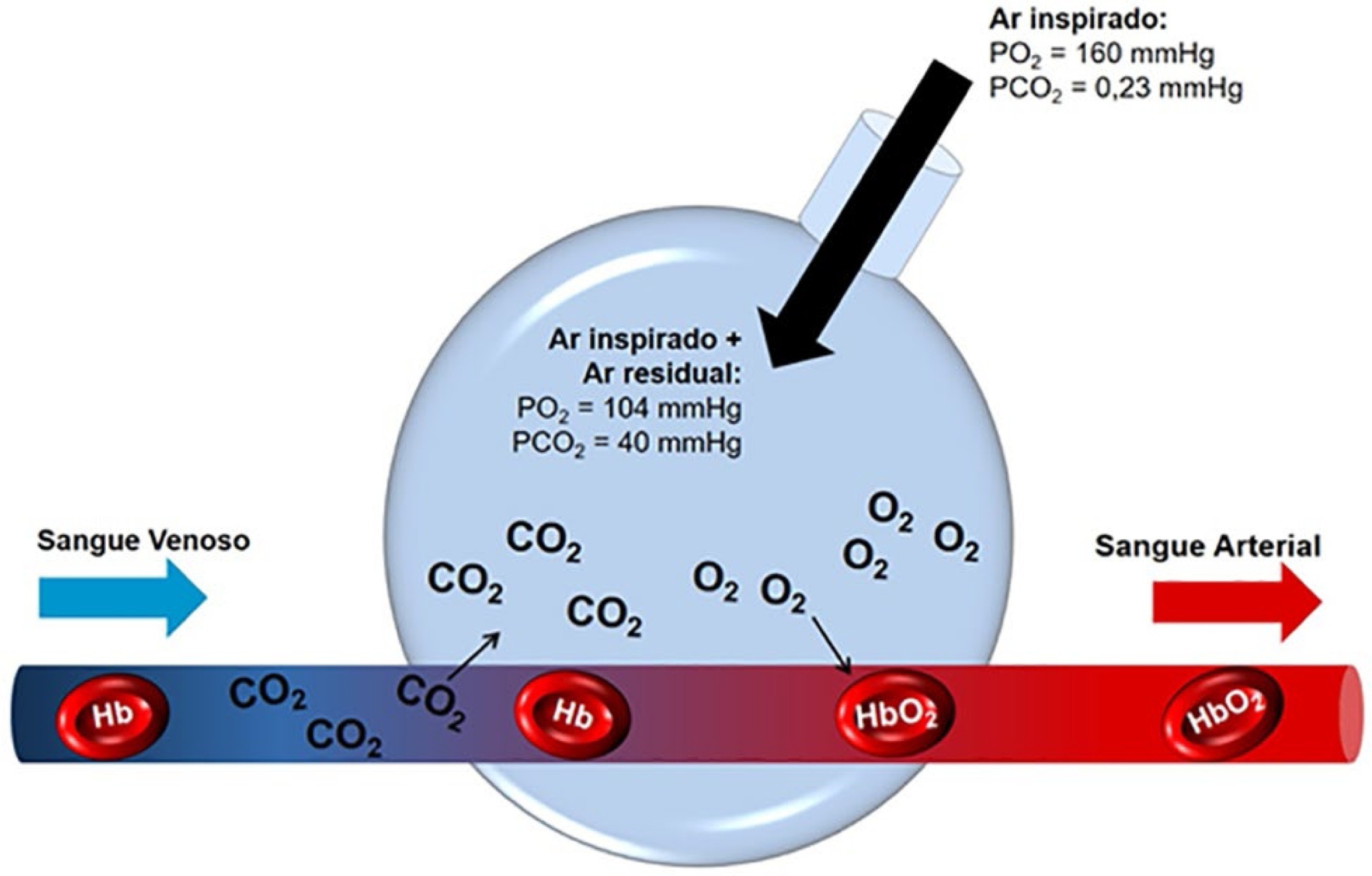
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária - ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):268-276
DOI 10.5935/0103-507X.20200043
To conduct a national survey of intensive care professionals to identify the practices for promoting sleep in adult intensive care units in Brazil and describe the professionals’ perceptions of the importance of sleep for patients.
An electronic questionnaire was distributed by the clinical research cooperation network of the Associação de Medicina Intensiva Brasileira and by the Brazilian Research in Intensive Care Network to physicians and nurses registered with the association. The questionnaire evaluated the profile of the respondents, the profile of their intensive care units, whether protocols for promoting sleep were present, the pharmacological and nonpharmacological measures typically employed in the unit, and the professionals’ perceptions regarding sleep in critically ill patients.
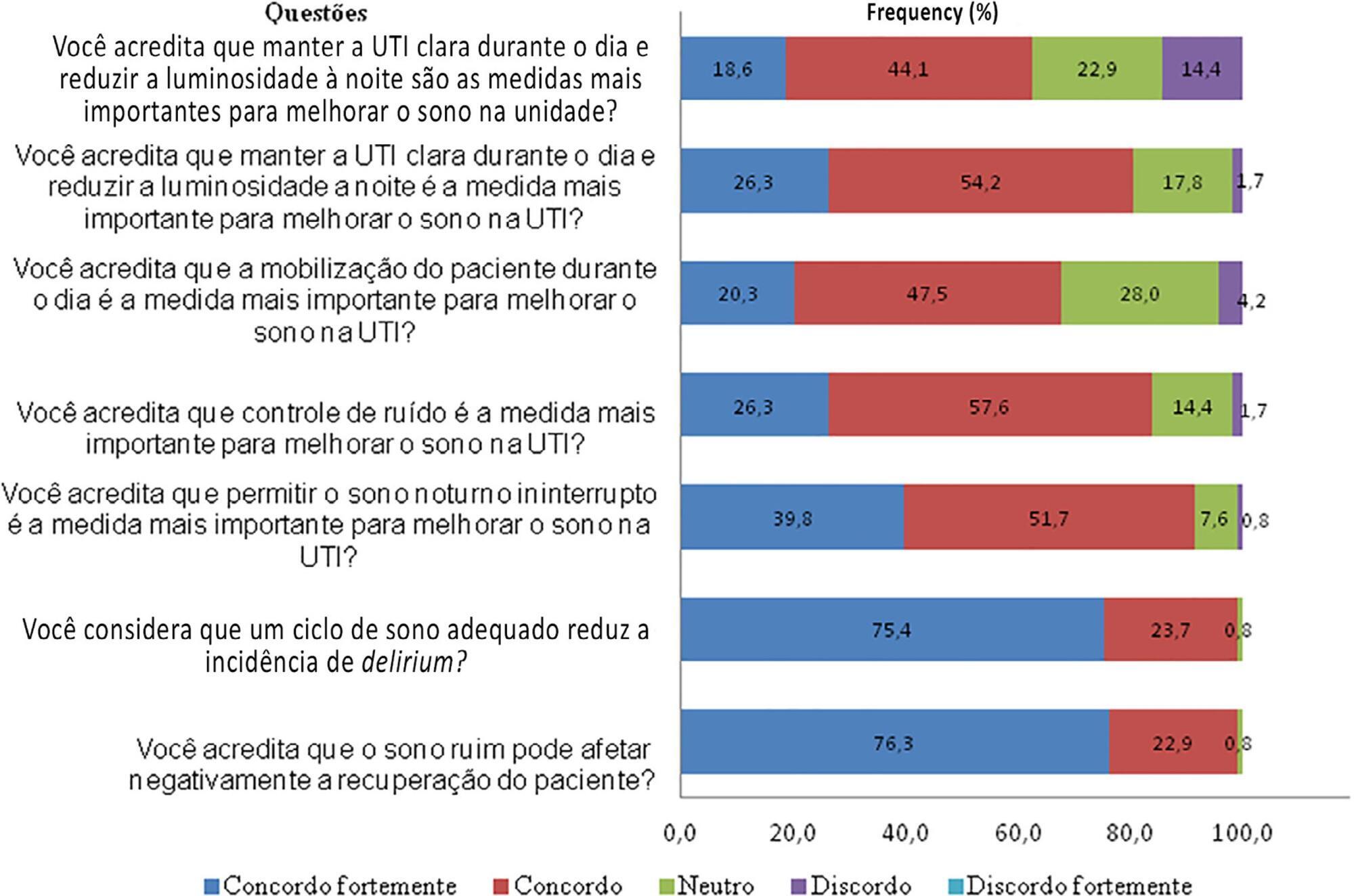
A total of 118 questionnaires were evaluated. The Southeast region of the country was the most represented (50 questionnaires, 42.4%). The majority of units had a clinical-surgical profile (93 questionnaires; 78.8%), and 26 had a continuous visitation policy (22.0%). Only 18 intensive care units (15.3%) reported having protocols for promoting sleep. The most cited measure for sleep promotion was reducing light during the night (95 questionnaires; 80.5%), which was more often performed in private intensive care units. Almost all of the responders (99%) believed that poor-quality sleep has a negative impact on patient recovery.
The responses to this Brazilian survey revealed that few intensive care units had a program for promoting sleep, although almost all participants recognized the importance of sleep in patient recovery.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):166-196
DOI 10.5935/0103-507X.20200039
Different therapies are currently used, considered, or proposed for the treatment of COVID-19; for many of those therapies, no appropriate assessment of effectiveness and safety was performed. This document aims to provide scientifically available evidence-based information in a transparent interpretation, to subsidize decisions related to the pharmacological therapy of COVID-19 in Brazil.
A group of 27 experts and methodologists integrated a task-force formed by professionals from the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB), the Brazilian Society of Infectious Diseases (Sociedad Brasileira de Infectologia - SBI) and the Brazilian Society of Pulmonology and Tisiology (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT). Rapid systematic reviews, updated on April 28, 2020, were conducted. The assessment of the quality of evidence and the development of recommendations followed the GRADE system. The recommendations were written on May 5, 8, and 13, 2020.
Eleven recommendations were issued based on low or very-low level evidence. We do not recommend the routine use of hydroxychloroquine, chloroquine, azithromycin, lopinavir/ritonavir, corticosteroids, or tocilizumab for the treatment of COVID-19. Prophylactic heparin should be used in hospitalized patients, however, no anticoagulation should be provided for patients without a specific clinical indication. Antibiotics and oseltamivir should only be considered for patients with suspected bacterial or influenza coinfection, respectively.
So far no pharmacological intervention was proven effective and safe to warrant its use in the routine treatment of COVID-19 patients; therefore such patients should ideally be treated in the context of clinical trials. The recommendations herein provided will be revised continuously aiming to capture newly generated evidence.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):224-228
DOI 10.5935/0103-507X.20200030
To estimate the reporting rates of coronavirus disease 2019 (COVID-19) cases for Brazil as a whole and states.
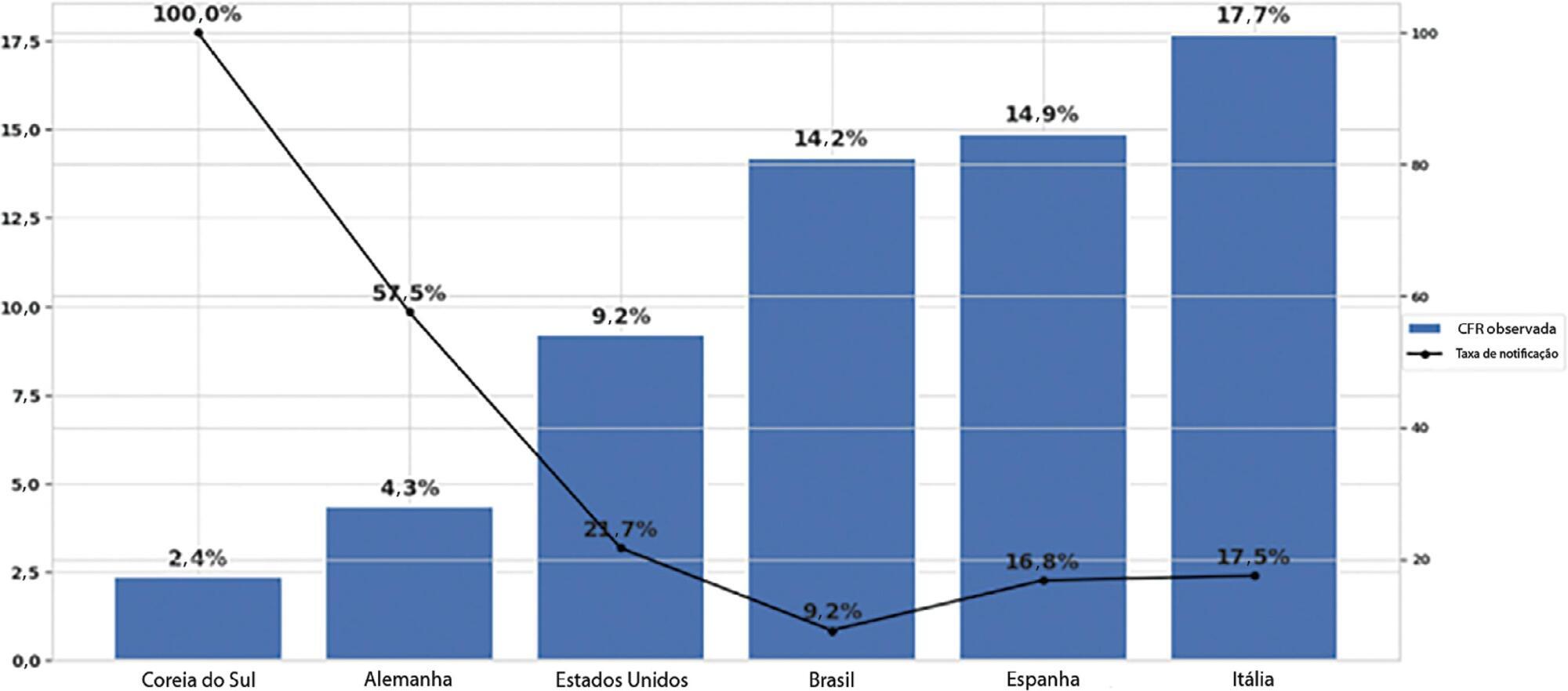
We estimated the actual number of COVID-19 cases using the reported number of deaths in Brazil and each state, and the expected case-fatality ratio from the World Health Organization. Brazil’s expected case-fatality ratio was also adjusted by the population’s age pyramid. Therefore, the notification rate can be defined as the number of confirmed cases (notified by the Ministry of Health) divided by the number of expected cases (estimated from the number of deaths).
The reporting rate for COVID-19 in Brazil was estimated at 9.2% (95%CI 8.8% - 9.5%), with all the states presenting rates below 30%. São Paulo and Rio de Janeiro, the most populated states in Brazil, showed small reporting rates (8.9% and 7.2%, respectively). The highest reporting rate occurred in Roraima (31.7%) and the lowest in Paraiba (3.4%).
The results indicated that the reporting of confirmed cases in Brazil is much lower as compared to other countries we analyzed. Therefore, decision-makers, including the government, fail to know the actual dimension of the pandemic, which may interfere with the determination of control measures.
Abstract
Rev Bras Ter Intensiva. 2020;32(1):17-27
DOI 10.5935/0103-507X.20200005
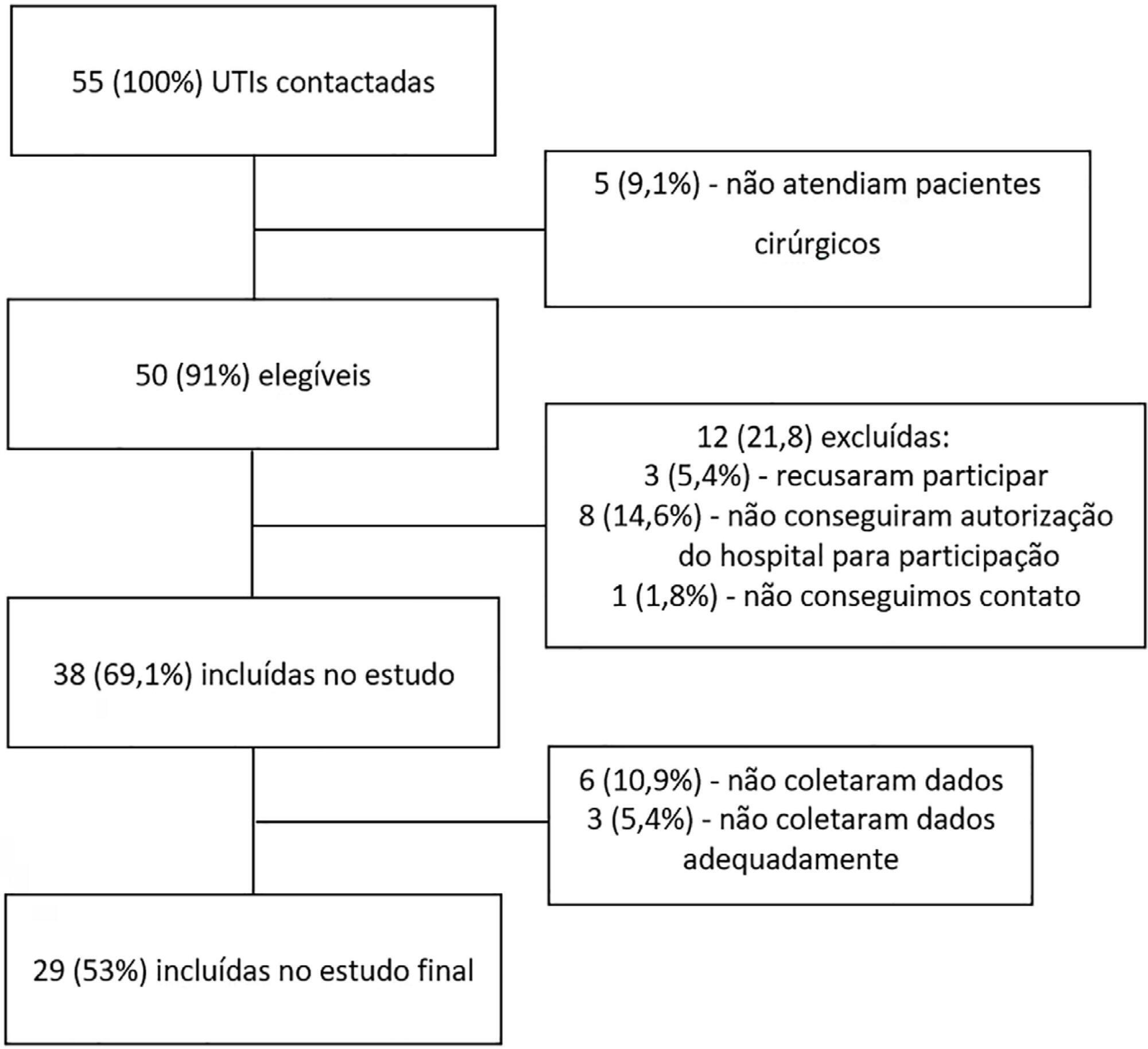
To define the epidemiological profile and the main determinants of morbidity and mortality in noncardiac high surgical risk patients in Brazil.
This was a prospective, observational and multicenter study. All noncardiac surgical patients admitted to intensive care units, i.e., those considered high risk, within a 1-month period were evaluated and monitored daily for a maximum of 7 days in the intensive care unit to determine complications. The 28-day postoperative, intensive care unit and hospital mortality rates were evaluated.
Twenty-nine intensive care units participated in the study. Surgeries were performed in 25,500 patients, of whom 904 (3.5%) were high-risk (95% confidence interval - 95%CI 3.3% - 3.8%) and were included in the study. Of the participating patients, 48.3% were from private intensive care units, and 51.7% were from public intensive care units. The length of stay in the intensive care unit was 2.0 (1.0 - 4.0) days, and the length of hospital stay was 9.5 (5.4 - 18.6) days. The complication rate was 29.9% (95%CI 26.4 - 33.7), and the 28-day postoperative mortality rate was 9.6% (95%CI 7.4 - 12.1). The independent risk factors for complications were the Simplified Acute Physiology Score 3 (SAPS 3; odds ratio - OR = 1.02; 95%CI 1.01 - 1.03) and Sequential Organ Failure Assessment Score (SOFA) on admission to the intensive care unit (OR = 1.17; 95%CI 1.09 - 1.25), surgical time (OR = 1.001, 95%CI 1.000 - 1.002) and emergency surgeries (OR = 1.93, 95%CI, 1.10 - 3.38). In addition, there were associations with 28-day mortality (OR = 1.032; 95%CI 1.011 - 1.052), SAPS 3 (OR = 1.041; 95%CI 1.107 - 1.279), SOFA (OR = 1.175, 95%CI 1.069 - 1.292) and emergency surgeries (OR = 2.509; 95%CI 1.040 - 6.051).
Higher prognostic scores, elderly patients, longer surgical times and emergency surgeries were strongly associated with higher 28-day mortality and more complications during the intensive care unit stay.
Abstract
Rev Bras Ter Intensiva. 2019;31(3):403-409
DOI 10.5935/0103-507X.20190050
Brain death, defined as the complete and irreversible loss of brain functions, has a history that is linked to the emergence of intensive care units and the advancement of artificial ventilatory support. In Brazil, by federal law, the criteria for the diagnosis of brain death have been defined by the Federal Council of Medicine since 1997 and apply to the entire Brazilian territory. Resolution 2,173/2017 of the Federal Council of Medicine updated the criteria for diagnosing brain death. These changes include the following: the requirement for the patient to meet specific physiological prerequisites and for the physician to provide optimized care to the patient before starting the procedures for diagnosing brain death and to perform complementary tests, as well as the need for specific training for physicians who make this diagnosis. Other changes include the reduction of the time interval between the two clinical examinations, the possibility of continuing procedures in the presence of unilateral ear or eye injury, the performance of a single apnea test and the creation of a statement of brain death determination that includes the recording of all procedures in a single document. This document, despite the controversy surrounding it, increases the safety necessary when establishing a diagnosis of such importance and has positive implications that extend beyond the patient and the physician to reach the entire health system.
Abstract
Rev Bras Ter Intensiva. 2019;31(1):71-78
DOI 10.5935/0103-507X.20190013
To compare the clinical characteristics and outcomes of patients with community-acquired and hospital-acquired sepsis.
This is a retrospective cohort study that included all patients with a diagnosis of sepsis detected between January 2010 and December 2015 at a private hospital in southern Brazil. Outcomes (mortality, intensive care unit and hospital lengths of stay) were measured by analyzing electronic records.
There were 543 hospitalized patients with a diagnosis of sepsis, with a frequency of 90.5 (85 to 105) cases/year. Of these, 319 (58%) cases were classified as hospital-acquired sepsis. This group exhibited more severe disease and had a larger number of organ dysfunctions, with higher hospital [8 (8 - 10) versus 23 (20 - 27) days; p < 0.001] and intensive care unit [5 (4 - 7) versus 8.5 (7 - 10); p < 0.001] lengths of stay and higher in-hospital mortality (30.7% versus 15.6%; p < 0.001) than those with community-acquired sepsis. After adjusting for age, APACHE II scores, and hemodynamic and respiratory dysfunction, hospital-acquired sepsis remained associated with increased mortality (OR 1.96; 95%CI 1.15 - 3.32, p = 0.013).
The present results contribute to the definition of the epidemiological profile of sepsis in the sample studied, in which hospital-acquired sepsis was more severe and was associated with higher mortality.
Abstract
Rev Bras Ter Intensiva. 2018;30(3):327-332
DOI 10.5935/0103-507X.20180044
To evaluate the practice of dentistry in intensive care units.
An observational survey study was conducted in which questionnaires were sent via the online platform for collaboration in intensive care research in Brazil (AMIBnet). The study was carried out from June to October 2017. The questionnaires, which contained 26 closed questions about hospitals and dentistry practices in the intensive care units, were sent to 4,569 professionals from different specialties practicing in the units.
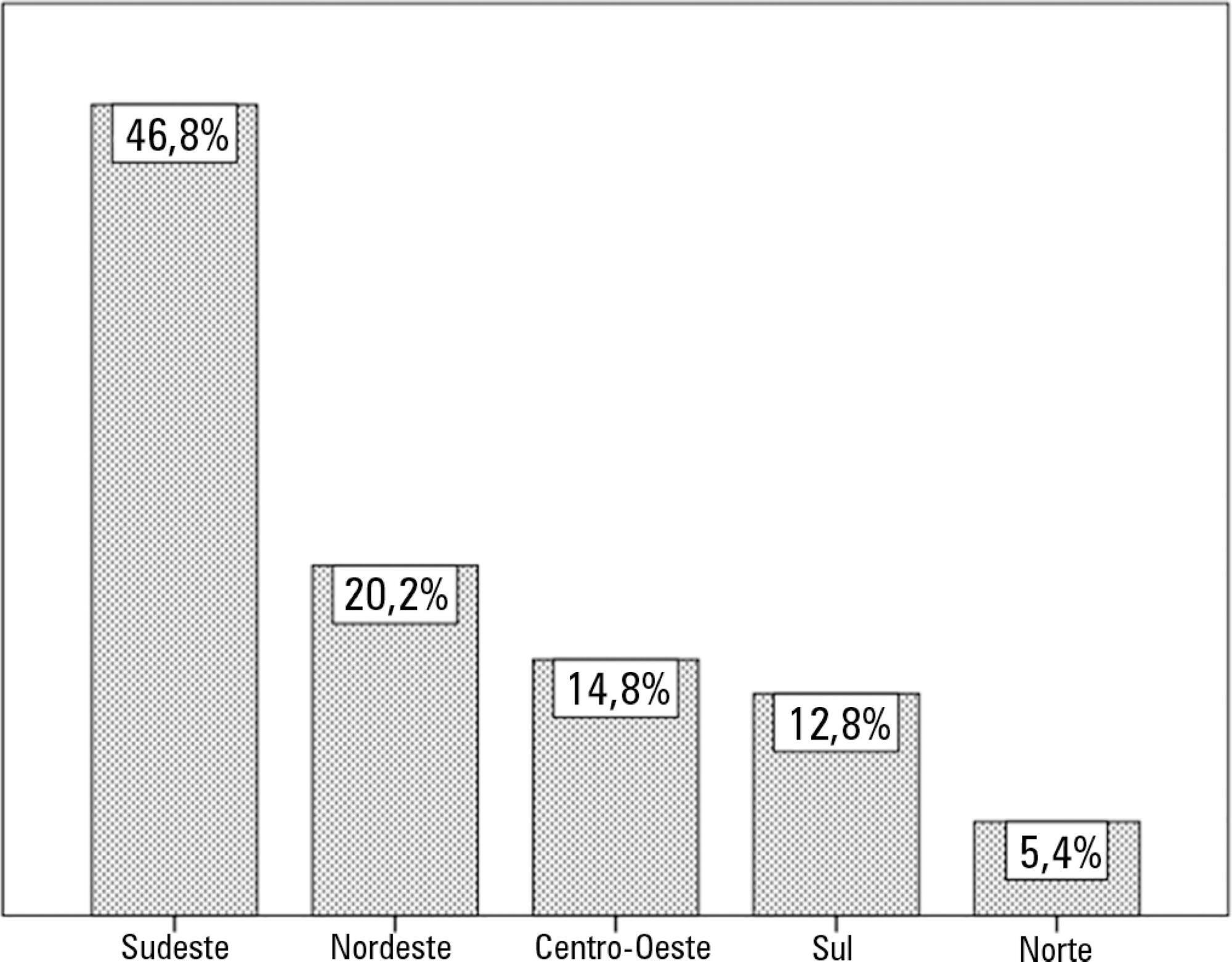
In total, 203 questionnaires were returned, resulting in a response rate of 4.44%. Most of the responses were from intensive care units in the Southeast region of the country (46.8%). Public hospitals (37.9%) and private hospitals (36.4%) had similar participation rates. Of the respondents, 55% indicated that a bedside dentistry service was present, and they were provided in different ways.
The presence of dentistry services and oral health service delivery training and protocols were correlated. The oral care methods varied greatly among the intensive care units surveyed.