You searched for:"Suzana Margareth Ajeje Lobo"
We found (14) results for your search.-
Special Article
The II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
Abstract
Special ArticleThe II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
DOI 10.5935/2965-2774.20230136-pt
Views22ABSTRACT
Objective:
To update the recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Methods:
Experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Results:
Twenty-one recommendations were generated, including strong recommendations for the use of corticosteroids in patients using supplemental oxygen and conditional recommendations for the use of tocilizumab and baricitinib for patients on supplemental oxygen or on noninvasive ventilation and anticoagulants to prevent thromboembolism. Due to suspension of use authorization, it was not possible to make recommendations regarding the use of casirivimab + imdevimab. Strong recommendations against the use of azithromycin in patients without suspected bacterial infection, hydroxychloroquine, convalescent plasma, colchicine, and lopinavir + ritonavir and conditional recommendations against the use of ivermectin and remdesivir were made.
Conclusion:
New recommendations for the treatment of hospitalized patients with COVID-19 were generated, such as those for tocilizumab and baricitinib. Corticosteroids and prophylaxis for thromboembolism are still recommended, the latter with conditional recommendation. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and to promote resource economy.
Keywords:BrazilCoronavirus infectionsCOVID-19COVID-19/drug therapyHealth planning guidelinesSARS-CoV-2See more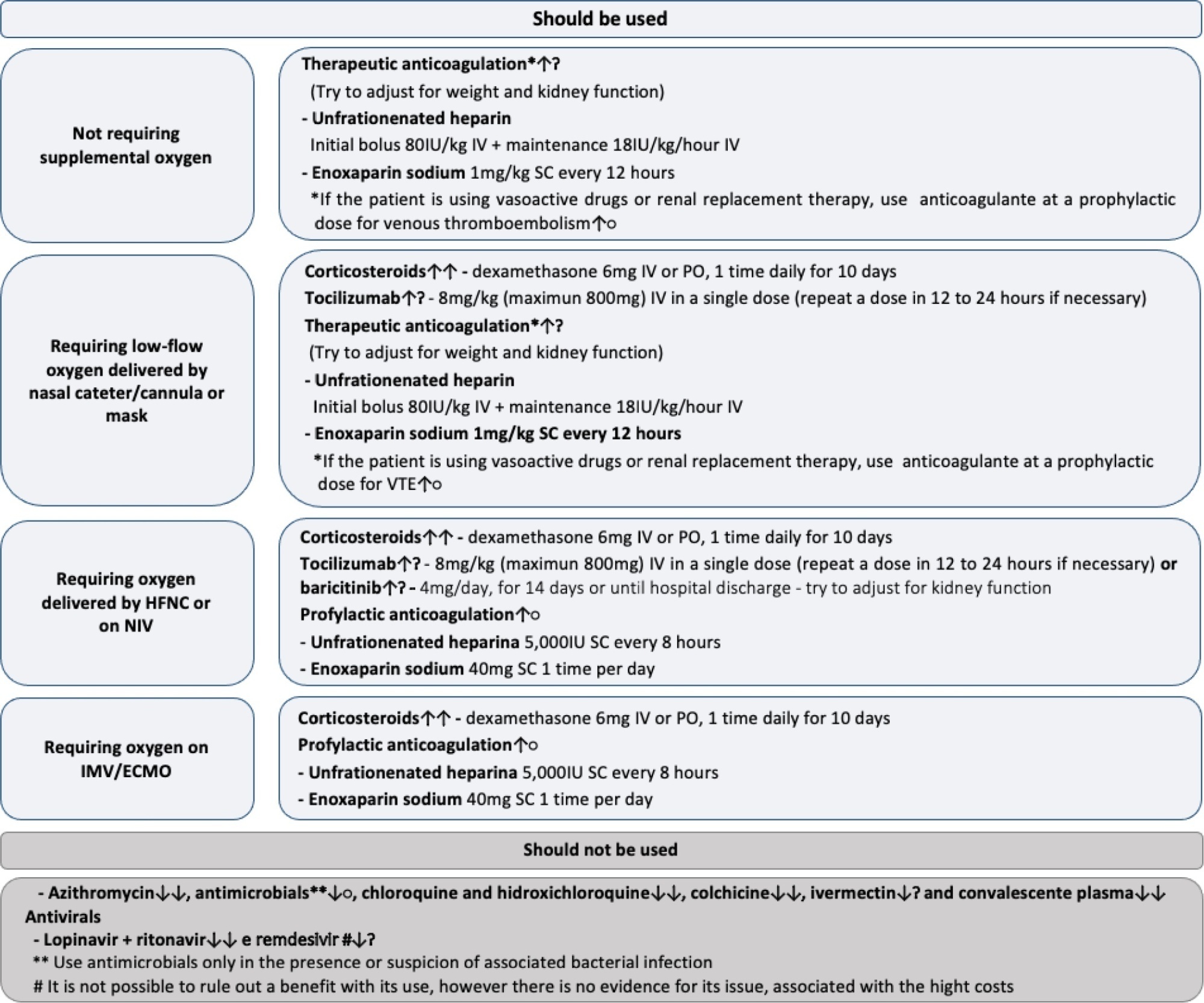
-
Special Article
Dapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
Abstract
Special ArticleDapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
DOI 10.5935/2965-2774.20230129-pt
Views9ABSTRACT
Background:
Critical illness is a major ongoing health care burden worldwide and is associated with high mortality rates. Sodium-glucose cotransporter-2 inhibitors have consistently shown benefits in cardiovascular and renal outcomes. The effects of sodium-glucose cotransporter-2 inhibitors in acute illness have not been properly investigated.
Methods:
DEFENDER is an investigator-initiated, multicenter, randomized, open-label trial designed to evaluate the efficacy and safety of dapagliflozin in 500 adult participants with acute organ dysfunction who are hospitalized in the intensive care unit. Eligible participants will be randomized 1:1 to receive dapagliflozin 10mg plus standard of care for up to 14 days or standard of care alone. The primary outcome is a hierarchical composite of hospital mortality, initiation of kidney replacement therapy, and intensive care unit length of stay, up to 28 days. Safety will be strictly monitored throughout the study.
Conclusion:
DEFENDER is the first study designed to investigate the use of a sodium-glucose cotransporter-2 inhibitor in general intensive care unit patients with acute organ dysfunction. It will provide relevant information on the use of drugs of this promising class in critically ill patients.
ClinicalTrials.gov registry:
NCT05558098
Keywords:Critical care outcomesCritical illnessOrgan dysfunctionSodium-glucose transporter 2 inhibitorsSee more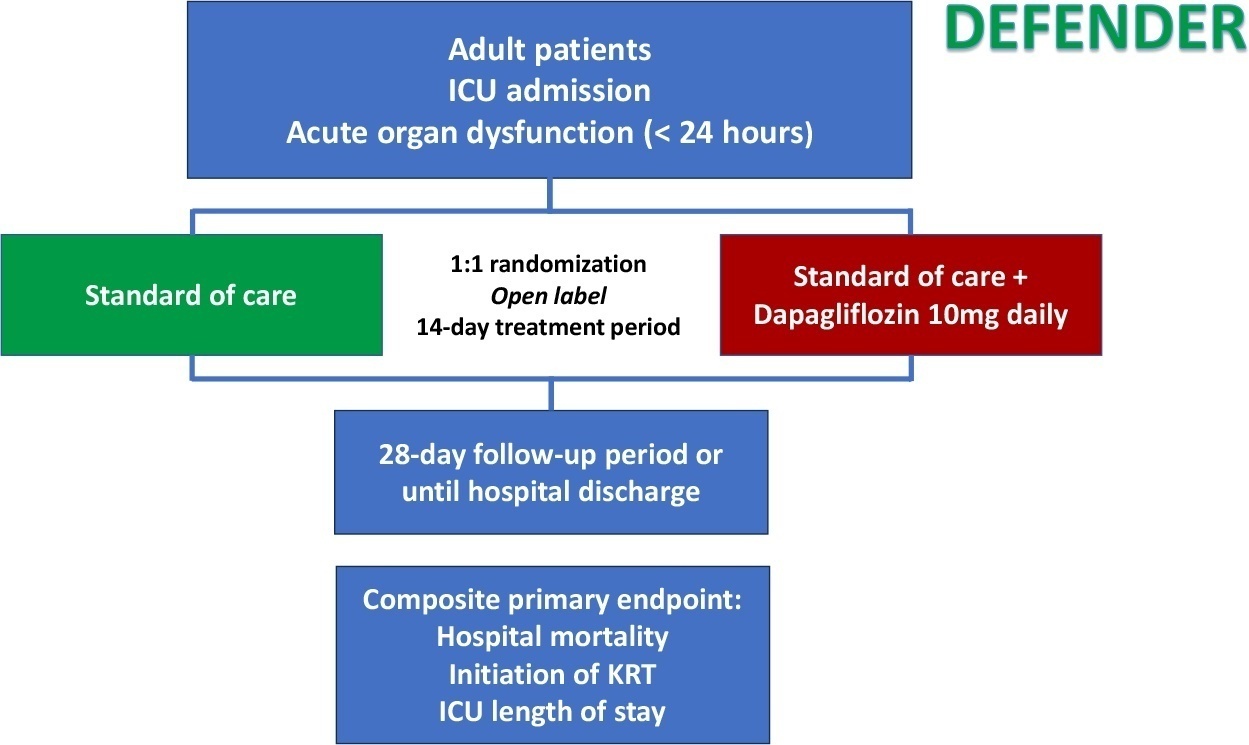
-
Commentary
Leveraging a national cloud-based intensive care registry for COVID-19 surveillance, research and case-mix evaluation in Brazil
Rev Bras Ter Intensiva. 2022;34(2):205-209
Abstract
CommentaryLeveraging a national cloud-based intensive care registry for COVID-19 surveillance, research and case-mix evaluation in Brazil
Rev Bras Ter Intensiva. 2022;34(2):205-209
DOI 10.5935/0103-507X.20220016-en
Views5INTRODUCTIONLarge national databases of intensive care units (ICUs) generate valuable information for the management and guidance of public policies.() These national ICU registries were established in high-income countries more than 20 years ago. Their contribution to the understanding of case mix and outcomes of ICU patients as well as to clinical research and quality improvement […]See more
-
Special Article
Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19: Joint guideline of Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia, Sociedade Brasileira de Reumatologia
Rev Bras Ter Intensiva. 2022;34(1):1-12
Abstract
Special ArticleBrazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19: Joint guideline of Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia, Sociedade Brasileira de Reumatologia
Rev Bras Ter Intensiva. 2022;34(1):1-12
DOI 10.5935/0103-507X.20220001-en
Views7See moreABSTRACT
Objective:
Several therapies are being used or proposed for COVID-19, and many lack appropriate evaluations of their effectiveness and safety. The purpose of this document is to develop recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Methods:
A group of 27 experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Results:
Sixteen recommendations were generated. They include strong recommendations for the use of corticosteroids in patients using supplemental oxygen, the use of anticoagulants at prophylactic doses to prevent thromboembolism and the nonuse of antibiotics in patients without suspected bacterial infection. It was not possible to make a recommendation regarding the use of tocilizumab in patients hospitalized with COVID-19 using oxygen due to uncertainties regarding the availability of and access to the drug. Strong recommendations against the use of hydroxychloroquine, convalescent plasma, colchicine, lopinavir + ritonavir and antibiotics in patients without suspected bacterial infection and also conditional recommendations against the use of casirivimab + imdevimab, ivermectin and rendesivir were made.
Conclusion:
To date, few therapies have proven effective in the treatment of hospitalized patients with COVID-19, and only corticosteroids and prophylaxis for thromboembolism are recommended. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and promote economical resource use.

-
Original Article
Epidemiology and outcome of high-surgical-risk patients admitted to an intensive care unit in Brazil
Rev Bras Ter Intensiva. 2020;32(1):17-27
Abstract
Original ArticleEpidemiology and outcome of high-surgical-risk patients admitted to an intensive care unit in Brazil
Rev Bras Ter Intensiva. 2020;32(1):17-27
DOI 10.5935/0103-507X.20200005
Views1ABSTRACT
Objective:
To define the epidemiological profile and the main determinants of morbidity and mortality in noncardiac high surgical risk patients in Brazil.
Methods:
This was a prospective, observational and multicenter study. All noncardiac surgical patients admitted to intensive care units, i.e., those considered high risk, within a 1-month period were evaluated and monitored daily for a maximum of 7 days in the intensive care unit to determine complications. The 28-day postoperative, intensive care unit and hospital mortality rates were evaluated.
Results:
Twenty-nine intensive care units participated in the study. Surgeries were performed in 25,500 patients, of whom 904 (3.5%) were high-risk (95% confidence interval – 95%CI 3.3% – 3.8%) and were included in the study. Of the participating patients, 48.3% were from private intensive care units, and 51.7% were from public intensive care units. The length of stay in the intensive care unit was 2.0 (1.0 – 4.0) days, and the length of hospital stay was 9.5 (5.4 – 18.6) days. The complication rate was 29.9% (95%CI 26.4 – 33.7), and the 28-day postoperative mortality rate was 9.6% (95%CI 7.4 – 12.1). The independent risk factors for complications were the Simplified Acute Physiology Score 3 (SAPS 3; odds ratio – OR = 1.02; 95%CI 1.01 – 1.03) and Sequential Organ Failure Assessment Score (SOFA) on admission to the intensive care unit (OR = 1.17; 95%CI 1.09 – 1.25), surgical time (OR = 1.001, 95%CI 1.000 – 1.002) and emergency surgeries (OR = 1.93, 95%CI, 1.10 – 3.38). In addition, there were associations with 28-day mortality (OR = 1.032; 95%CI 1.011 – 1.052), SAPS 3 (OR = 1.041; 95%CI 1.107 – 1.279), SOFA (OR = 1.175, 95%CI 1.069 – 1.292) and emergency surgeries (OR = 2.509; 95%CI 1.040 – 6.051).
Conclusion:
Higher prognostic scores, elderly patients, longer surgical times and emergency surgeries were strongly associated with higher 28-day mortality and more complications during the intensive care unit stay.
Keywords:BrazilIntensive care unitspostoperative carePostoperative complications/mortalitySurgical procedures, operative/epidemiologySurgical procedures, operative/mortalitySee more
-
Original Article
Adverse outcomes of delayed intensive care unit
Rev Bras Ter Intensiva. 2020;32(1):92-98
Abstract
Original ArticleAdverse outcomes of delayed intensive care unit
Rev Bras Ter Intensiva. 2020;32(1):92-98
DOI 10.5935/0103-507X.20200014
Views1See moreABSTRACT
Objective:
To examine the impact of delayed transfer from the emergency room into the intensive care unit on the length of intensive care unit stay and death.
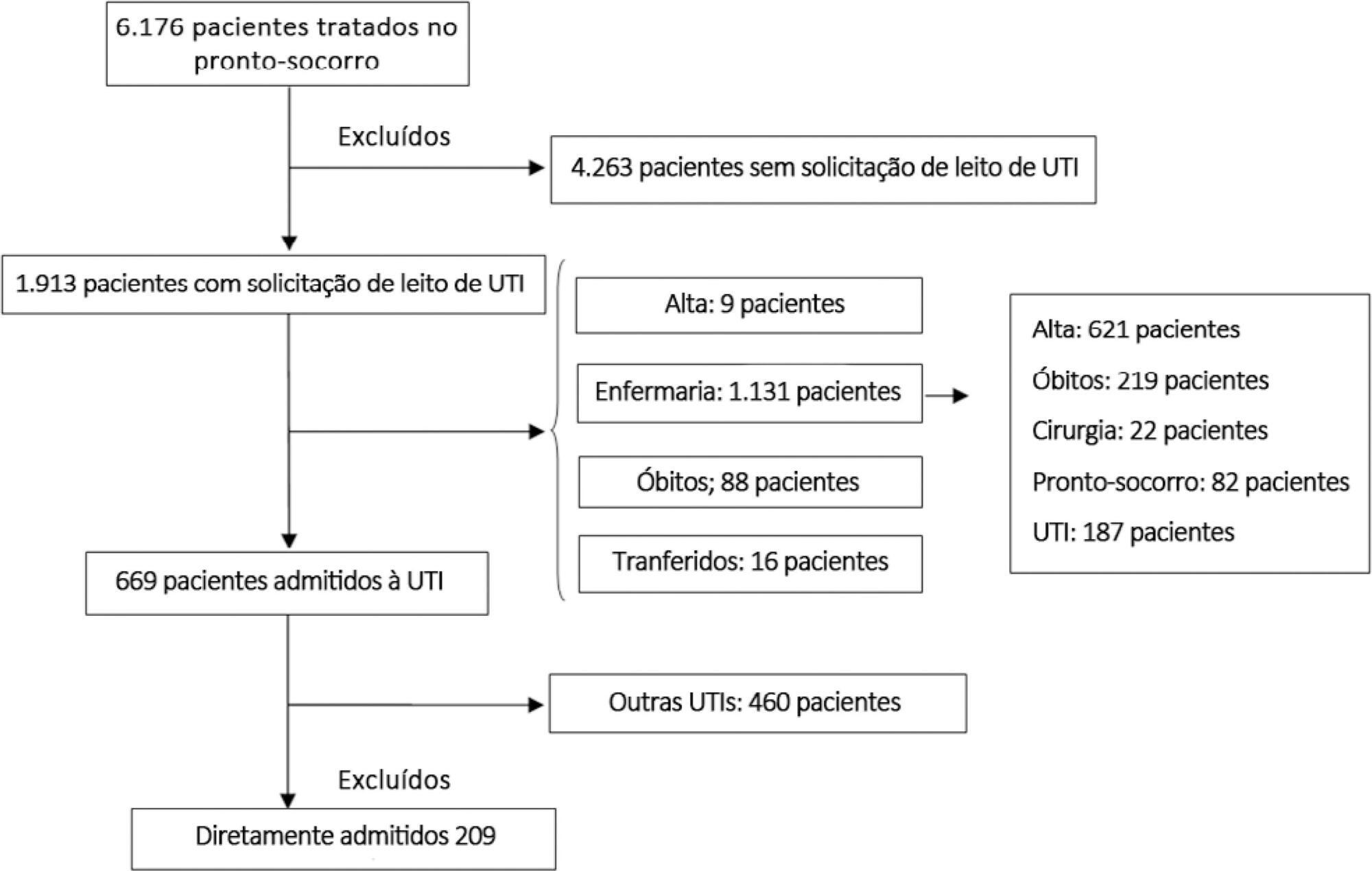
Methods:
This prospective, cohort study performed in a tertiary academic hospital obtained data from 1913 patients admitted to the emergency room with a documented request for admission into the intensive care unit. The patients admitted directly into the medical-surgical intensive care unit (n = 209) were categorized into tertiles according to their waiting time for intensive care unit admission (Group 1: < 637 min, Group 2: 637 to 1602 min, and Group 3: > 1602 min). Patients who stayed in the intensive care unit for longer than 3.2 days (median time of intensive care unit length of stay of all patients) were considered as having a prolonged intensive care unit stay.
Results:
A total of 6,176 patients were treated in the emergency room during the study period, among whom 1,913 (31%) required a bed in the intensive care unit. The median length of stay in the emergency room was 17 hours [9 to 33 hours]. Hospitalization for infection/sepsis was an independent predictor of prolonged intensive care unit stay (OR 2.75 95%CI 1.38 – 5.48, p = 0.004), but waiting time for intensive care unit admission was not. The mortality rate was higher in Group 3 (38%) than in Group 1 (31%) but the difference was not statistically significant.
Conclusion:
Delayed admission into the intensive care unit from the emergency room did not result in an increased intensive care unit stay or mortality.
-
Editorials
Brazilian guidelines in critical care: let’s face this challenge…
Rev Bras Ter Intensiva. 2016;28(3):213-214
Abstract
EditorialsBrazilian guidelines in critical care: let’s face this challenge…
Rev Bras Ter Intensiva. 2016;28(3):213-214
DOI 10.5935/0103-507X.20160046
Views0Clinical practice guidelines are useful tools to improve delivery of the best care, based on the best available evidence, for our patients. They help practitioners to make clinical decisions and might help to ensure the proper allocation of resources in public health policy.() Therefore, health care guidelines and their appropriate implementation are of interest to […]See more -
Original Articles
Septic shock: a major cause of hospital death after intensive care unit discharge
Rev Bras Ter Intensiva. 2015;27(1):51-56
Abstract
Original ArticlesSeptic shock: a major cause of hospital death after intensive care unit discharge
Rev Bras Ter Intensiva. 2015;27(1):51-56
DOI 10.5935/0103-507X.20150009
Views0See moreObjective:
To assess the causes and factors associated with the death of patients between intensive care unit discharge and hospital discharge.
Methods:
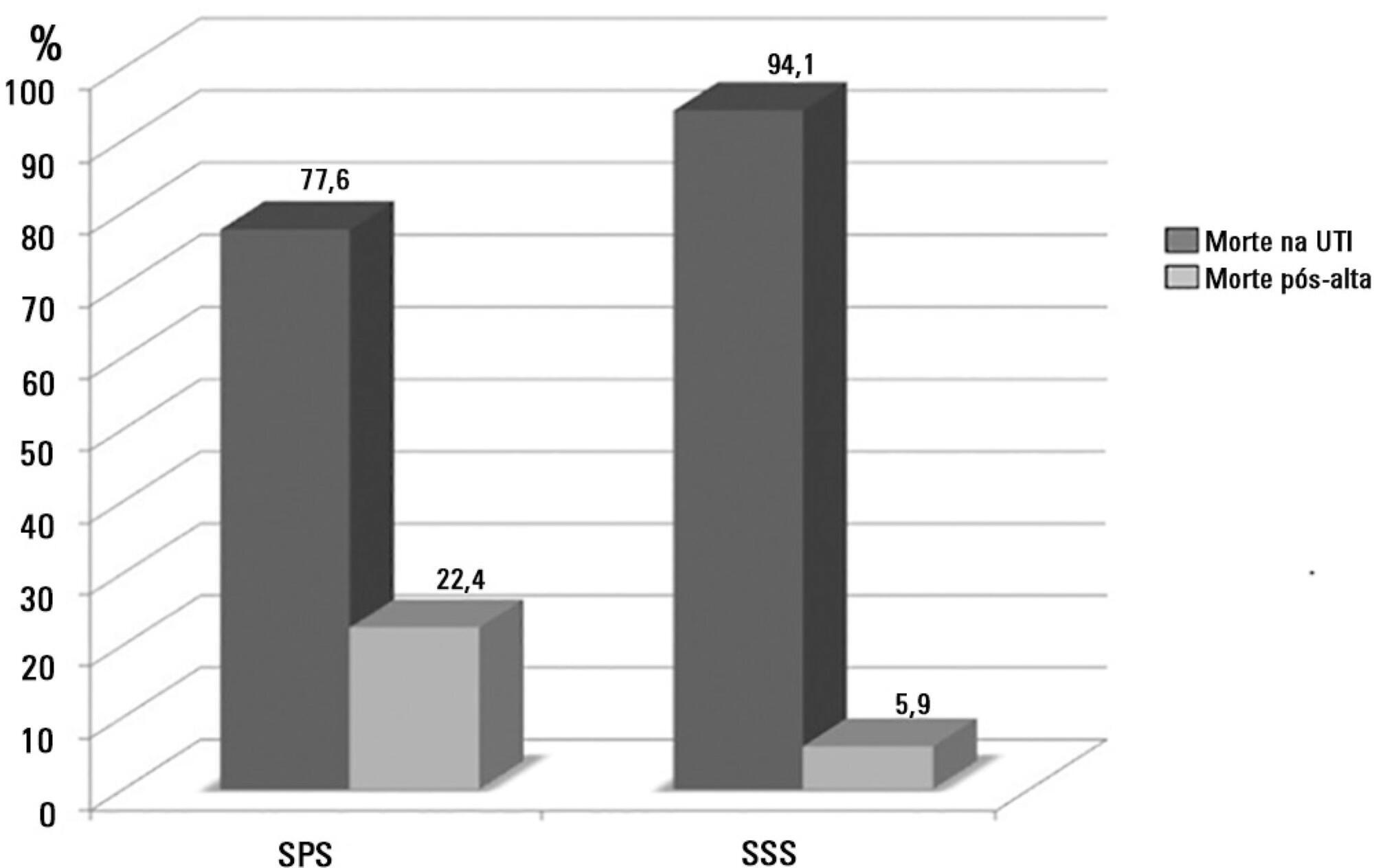
The present is a pilot, retrospective, observational cohort study. The records of all patients admitted to two units of a public/private university hospital from February 1, 2013 to April 30, 2013 were assessed. Demographic and clinical data, risk scores and outcomes were obtained from the Epimed monitoring system and confirmed in the electronic record system of the hospital. The relative risk and respective confidence intervals were calculated.
Results:
A total of 581 patients were evaluated. The mortality rate in the intensive care unit was 20.8% and in the hospital was 24.9%. Septic shock was the cause of death in 58.3% of patients who died after being discharged from the intensive care unit. Of the patients from the public health system, 73 (77.6%) died in the intensive care unit and 21 (22.4%) died in the hospital after being discharged from the unit. Of the patients from the Supplementary Health System, 48 (94.1%) died in the intensive care unit and 3 (5.9%) died in the hospital after being discharged from the unit (relative risk, 3.87%; 95% confidence interval, 1.21 – 12.36; p < 0.05). The post-discharge mortality rate was significantly higher in patients with intensive care unit hospitalization time longer than 6 days.
Conclusion:
The main cause of death of patients who were discharged from the intensive care unit and died in the ward before hospital discharge was septic shock. Coverage by the public healthcare system and longer hospitalization time in the intensive care unit were factors associated with death after discharge from the intensive care unit.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




