You searched for:"Adriano José Pereira"
We found (3) results for your search.-
Special Article
Dapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
Abstract
Special ArticleDapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
DOI 10.5935/2965-2774.20230129-pt
Views9ABSTRACT
Background:
Critical illness is a major ongoing health care burden worldwide and is associated with high mortality rates. Sodium-glucose cotransporter-2 inhibitors have consistently shown benefits in cardiovascular and renal outcomes. The effects of sodium-glucose cotransporter-2 inhibitors in acute illness have not been properly investigated.
Methods:
DEFENDER is an investigator-initiated, multicenter, randomized, open-label trial designed to evaluate the efficacy and safety of dapagliflozin in 500 adult participants with acute organ dysfunction who are hospitalized in the intensive care unit. Eligible participants will be randomized 1:1 to receive dapagliflozin 10mg plus standard of care for up to 14 days or standard of care alone. The primary outcome is a hierarchical composite of hospital mortality, initiation of kidney replacement therapy, and intensive care unit length of stay, up to 28 days. Safety will be strictly monitored throughout the study.
Conclusion:
DEFENDER is the first study designed to investigate the use of a sodium-glucose cotransporter-2 inhibitor in general intensive care unit patients with acute organ dysfunction. It will provide relevant information on the use of drugs of this promising class in critically ill patients.
ClinicalTrials.gov registry:
NCT05558098
Keywords:Critical care outcomesCritical illnessOrgan dysfunctionSodium-glucose transporter 2 inhibitorsSee more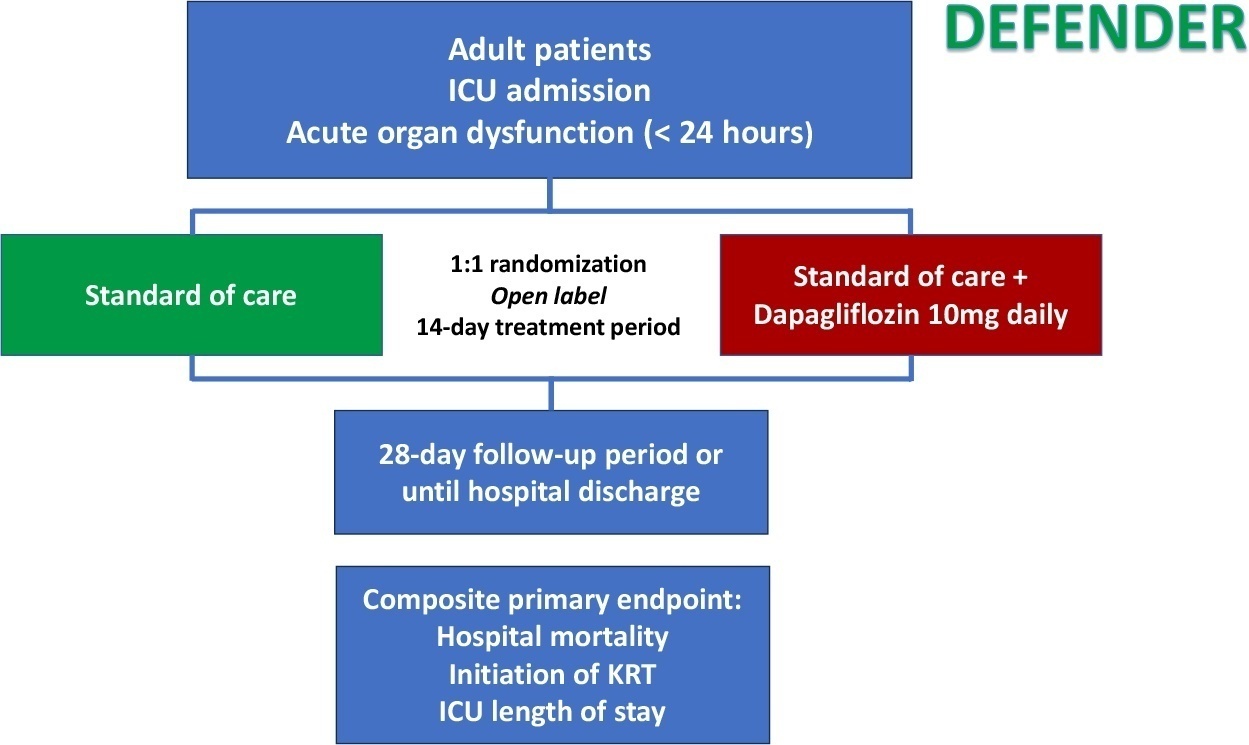
-
Letter to the Editor
Patient-level costs of central line-associated bloodstream infections caused by multidrug-resistant microorganisms in a public intensive care unit in Brazil: a retrospective cohort study
Rev Bras Ter Intensiva. 2022;34(4):529-533
Abstract
Letter to the EditorPatient-level costs of central line-associated bloodstream infections caused by multidrug-resistant microorganisms in a public intensive care unit in Brazil: a retrospective cohort study
Rev Bras Ter Intensiva. 2022;34(4):529-533
DOI 10.5935/0103-507X.20220313-en
Views1TO THE EDITOR, Hospital-acquired infections (HAIs) are a major threat to patients and health care systems.() Hospital-acquired infections are associated with increased mortality and prolonged hospital length of stay.() However, it is not clear whether HAIs caused by multidrug-resistant (MDR) pathogens acquired in intensive care units (ICUs) are associated with increased costs when compared to […]See more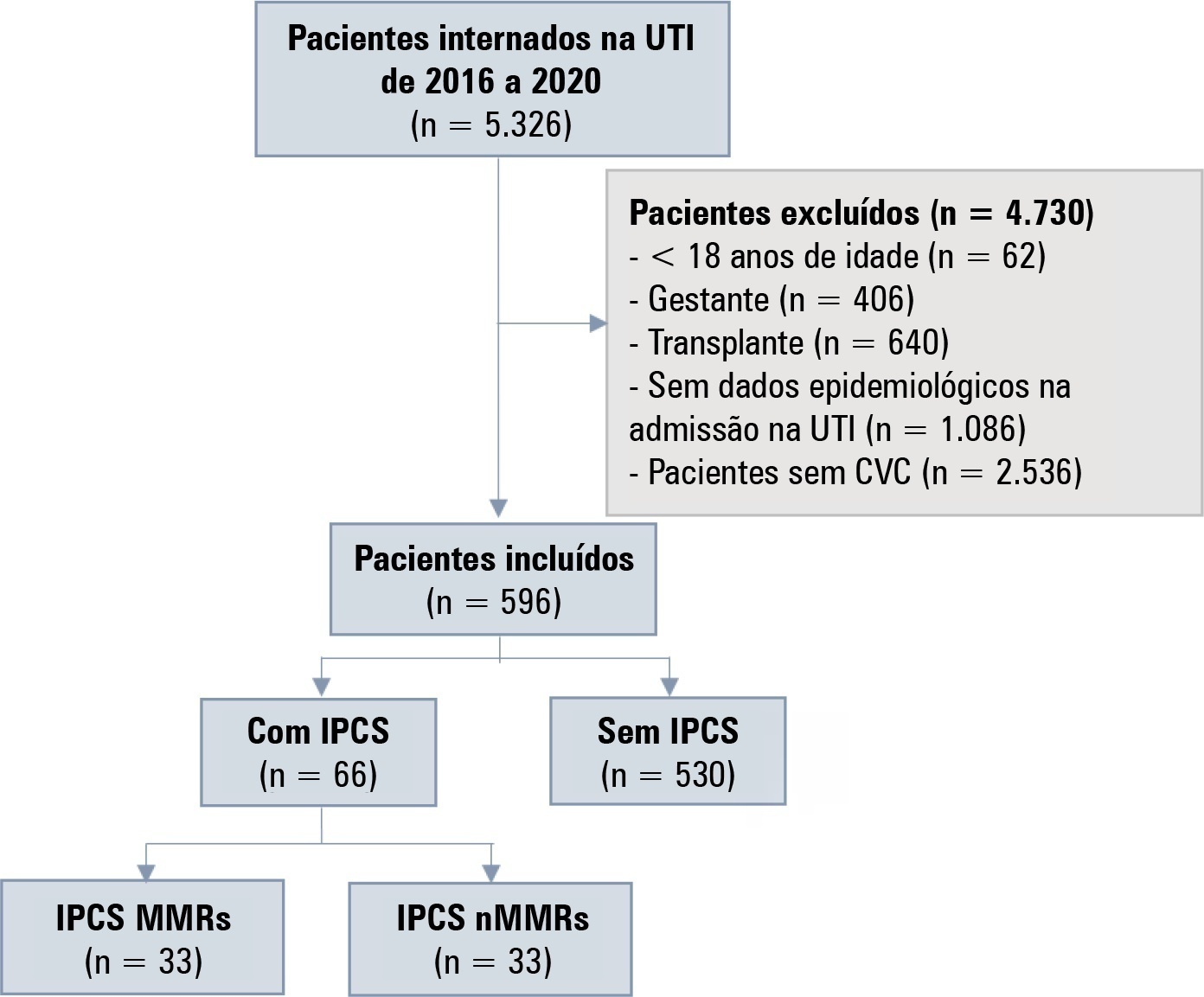
-
Special Article
Statistical analysis of a cluster-randomized clinical trial on adult general intensive care units in Brazil: TELE-critical care verSus usual Care On ICU PErformance (TELESCOPE) trial
Rev Bras Ter Intensiva. 2022;34(1):87-95
Abstract
Special ArticleStatistical analysis of a cluster-randomized clinical trial on adult general intensive care units in Brazil: TELE-critical care verSus usual Care On ICU PErformance (TELESCOPE) trial
Rev Bras Ter Intensiva. 2022;34(1):87-95
DOI 10.5935/0103-507x.20220003-en
Views2ABSTRACT
Objective:
The TELE-critical Care verSus usual Care On ICU PErformance (TELESCOPE) trial aims to assess whether a complex telemedicine intervention in intensive care units, which focuses on daily multidisciplinary rounds performed by remote intensivists, will reduce intensive care unit length of stay compared to usual care.
Methods:
The TELESCOPE trial is a national, multicenter, controlled, open label, cluster randomized trial. The study tests the effectiveness of daily multidisciplinary rounds conducted by an intensivist through telemedicine in Brazilian intensive care units. The protocol was approved by the local Research Ethics Committee of the coordinating study center and by the local Research Ethics Committee from each of the 30 intensive care units, following Brazilian legislation. The trial is registered with ClinicalTrials. gov (NCT03920501). The primary outcome is intensive care unit length of stay, which will be analyzed accounting for the baseline period and cluster structure of the data and adjusted by prespecified covariates. Secondary exploratory outcomes included intensive care unit performance classification, in-hospital mortality, incidence of nosocomial infections, ventilator-free days at 28 days, rate of patients receiving oral or enteral feeding, rate of patients under light sedation or alert and calm, and rate of patients under normoxemia.
Conclusion:
According to the trial’s best practice, we report our statistical analysis prior to locking the database and beginning analyses. We anticipate that this reporting practice will prevent analysis bias and improve the interpretation of the reported results.
Keywords:BrazilCritical careData interpretation, statisticalHospital mortalityIntensive care unitsLength of stayPatient care teamResearch designTelemedicineSee more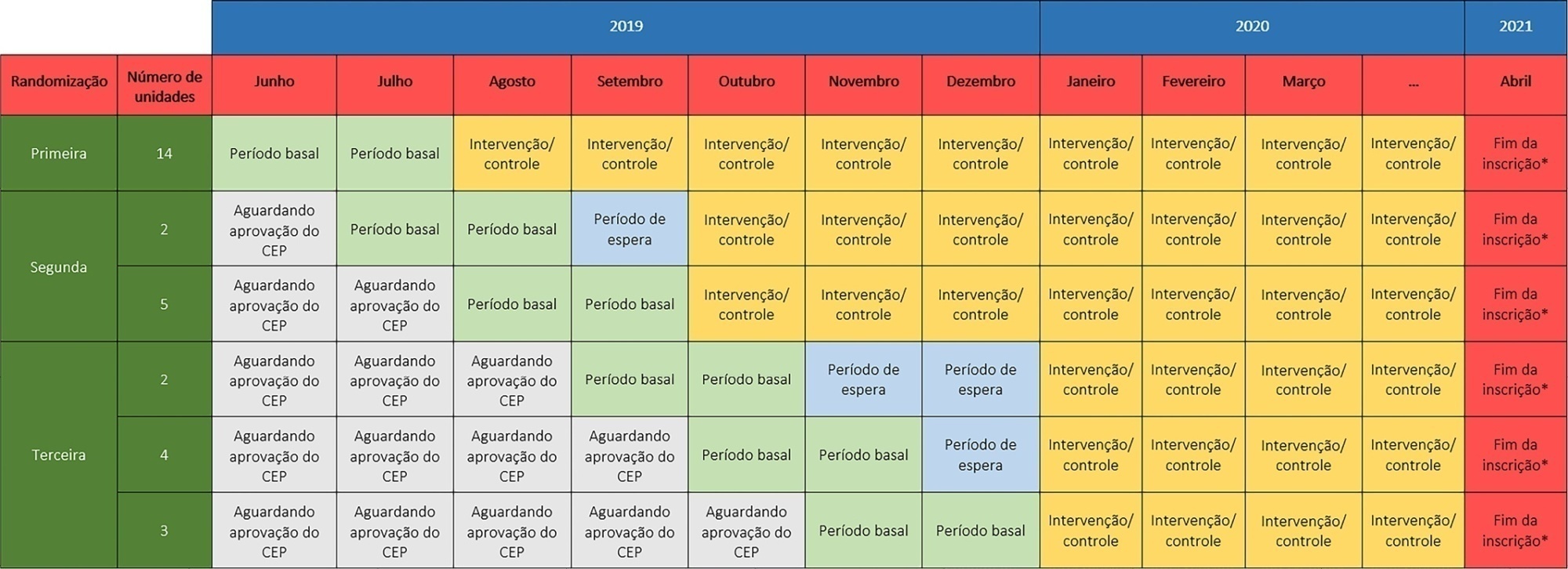
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




