You searched for:"Alexandre Biasi Cavalcanti"
We found (31) results for your search.-
Special Article
Statistical analysis of a cluster-randomized clinical trial on adult general intensive care units in Brazil: TELE-critical care verSus usual Care On ICU PErformance (TELESCOPE) trial
Rev Bras Ter Intensiva. 2022;34(1):87-95
Abstract
Special ArticleStatistical analysis of a cluster-randomized clinical trial on adult general intensive care units in Brazil: TELE-critical care verSus usual Care On ICU PErformance (TELESCOPE) trial
Rev Bras Ter Intensiva. 2022;34(1):87-95
DOI 10.5935/0103-507x.20220003-en
Views2ABSTRACT
Objective:
The TELE-critical Care verSus usual Care On ICU PErformance (TELESCOPE) trial aims to assess whether a complex telemedicine intervention in intensive care units, which focuses on daily multidisciplinary rounds performed by remote intensivists, will reduce intensive care unit length of stay compared to usual care.
Methods:
The TELESCOPE trial is a national, multicenter, controlled, open label, cluster randomized trial. The study tests the effectiveness of daily multidisciplinary rounds conducted by an intensivist through telemedicine in Brazilian intensive care units. The protocol was approved by the local Research Ethics Committee of the coordinating study center and by the local Research Ethics Committee from each of the 30 intensive care units, following Brazilian legislation. The trial is registered with ClinicalTrials. gov (NCT03920501). The primary outcome is intensive care unit length of stay, which will be analyzed accounting for the baseline period and cluster structure of the data and adjusted by prespecified covariates. Secondary exploratory outcomes included intensive care unit performance classification, in-hospital mortality, incidence of nosocomial infections, ventilator-free days at 28 days, rate of patients receiving oral or enteral feeding, rate of patients under light sedation or alert and calm, and rate of patients under normoxemia.
Conclusion:
According to the trial’s best practice, we report our statistical analysis prior to locking the database and beginning analyses. We anticipate that this reporting practice will prevent analysis bias and improve the interpretation of the reported results.
Keywords:BrazilCritical careData interpretation, statisticalHospital mortalityIntensive care unitsLength of stayPatient care teamResearch designTelemedicineSee more
-
Special Article
Hemodynamic phenotype-based, capillary refill time-targeted resuscitation in early septic shock: The ANDROMEDA-SHOCK-2 Randomized Clinical Trial study protocol
Rev Bras Ter Intensiva. 2022;34(1):96-106
Abstract
Special ArticleHemodynamic phenotype-based, capillary refill time-targeted resuscitation in early septic shock: The ANDROMEDA-SHOCK-2 Randomized Clinical Trial study protocol
Rev Bras Ter Intensiva. 2022;34(1):96-106
DOI 10.5935/0103-507X.20220004-en
Views5ABSTRACT
Background:
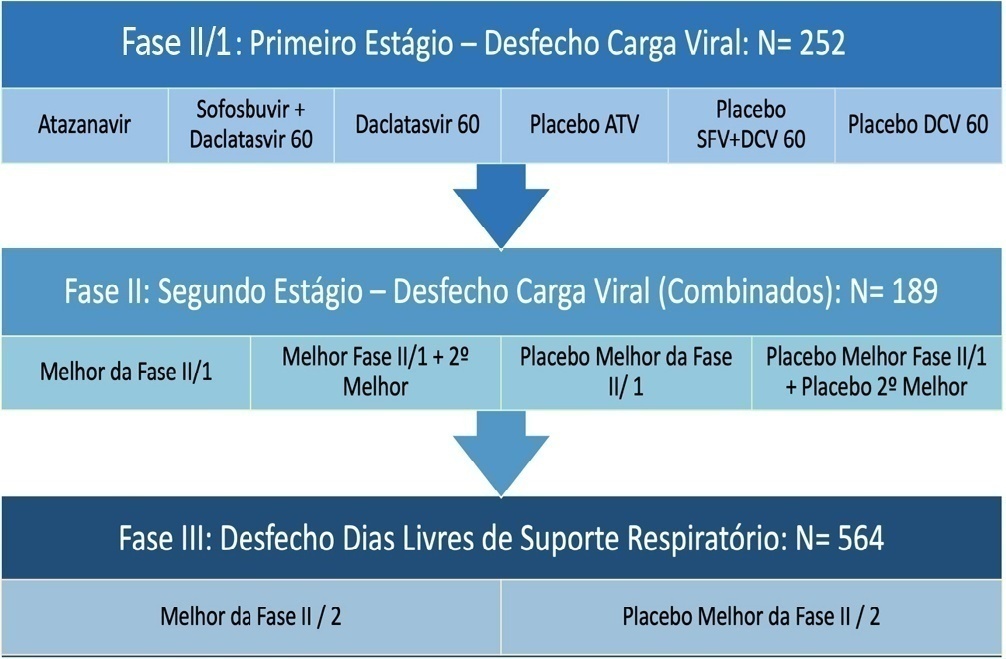
Early reversion of sepsis-induced tissue hypoperfusion is essential for survival in septic shock. However, consensus regarding the best initial resuscitation strategy is lacking given that interventions designed for the entire population with septic shock might produce unnecessary fluid administration. This article reports the rationale, study design and analysis plan of the ANDROMEDA-2 study, which aims to determine whether a peripheral perfusion-guided strategy consisting of capillary refill time-targeted resuscitation based on clinical and hemodynamic phenotypes is associated with a decrease in a composite outcome of mortality, time to organ support cessation, and hospital length of stay compared to standard care in patients with early (< 4 hours of diagnosis) septic shock.
Methods:
The ANDROMEDA-2 study is a multicenter, multinational randomized controlled trial. In the intervention group, capillary refill time will be measured hourly for 6 hours. If abnormal, patients will enter an algorithm starting with pulse pressure assessment. Patients with pulse pressure less than 40mmHg will be tested for fluid responsiveness and receive fluids accordingly. In patients with pulse pressure > 40mmHg, norepinephrine will be titrated to maintain diastolic arterial pressure > 50mmHg. Patients who fail to normalize capillary refill time after the previous steps will be subjected to critical care echocardiography for cardiac dysfunction evaluation and subsequent management. Finally, vasopressor and inodilator tests will be performed to further optimize perfusion. A sample size of 1,500 patients will provide 88% power to demonstrate superiority of the capillary refill time-targeted strategy.
Conclusions:
If hemodynamic phenotype-based, capillary refill time-targeted resuscitation demonstrates to be a superior strategy, care processes in septic shock resuscitation can be optimized with bedside tools.
Keywords:AlgorithmCapillary refill timeCritical careEchocardiographyLength of staynorepinephrinePerfusionPhenotypeSepsisSeptic shockSee more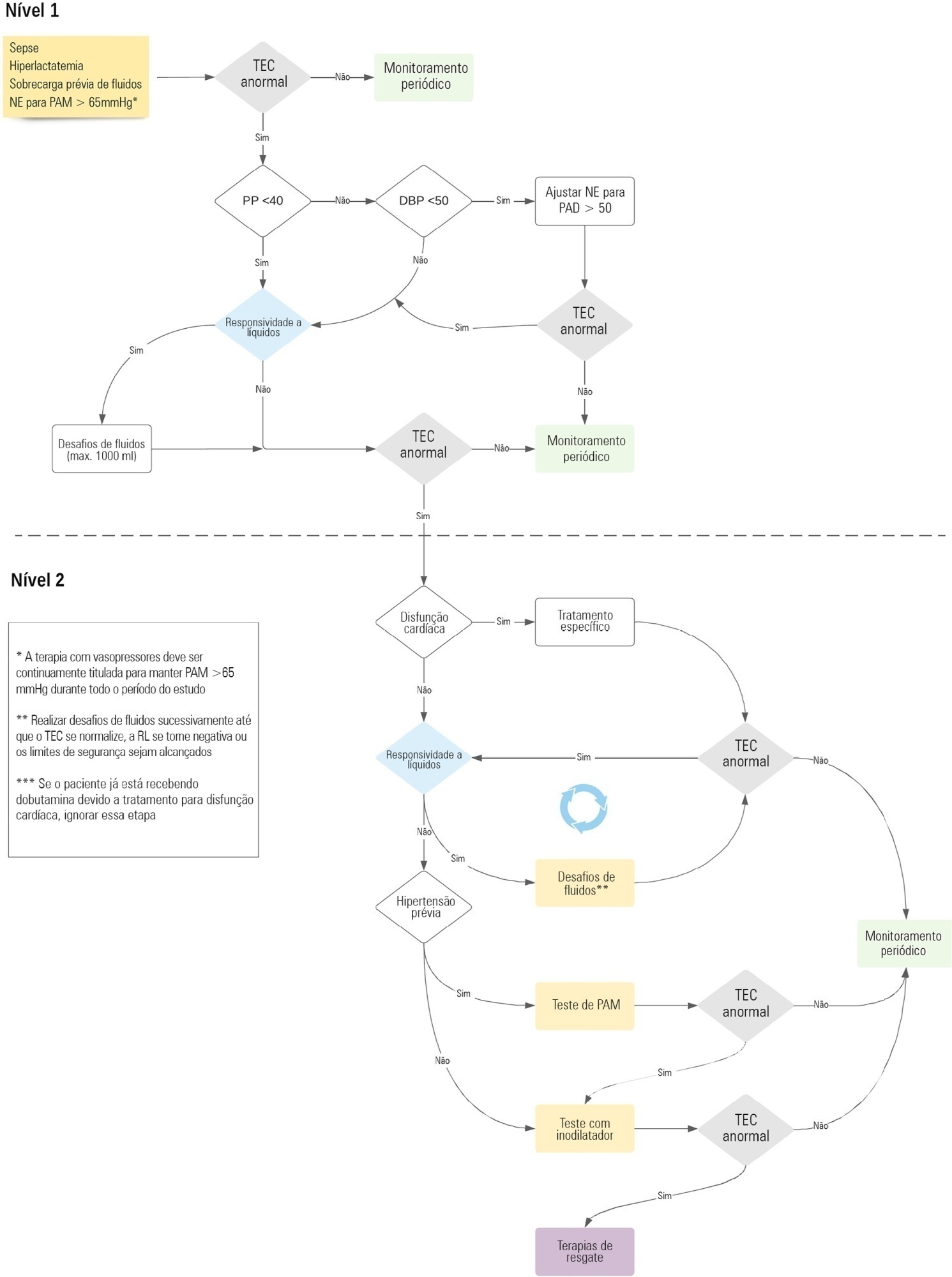
-
Letter to the Editor
Epistaxis as a complication of high-flow nasal cannula therapy in adults
Rev Bras Ter Intensiva. 2021;33(4):640-643
Abstract
Letter to the EditorEpistaxis as a complication of high-flow nasal cannula therapy in adults
Rev Bras Ter Intensiva. 2021;33(4):640-643
DOI 10.5935/0103-507X.20210090
Views1INTRODUCTIONHigh-flow nasal cannula (HFNC) therapy has emerged as a valuable therapy for adult patients with acute respiratory failure(,) or to prevent postextubation respiratory failure.(,) Currently, HFNC therapy is recommended in guidelines on the management of patients with coronavirus 2019 disease (COVID-19).() HFNC therapy delivers humidified and heated gas at a high flow that can exceed […]See more -
Original Article
Resuscitation fluid practices in Brazilian intensive care units: a secondary analysis of Fluid-TRIPS
Rev Bras Ter Intensiva. 2021;33(2):206-218
Abstract
Original ArticleResuscitation fluid practices in Brazilian intensive care units: a secondary analysis of Fluid-TRIPS
Rev Bras Ter Intensiva. 2021;33(2):206-218
DOI 10.5935/0103-507X.20210028
Views1See moreAbstract
Objective:
To describe fluid resuscitation practices in Brazilian intensive care units and to compare them with those of other countries participating in the Fluid-TRIPS.
Methods:
This was a prospective, international, cross-sectional, observational study in a convenience sample of intensive care units in 27 countries (including Brazil) using the Fluid-TRIPS database compiled in 2014. We described the patterns of fluid resuscitation use in Brazil compared with those in other countries and identified the factors associated with fluid choice.
Results:
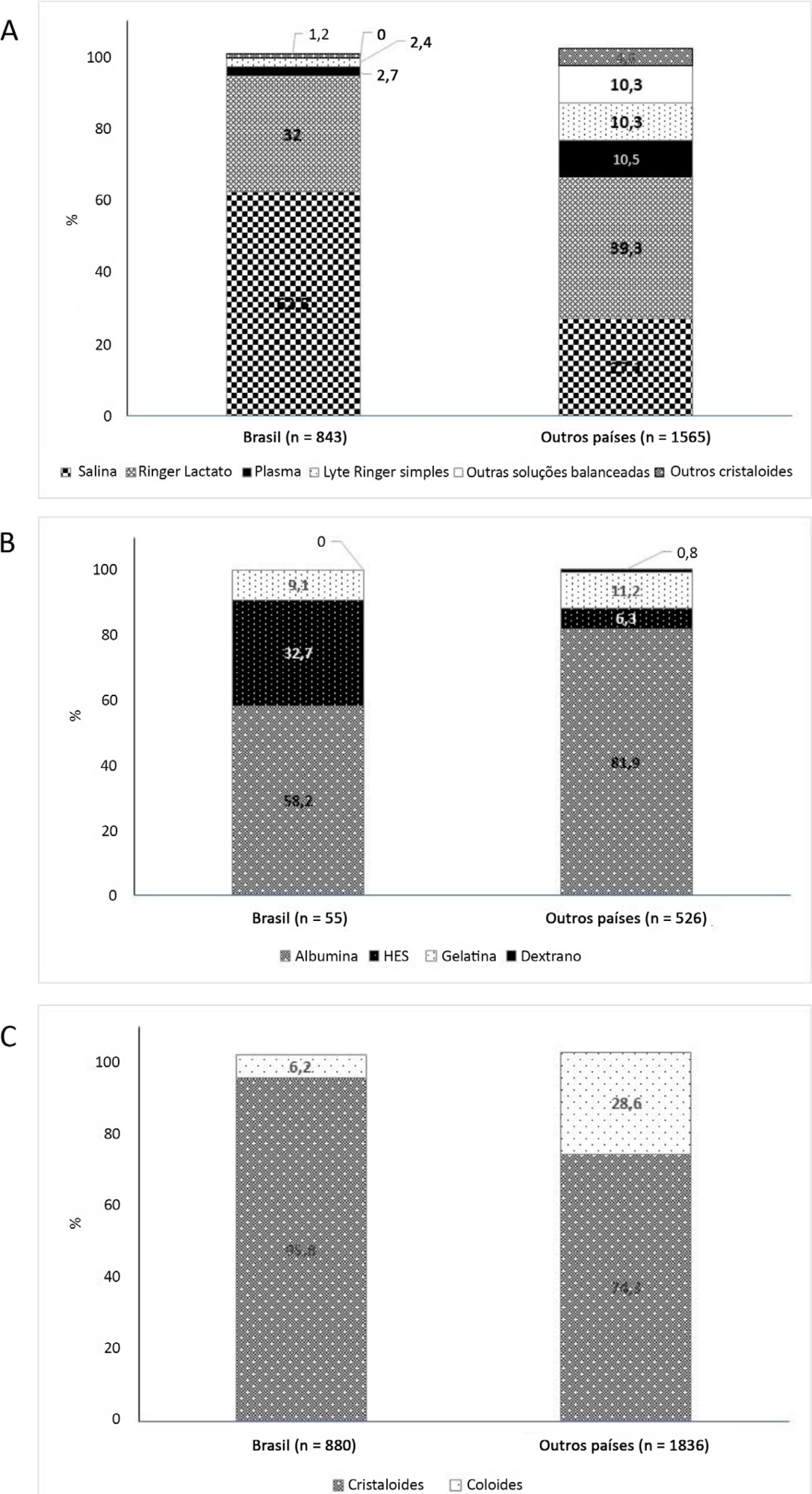
On the study day, 3,214 patients in Brazil and 3,493 patients in other countries were included, of whom 16.1% and 26.8% (p < 0.001) received fluids, respectively. The main indication for fluid resuscitation was impaired perfusion and/or low cardiac output (Brazil: 71.7% versus other countries: 56.4%, p < 0.001). In Brazil, the percentage of patients receiving crystalloid solutions was higher (97.7% versus 76.8%, p < 0.001), and 0.9% sodium chloride was the most commonly used crystalloid (62.5% versus 27.1%, p < 0.001). The multivariable analysis suggested that the albumin levels were associated with the use of both crystalloids and colloids, whereas the type of fluid prescriber was associated with crystalloid use only.
Conclusion:
Our results suggest that crystalloids are more frequently used than colloids for fluid resuscitation in Brazil, and this discrepancy in frequencies is higher than that in other countries. Sodium chloride (0.9%) was the crystalloid most commonly prescribed. Serum albumin levels and the type of fluid prescriber were the factors associated with the choice of crystalloids or colloids for fluid resuscitation.
-
Special Article
Brazilian guidelines for the management of brain-dead potential organ donors. The task force of the Associação de Medicina Intensiva Brasileira, Associação Brasileira de Transplantes de Órgãos, Brazilian Research in Critical Care Network, and the General Coordination of the National Transplant System
Rev Bras Ter Intensiva. 2021;33(1):1-11
Abstract
Special ArticleBrazilian guidelines for the management of brain-dead potential organ donors. The task force of the Associação de Medicina Intensiva Brasileira, Associação Brasileira de Transplantes de Órgãos, Brazilian Research in Critical Care Network, and the General Coordination of the National Transplant System
Rev Bras Ter Intensiva. 2021;33(1):1-11
DOI 10.5935/0103-507X.20210001
Views5See moreAbstract
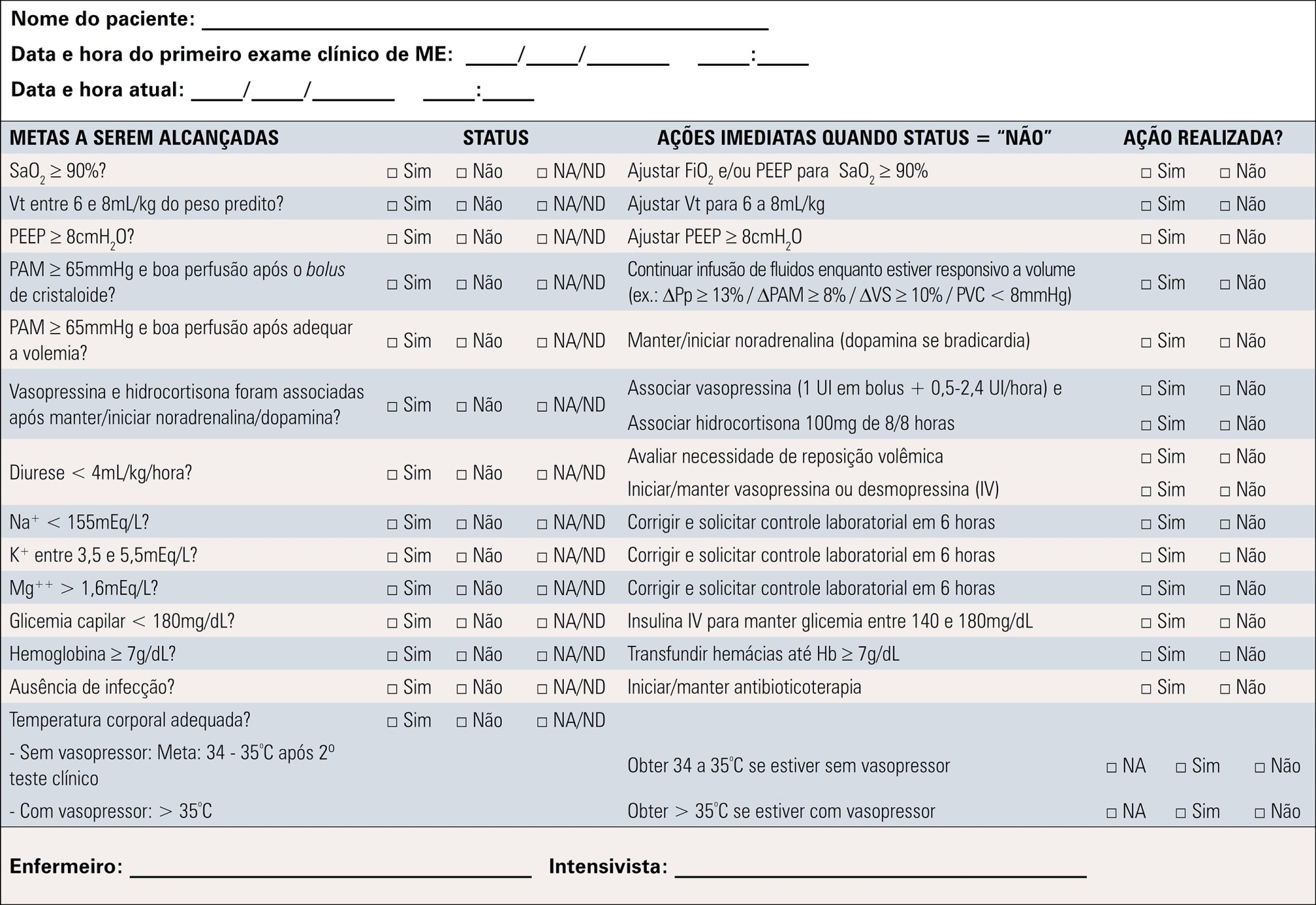
Objective:
To contribute to updating the recommendations for brain-dead potential organ donor management.
Methods:
A group of 27 experts, including intensivists, transplant coordinators, transplant surgeons, and epidemiologists, answered questions related to the following topics were divided into mechanical ventilation, hemodynamics, endocrine-metabolic management, infection, body temperature, blood transfusion, and checklists use. The outcomes considered were cardiac arrests, number of organs removed or transplanted as well as function / survival of transplanted organs. The quality of evidence of the recommendations was assessed using the Grading of Recommendations Assessment, Development, and Evaluation system to classify the recommendations.
Results:
A total of 19 recommendations were drawn from the expert panel. Of these, 7 were classified as strong, 11 as weak and 1 was considered a good clinical practice.
Conclusion:
Despite the agreement among panel members on most recommendations, the grade of recommendation was mostly weak.
-
Special Article
Quality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
Abstract
Special ArticleQuality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
Views0See moreAbstract
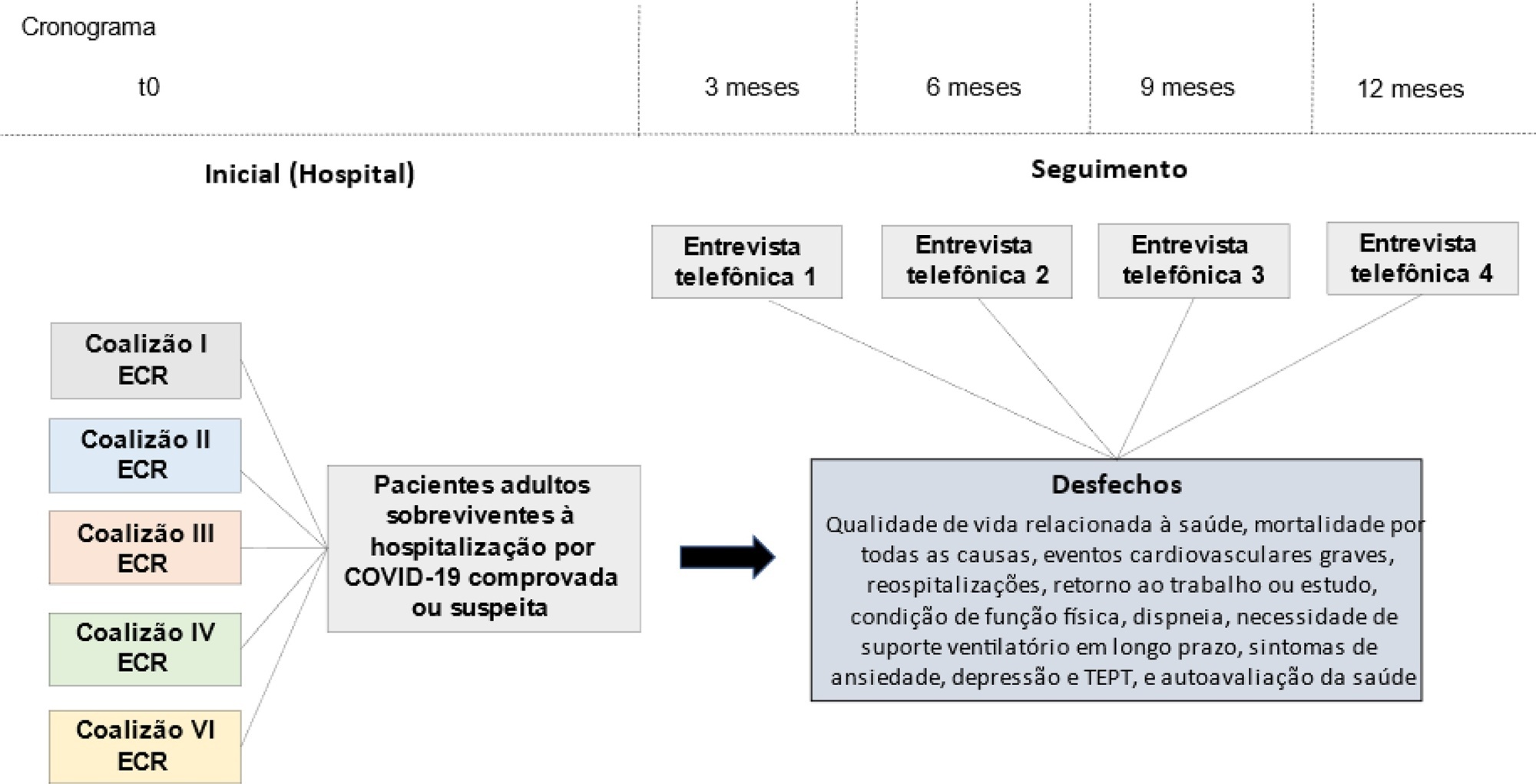
Introduction:
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
Methods:
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
Ethics and dissemination:
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
-
Original Article
Statistical analysis plan for the Balanced Solution versus Saline in Intensive Care Study (BaSICS)
Rev Bras Ter Intensiva. 2020;32(4):493-505
Abstract
Original ArticleStatistical analysis plan for the Balanced Solution versus Saline in Intensive Care Study (BaSICS)
Rev Bras Ter Intensiva. 2020;32(4):493-505
DOI 10.5935/0103-507X.20200081
Views1See moreAbstract
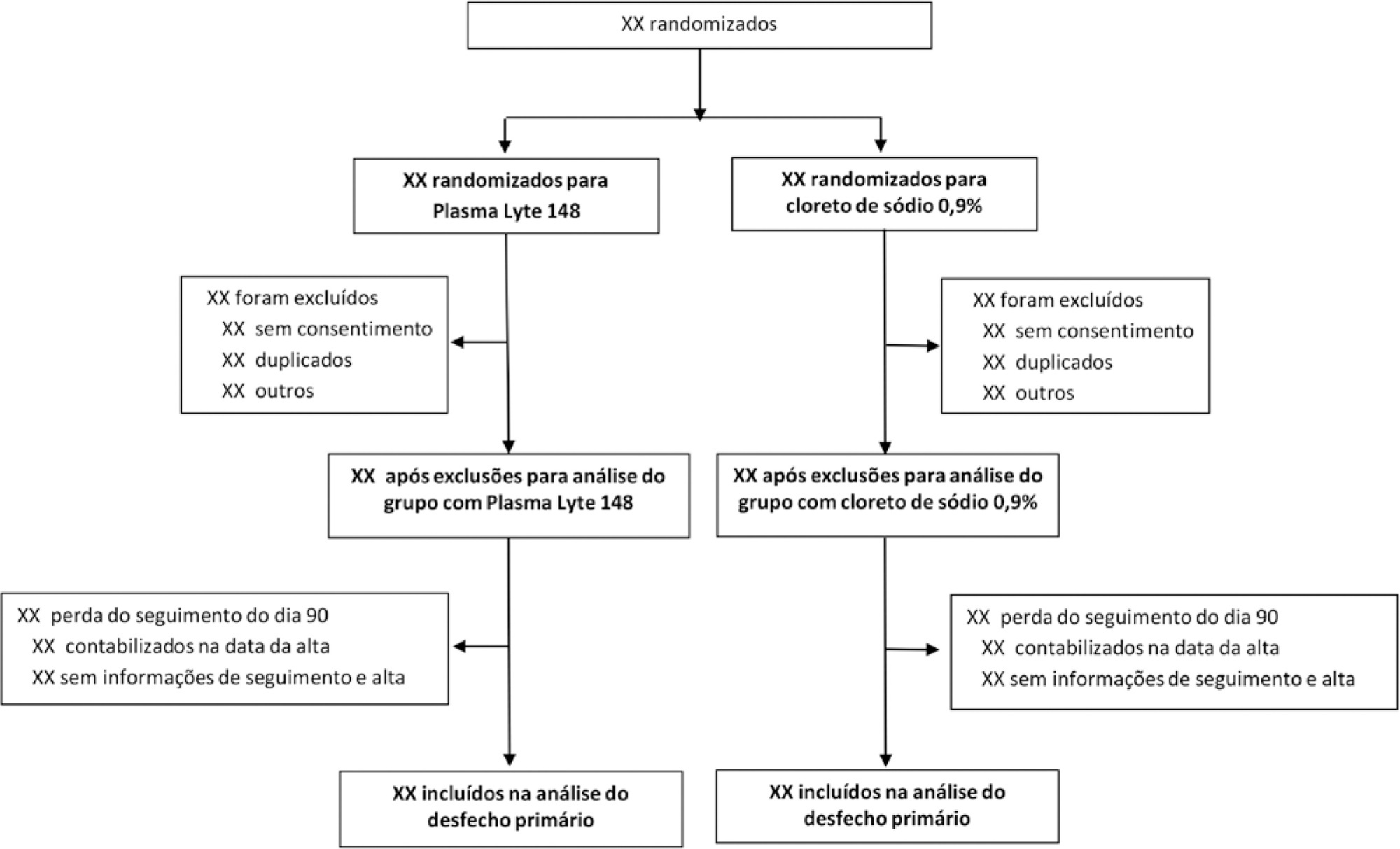
Objective:
To report the statistical analysis plan (first version) for the Balanced Solutions versus Saline in Intensive Care Study (BaSICS).
Methods:
BaSICS is a multicenter factorial randomized controlled trial that will assess the effects of Plasma-Lyte 148 versus 0.9% saline as the fluid of choice in critically ill patients, as well as the effects of a slow (333mL/h) versus rapid (999mL/h) infusion speed during fluid challenges, on important patient outcomes. The fluid type will be blinded for investigators, patients and the analyses. No blinding will be possible for the infusion speed for the investigators, but all analyses will be kept blinded during the analysis procedure.
Results:
BaSICS will have 90-day mortality as its primary endpoint, which will be tested using mixed-effects Cox proportional hazard models, considering sites as a random variable (frailty models) adjusted for age, organ dysfunction and admission type. Important secondary endpoints include renal replacement therapy up to 90 days, acute renal failure, organ dysfunction at days 3 and 7, and mechanical ventilation-free days within 28 days.
Conclusion:
This manuscript provides details on the first version of the statistical analysis plan for the BaSICS trial and will guide the study’s analysis when follow-up is finished.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis