You searched for:"Alexandre Biasi Cavalcanti"
We found (31) results for your search.-
Review Article
Challenges for the development of alternative low-cost ventilators during COVID-19 pandemic in Brazil
Rev Bras Ter Intensiva. 2020;32(3):444-457
Abstract
Review ArticleChallenges for the development of alternative low-cost ventilators during COVID-19 pandemic in Brazil
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
Views0ABSTRACT
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária – ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Keywords:Biomedical engineeringBrazilCOVID-19Positive pressure respirationRespiration, artificialSevere acute respiratory syndromeVentilators, mechanicalSee more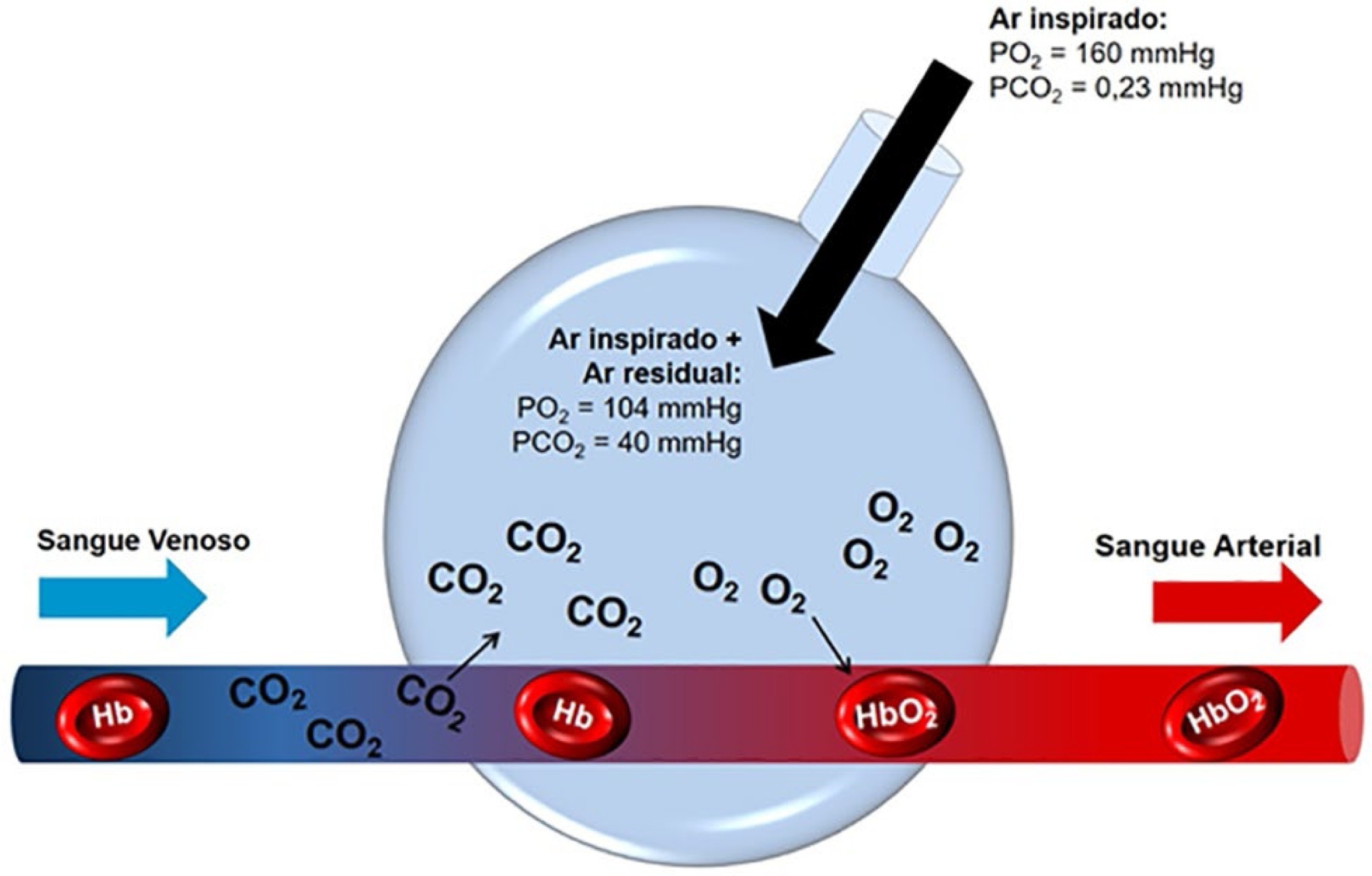
-
Original Article
COVID-19-associated ARDS treated with DEXamethasone (CoDEX): study design and rationale for a randomized trial
Rev Bras Ter Intensiva. 2020;32(3):354-362
Abstract
Original ArticleCOVID-19-associated ARDS treated with DEXamethasone (CoDEX): study design and rationale for a randomized trial
Rev Bras Ter Intensiva. 2020;32(3):354-362
DOI 10.5935/0103-507X.20200063
Views0Abstract
Objective:
The infection caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spreads worldwide and is considered a pandemic. The most common manifestation of SARS-CoV-2 infection (coronavirus disease 2019 – COVID-19) is viral pneumonia with varying degrees of respiratory compromise and up to 40% of hospitalized patients might develop acute respiratory distress syndrome. Several clinical trials evaluated the role of corticosteroids in non-COVID-19 acute respiratory distress syndrome with conflicting results. We designed a trial to evaluate the effectiveness of early intravenous dexamethasone administration on the number of days alive and free of mechanical ventilation within 28 days after randomization in adult patients with moderate or severe acute respiratory distress syndrome due to confirmed or probable COVID-19.
Methods:
This is a pragmatic, prospective, randomized, stratified, multicenter, open-label, controlled trial including 350 patients with early-onset (less than 48 hours before randomization) moderate or severe acute respiratory distress syndrome, defined by the Berlin criteria, due to COVID-19. Eligible patients will be randomly allocated to either standard treatment plus dexamethasone (Intervention Group) or standard treatment without dexamethasone (Control Group). Patients in the intervention group will receive dexamethasone 20mg intravenous once daily for 5 days, followed by dexamethasone 10mg IV once daily for additional 5 days or until intensive care unit discharge, whichever occurs first. The primary outcome is ventilator-free days within 28 days after randomization, defined as days alive and free from invasive mechanical ventilation. Secondary outcomes are all-cause mortality rates at day 28, evaluation of the clinical status at day 15 assessed with a 6-level ordinal scale, mechanical ventilation duration from randomization to day 28, Sequential Organ Failure Assessment Score evaluation at 48 hours, 72 hours and 7 days and intensive care unit -free days within 28.
Keywords:Adrenal cortex hormonesCoronavirusCOVID-19Critical careDexamethasoneRespiratory distress syndrome, adultSee more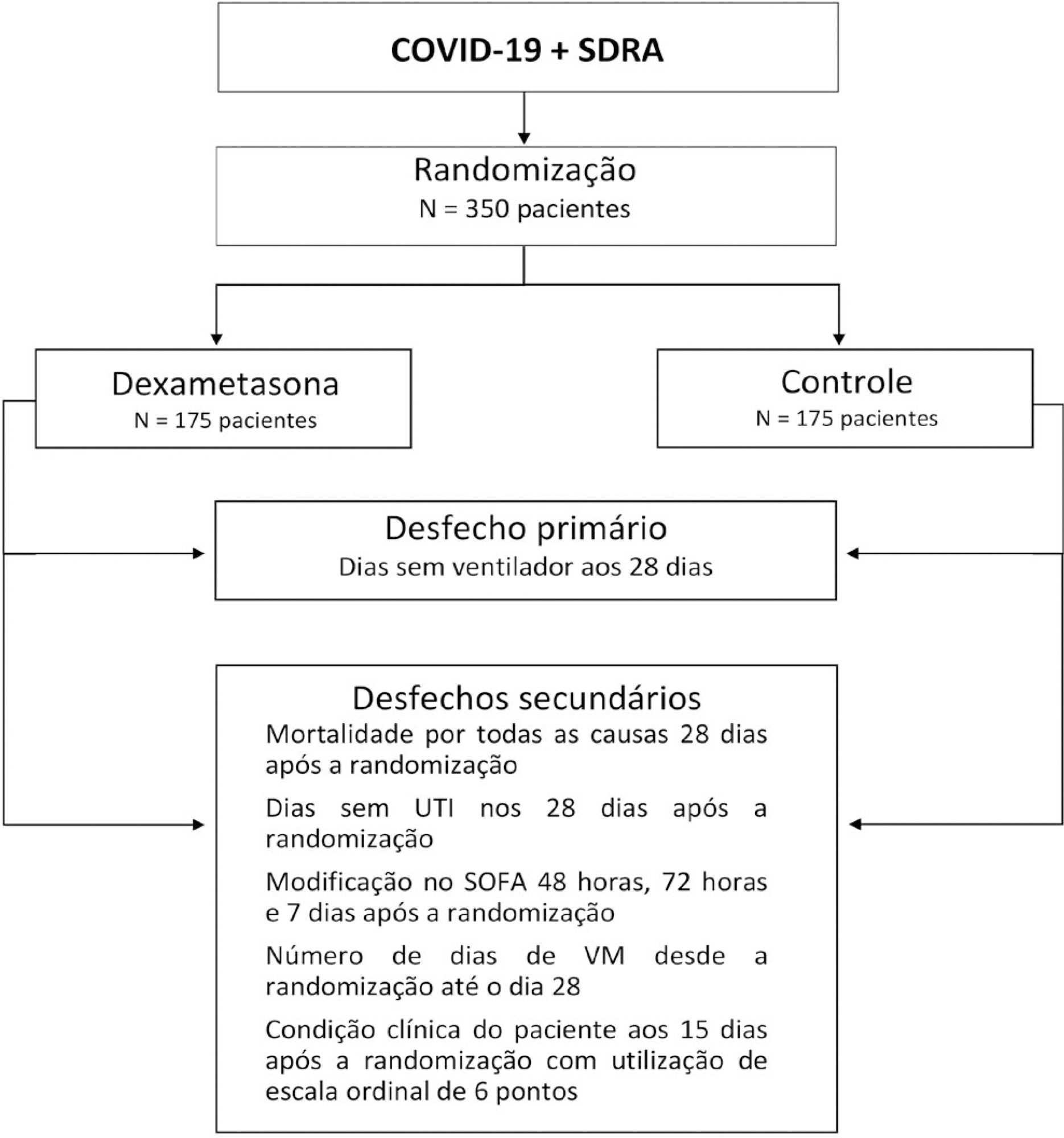
-
Original Article
Rationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
Abstract
Original ArticleRationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Views0See moreABSTRACT
Introduction:
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
Methods and analysis:
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
Ethics and dissemination:
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

-
Original Article
Availability of resources to treat sepsis in Brazil: a random sample of Brazilian institutions
Rev Bras Ter Intensiva. 2019;31(2):193-201
Abstract
Original ArticleAvailability of resources to treat sepsis in Brazil: a random sample of Brazilian institutions
Rev Bras Ter Intensiva. 2019;31(2):193-201
DOI 10.5935/0103-507X.20190033
Views1ABSTRACT
Objective:
To characterize resource availability from a nationally representative random sample of intensive care units in Brazil.
Methods:
A structured online survey of participating units in the Sepsis PREvalence Assessment Database (SPREAD) study, a nationwide 1-day point prevalence survey to assess the burden of sepsis in Brazil, was sent to the medical director of each unit.
Results:
A representative sample of 277 of the 317 invited units responded to the resources survey. Most of the hospitals had fewer than 500 beds (94.6%) with a median of 14 beds in the intensive care unit. Providing care for public-insured patients was the main source of income in two-thirds of the surveyed units. Own microbiology laboratory was not available for 26.8% of the surveyed intensive care units, and 10.5% did not always have access to blood cultures. Broad spectrum antibiotics were not always available in 10.5% of surveyed units, and 21.3% could not always measure lactate within three hours. Those institutions with a high resource availability (158 units, 57%) were usually larger and preferentially served patients from the private health system compared to institutions without high resource availability. Otherwise, those without high resource availability did not always have broad-spectrum antibiotics (24.4%), vasopressors (4.2%) or crystalloids (7.6%).
Conclusion:
Our study indicates that a relevant number of units cannot perform basic monitoring and therapeutic interventions in septic patients. Our results highlight major opportunities for improvement to adhere to simple but effective interventions in Brazil.
Keywords:Brazil/epidemiologyCritical careDeveloping countriesEpidemiological monitoringHealth resourcesIntensive care unitsSepsis/epidemiologyTherapeuticsSee more -
Original Article
Quality of life after intensive care unit: a multicenter cohort study protocol for assessment of long-term outcomes among intensive care survivors in Brazil
Rev Bras Ter Intensiva. 2018;30(4):405-413
Abstract
Original ArticleQuality of life after intensive care unit: a multicenter cohort study protocol for assessment of long-term outcomes among intensive care survivors in Brazil
Rev Bras Ter Intensiva. 2018;30(4):405-413
DOI 10.5935/0103-507X.20180063
Views0ABSTRACT
Objective:
To establish the prevalence of physical, cognitive and psychiatric disabilities, associated factors and their relationship with the qualities of life of intensive care survivors in Brazil.
Methods:
A prospective multicenter cohort study is currently being conducted at 10 adult medical-surgical intensive care units representative of the 5 Brazilian geopolitical regions. Patients aged ≥ 18 years who are discharged from the participating intensive care units and stay 72 hours or more in the intensive care unit for medical or emergency surgery admissions or 120 hours or more for elective surgery admissions are consecutively included. Patients are followed up for a period of one year by means of structured telephone interviews conducted at 3, 6 and 12 months after discharge from the intensive care unit. The outcomes are functional dependence, cognitive dysfunction, anxiety and depression symptoms, posttraumatic stress symptoms, health-related quality of life, rehospitalization and long-term mortality.
Discussion:
The present study has the potential to contribute to current knowledge of the prevalence and factors associated with postintensive care syndrome among adult intensive care survivors in Brazil. In addition, an association might be established between postintensive care syndrome and health-related quality of life.
Keywords:AnxietyCognitive dysfunctionCritical care outcomesDepressionDisabled personsQuality of lifeStress disorders, PosttraumaticSee more
-
Commentary
Sepsis is an important healthcare burden in Latin America: a call to action!
Rev Bras Ter Intensiva. 2018;30(4):402-404
Abstract
CommentarySepsis is an important healthcare burden in Latin America: a call to action!
Rev Bras Ter Intensiva. 2018;30(4):402-404
DOI 10.5935/0103-507X.20180061
Views0Sepsis is a life-threatening organ dysfunction caused by a dysregulated host response to infection. The precise incidence of sepsis is unknown and there is a lack of populational studies on the disease, especially from low and middle-income countries. However, extrapolations of populational data from high-income countries suggest a number of 30 million sepsis cases annually […]See more -
Original Article
Statistical analysis plan for early goal-directed therapy using a physiological holistic view – the ANDROMEDA-SHOCK: a randomized controlled trial
Rev Bras Ter Intensiva. 2018;30(3):253-263
Abstract
Original ArticleStatistical analysis plan for early goal-directed therapy using a physiological holistic view – the ANDROMEDA-SHOCK: a randomized controlled trial
Rev Bras Ter Intensiva. 2018;30(3):253-263
DOI 10.5935/0103-507X.20180041
Views0See moreABSTRACT
Background:
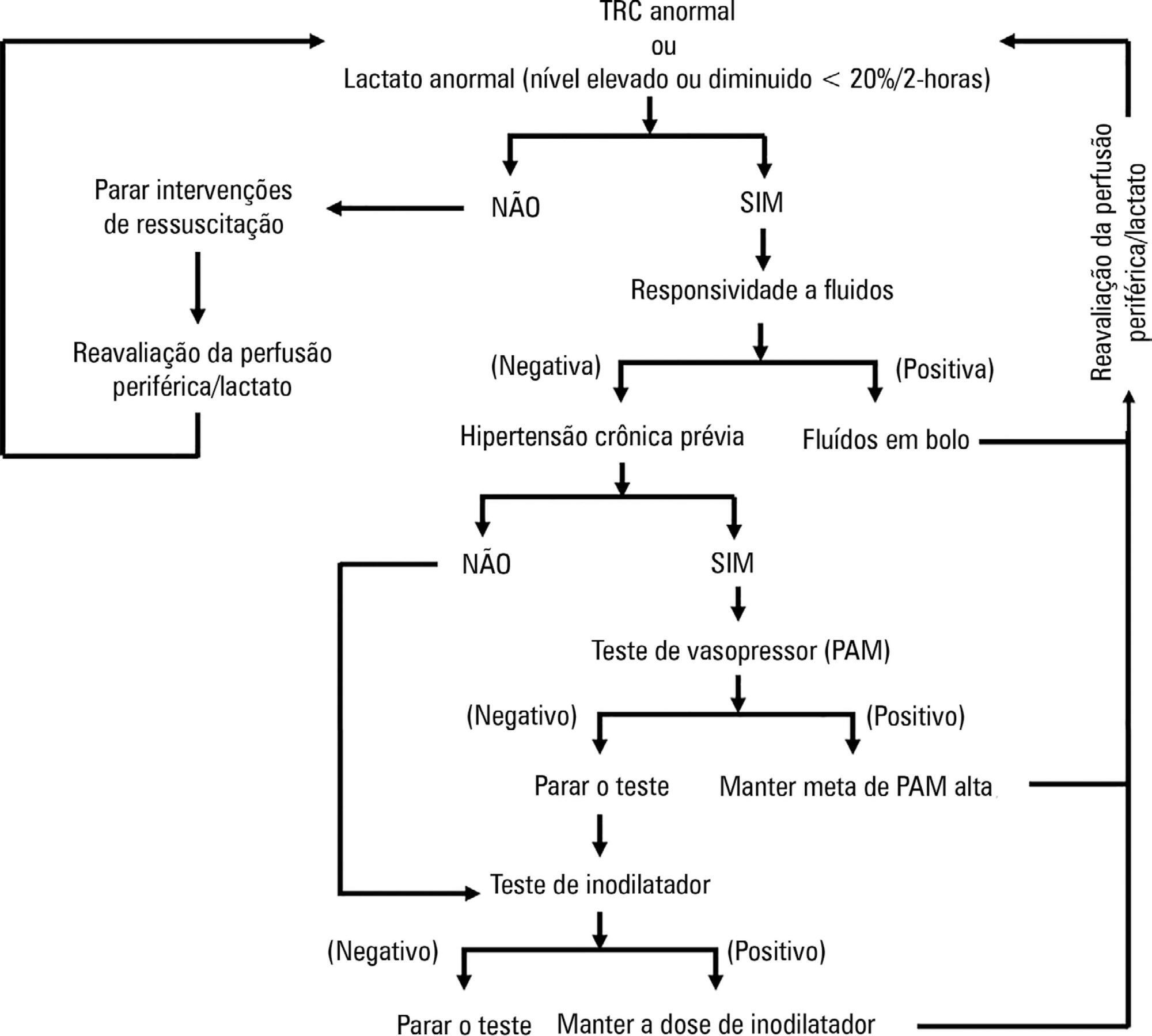
ANDROMEDA-SHOCK is an international, multicenter, randomized controlled trial comparing peripheral perfusion-targeted resuscitation to lactate-targeted resuscitation in patients with septic shock in order to test the hypothesis that resuscitation targeting peripheral perfusion will be associated with lower morbidity and mortality.
Objective:
To report the statistical analysis plan for the ANDROMEDA-SHOCK trial.
Methods:
We describe the trial design, primary and secondary objectives, patients, methods of randomization, interventions, outcomes, and sample size. We describe our planned statistical analysis for the primary, secondary and tertiary outcomes. We also describe the subgroup and sensitivity analyses. Finally, we provide details for presenting our results, including mock tables showing baseline characteristics, the evolution of hemodynamic and perfusion variables, and the effects of treatments on outcomes.
Conclusion:
According to the best trial practice, we report our statistical analysis plan and data management plan prior to locking the database and initiating the analyses. We anticipate that this procedure will prevent analysis bias and enhance the utility of the reported results.
-
Original Article
Existing capacity for renal replacement therapy and site-specific practices for managing acute kidney injury at centers participating in the BaSICS trial
Rev Bras Ter Intensiva. 2018;30(3):264-285
Abstract
Original ArticleExisting capacity for renal replacement therapy and site-specific practices for managing acute kidney injury at centers participating in the BaSICS trial
Rev Bras Ter Intensiva. 2018;30(3):264-285
DOI 10.5935/0103-507X.20180058
Views0See moreABSTRACT
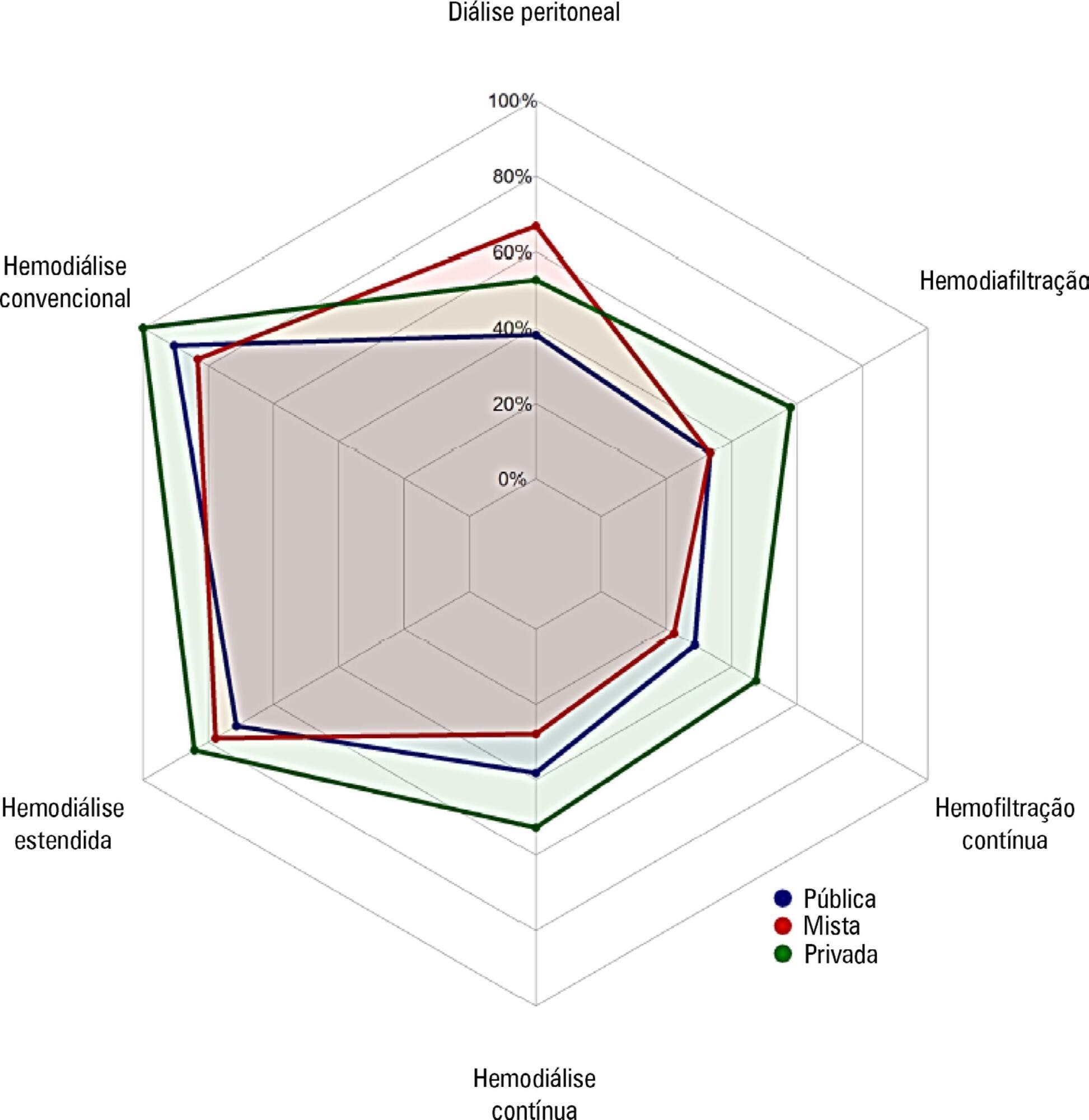
Objective:
To investigate the existing capacity for renal replacement therapy and site-specific practices for managing acute kidney injury at centers participating in the BaSICS trial.
Methods:
A questionnaire was provided to the chairs of 61 intensive care units enrolled in a randomized clinical trial in Brazil. A total of 124 physicians completed the questionnaire.
Results:
Approximately 15% of the patients admitted to the analyzed intensive care units received renal replacement therapy at the time of data collection. At least one renal replacement method was available in all of the analyzed units. Continuous methods were available more frequently at the private units than at the public units. The time from indication to onset of treatment was longer at the public units than at private units. The main obstacles to treatment initiation at public intensive care units were related to the availability of equipment and personnel, while the main bottleneck at private units was the nephrologist assessment. A considerable proportion of the participants stated that they would change their approach to renal replacement therapy if there were no limitations on the availability of methods in their units.
Conclusion:
There was wide variation in the availability of resources for renal replacement therapy and in the management of acute kidney injury in Brazilian intensive care units. This information should be taken into account when planning clinical trials focused on this topic in Brazil.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




