Abstract
Critical Care Science. 2024;36:e20240208en
DOI 10.62675/2965-2774.20240208-en
To evaluate the association between driving pressure and tidal volume based on predicted body weight and mortality in a cohort of patients with acute respiratory distress syndrome caused by COVID-19.
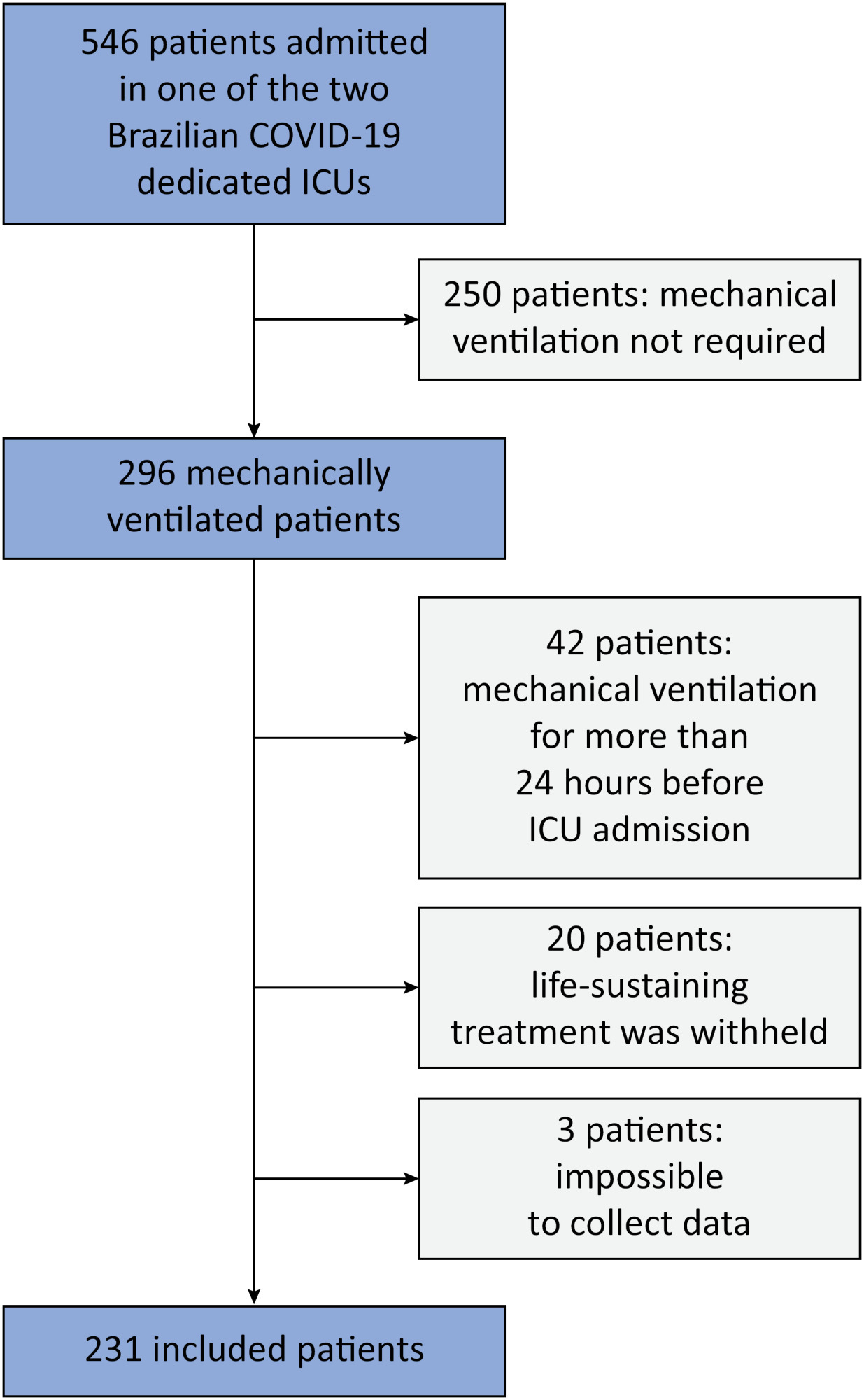
This was a prospective, observational study that included patients with acute respiratory distress syndrome due to COVID-19 admitted to two intensive care units. We performed multivariable analyses to determine whether driving pressure and tidal volume/kg predicted body weight on the first day of mechanical ventilation, as independent variables, are associated with hospital mortality.
We included 231 patients. The mean age was 64 (53 - 74) years, and the mean Simplified Acute and Physiology Score 3 score was 45 (39 - 54). The hospital mortality rate was 51.9%. Driving pressure was independently associated with hospital mortality (odds ratio 1.21, 95%CI 1.04 - 1.41 for each cm H2O increase in driving pressure, p = 0.01). Based on a double stratification analysis, we found that for the same level of tidal volume/kg predicted body weight, the risk of hospital death increased with increasing driving pressure. However, changes in tidal volume/kg predicted body weight were not associated with mortality when they did not lead to an increase in driving pressure.
In patients with acute respiratory distress syndrome caused by COVID-19, exposure to higher driving pressure, as opposed to higher tidal volume/kg predicted body weight, is associated with greater mortality. These results suggest that driving pressure might be a primary target for lung-protective mechanical ventilation in these patients.
Abstract
Critical Care Science. 2024;36:e20240088en
DOI 10.62675/2965-2774.20240088-en
Abstract
Critical Care Science. 2024;36:e20240196en
DOI 10.62675/2965-2774.20240196-en
To provide insights into the potential benefits of goal-directed therapy guided by FloTrac in reducing postoperative complications and improving outcomes.
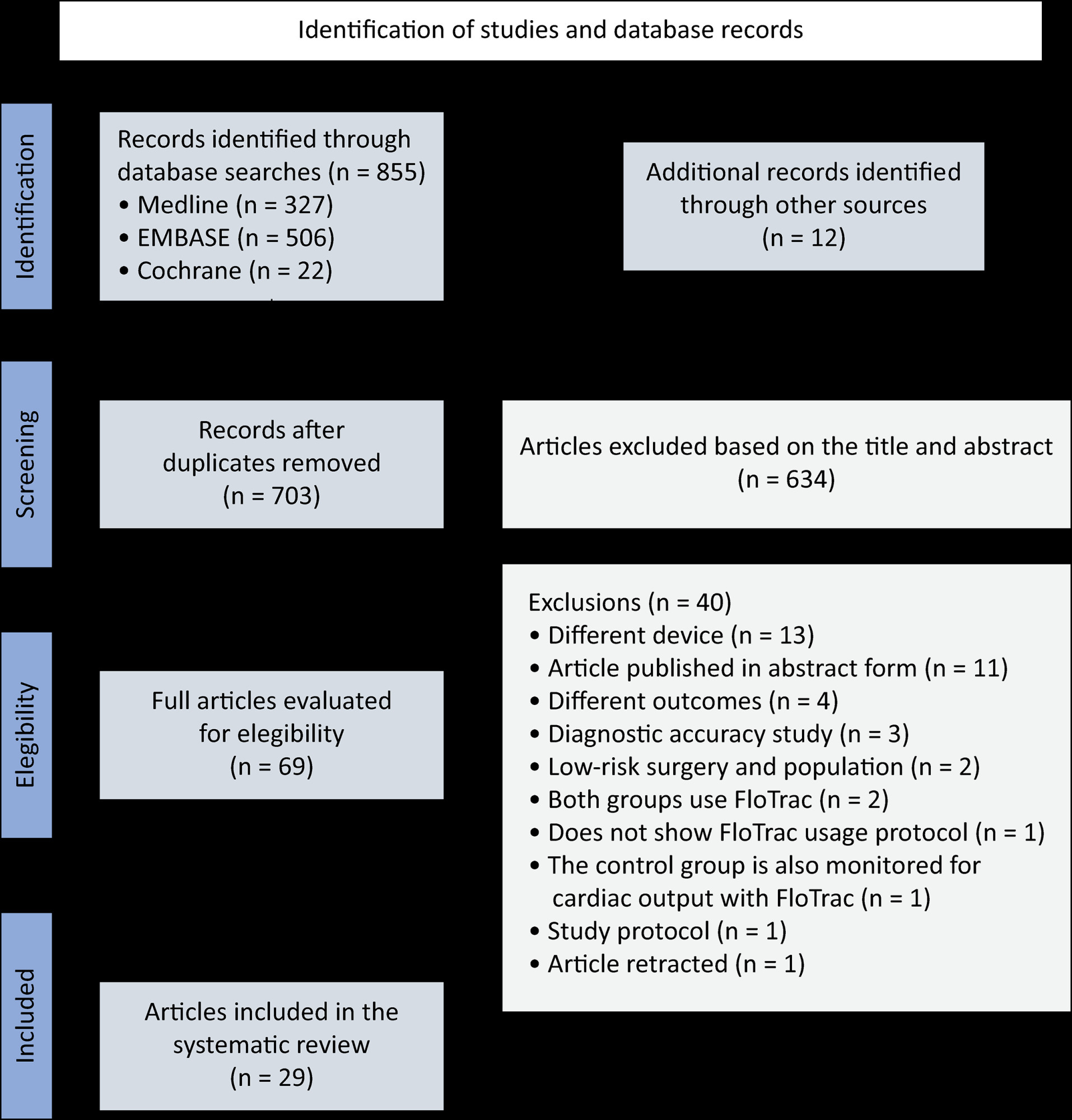
We performed a systematic review and meta-analysis of randomized controlled trials to evaluate goal-directed therapy guided by FloTrac in major surgery, comparing goal-directed therapy with usual care or invasive monitoring in cardiac and noncardiac surgery subgroups. The quality of the articles and evidence were evaluated with a risk of bias tool and GRADE.
We included 29 randomized controlled trials with 3,468 patients. Goal-directed therapy significantly reduced the duration of hospital stay (mean difference -1.43 days; 95%CI 2.07 to -0.79; I2 81%), intensive care unit stay (mean difference -0.77 days; 95%CI -1.18 to -0.36; I2 93%), and mechanical ventilation (mean difference -2.48 hours, 95%CI -4.10 to -0.86, I2 63%). There was no statistically significant difference in mortality, myocardial infarction, acute kidney injury or hypotension, but goal-directed therapy significantly reduced the risk of heart failure or pulmonary edema (RR 0.46; 95%CI 0.23 - 0.92; I2 0%).
Goal-directed therapy guided by the FloTrac sensor improved clinical outcomes and shortened the length of stay in the hospital and intensive care unit in patients undergoing major surgery. Further research can validate these results using specific protocols and better understand the potential benefits of FloTrac beyond these outcomes.
Abstract
Critical Care Science. 2024;36:e20240210en
DOI 10.62675/2965-2774.20240210-en
Driving pressure has been suggested to be the main driver of ventilator-induced lung injury and mortality in observational studies of acute respiratory distress syndrome. Whether a driving pressure-limiting strategy can improve clinical outcomes is unclear.
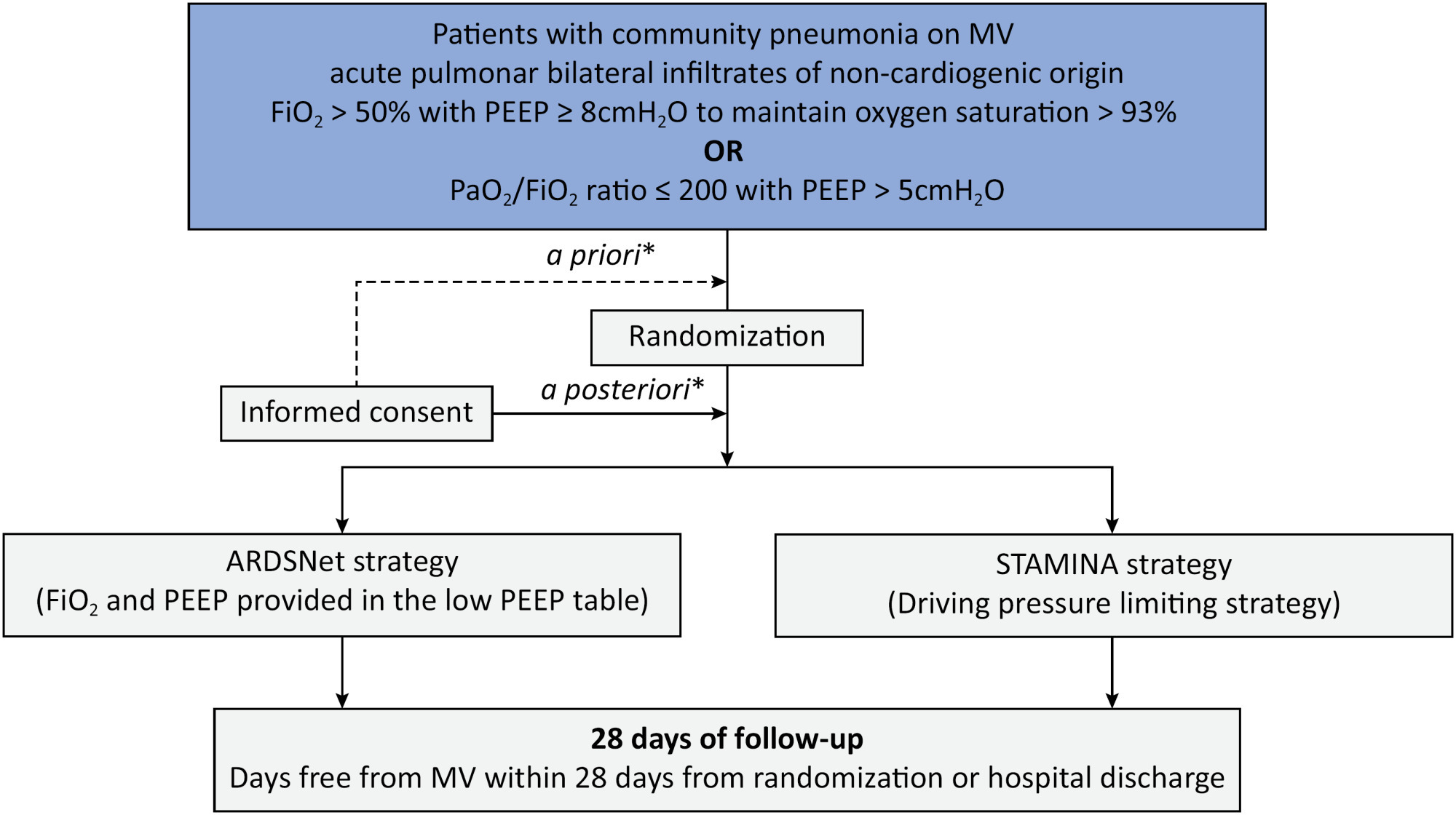
To describe the protocol and statistical analysis plan that will be used to test whether a driving pressure-limiting strategy including positive end-expiratory pressure titration according to the best respiratory compliance and reduction in tidal volume is superior to a standard strategy involving the use of the ARDSNet low-positive end-expiratory pressure table in terms of increasing the number of ventilator-free days in patients with acute respiratory distress syndrome due to community-acquired pneumonia.
The ventilator STrAtegy for coMmunIty acquired pNeumoniA (STAMINA) study is a randomized, multicenter, open-label trial that compares a driving pressure-limiting strategy to the ARDSnet low-positive end-expiratory pressure table in patients with moderate-to-severe acute respiratory distress syndrome due to community-acquired pneumonia admitted to intensive care units. We expect to recruit 500 patients from 20 Brazilian and 2 Colombian intensive care units. They will be randomized to a driving pressure-limiting strategy group or to a standard strategy using the ARDSNet low-positive end-expiratory pressure table. In the driving pressure-limiting strategy group, positive end-expiratory pressure will be titrated according to the best respiratory system compliance.
The primary outcome is the number of ventilator-free days within 28 days. The secondary outcomes are in-hospital and intensive care unit mortality and the need for rescue therapies such as extracorporeal life support, recruitment maneuvers and inhaled nitric oxide.
STAMINA is designed to provide evidence on whether a driving pressure-limiting strategy is superior to the ARDSNet low-positive end-expiratory pressure table strategy for increasing the number of ventilator-free days within 28 days in patients with moderate-to-severe acute respiratory distress syndrome. Here, we describe the rationale, design and status of the trial.
Abstract
Critical Care Science. 2024;36:e20240144en
DOI 10.62675/2965-2774.20240144-en
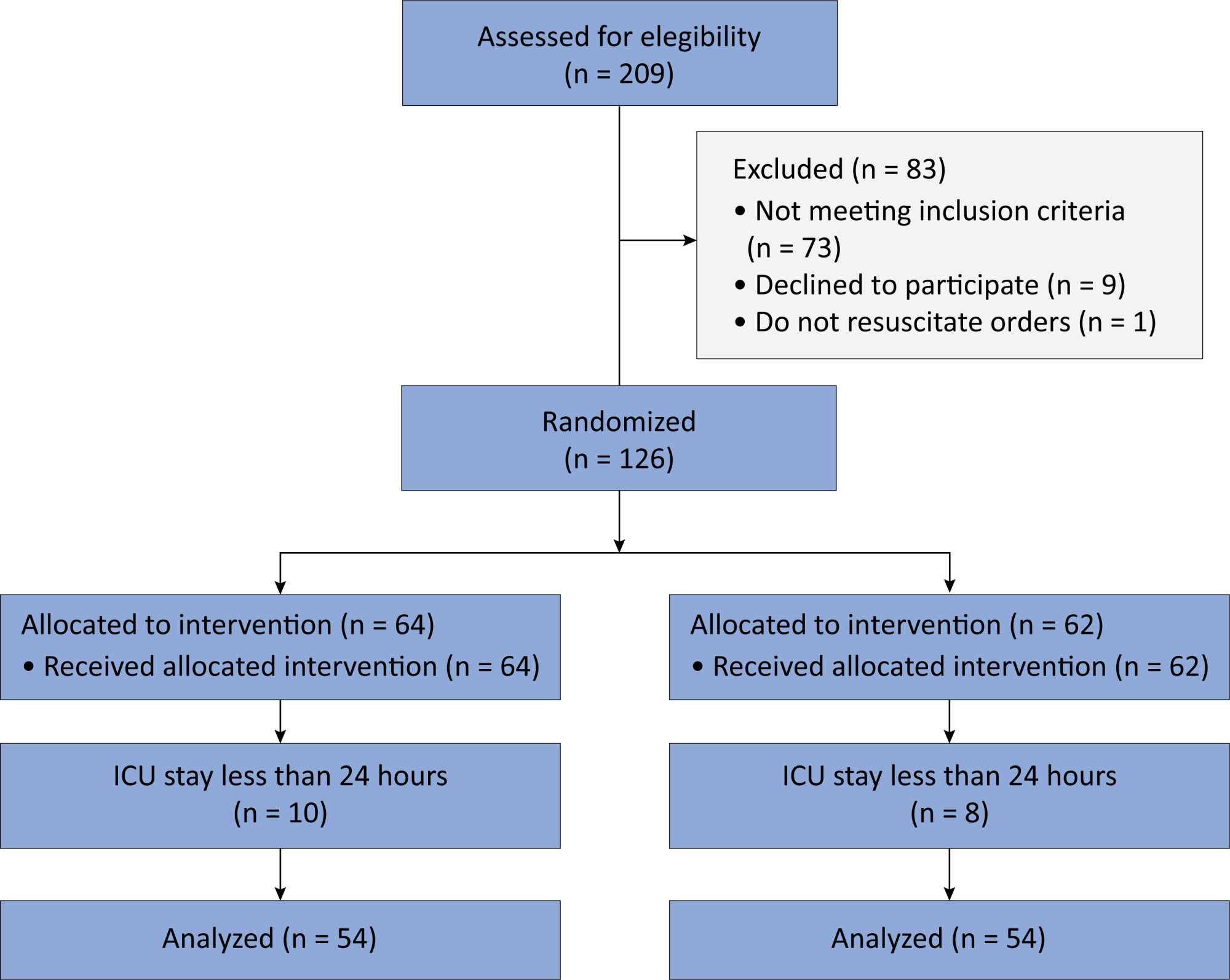
To determine whether enteral melatonin decreases the incidence of delirium in critically ill adults.
In this randomized controlled trial, adults were admitted to the intensive care unit and received either usual standard care alone (Control Group) or in combination with 3mg of enteral melatonin once a day at 9 PM (Melatonin Group). Concealment of allocation was done by serially numbered opaque sealed envelopes. The intensivist assessing delirium and the investigator performing the data analysis were blinded to the group allocation. The primary outcome was the incidence of delirium within 24 hours of the intensive care unit stay. The secondary outcomes were the incidence of delirium on Days 3 and 7, intensive care unit mortality, length of intensive care unit stay, duration of mechanical ventilation and Glasgow outcome score (at discharge).
We included 108 patients in the final analysis, with 54 patients in each group. At 24 hours of intensive care unit stay, there was no difference in the incidence of delirium between Melatonin and Control Groups (29.6 versus 46.2%; RR = 0.6; 95%CI 0.38 - 1.05; p = 0.11). No secondary outcome showed a statistically significant difference.
Enteral melatonin 3mg is not more effective at decreasing the incidence of delirium than standard care is in critically ill adults.
Abstract
Revista Brasileira de Terapia Intensiva. 2016;28(1):08-10
DOI 10.5935/0103-507X.20160008
Abstract
Revista Brasileira de Terapia Intensiva. 2013;25(1):1-2
Abstract
Revista brasileira de terapia intensiva. 2014;26(1):1-6
DOI 10.5935/0103-507X.20140001
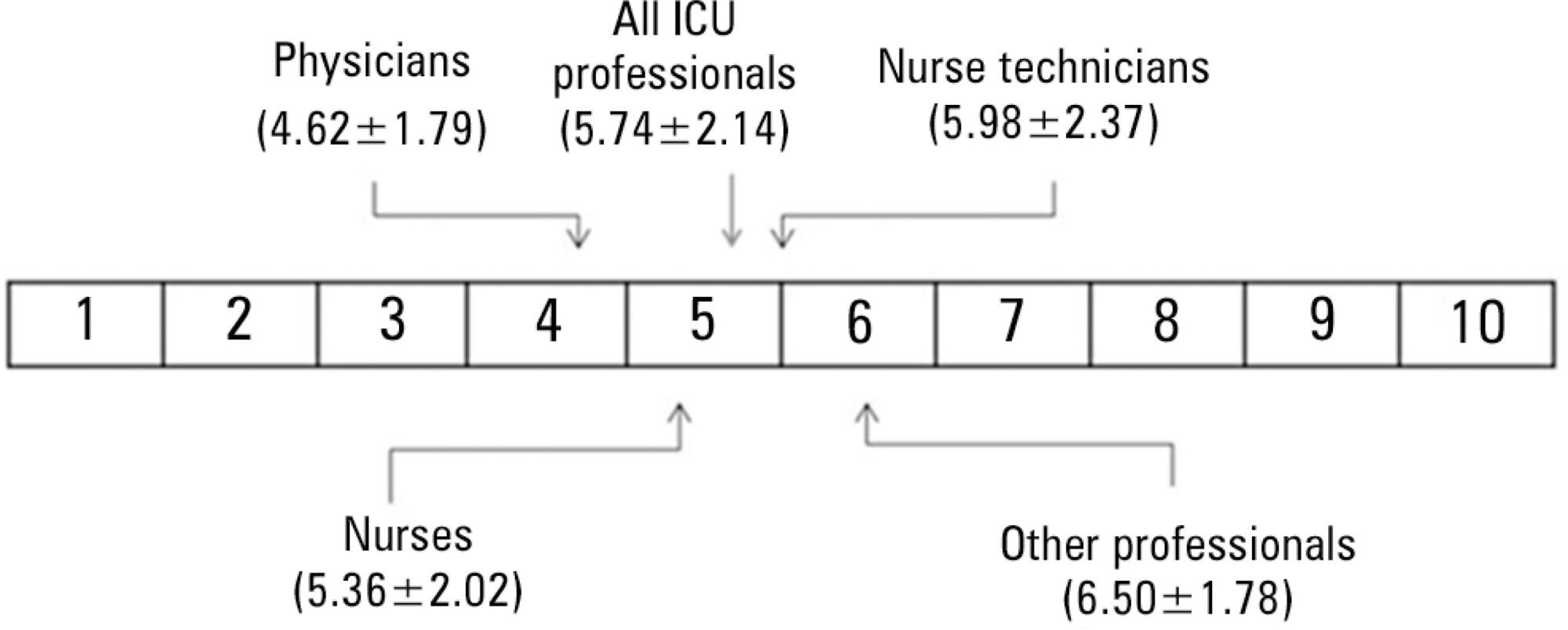
To evaluate the satisfaction of the intensive care unit staff with a computerized physician order entry and to compare the concept of the computerized physician order entry relevance among intensive care unit healthcare workers.
We performed a cross-sectional survey to assess the satisfaction of the intensive care unit staff with the computerized physician order entry in a 30-bed medical/surgical adult intensive care unit using a self-administered questionnaire. The questions used for grading satisfaction levels were answered according to a numerical scale that ranged from 1 point (low satisfaction) to 10 points (high satisfaction).
The majority of the respondents (n=250) were female (66%) between the ages of 30 and 35 years of age (69%). The overall satisfaction with the computerized physician order entry scored 5.74±2.14 points. The satisfaction was lower among physicians (n=42) than among nurses, nurse technicians, respiratory therapists, clinical pharmacists and diet specialists (4.62±1.79 versus 5.97±2.14, p<0.001); satisfaction decreased with age (p<0.001). Physicians scored lower concerning the potential of the computerized physician order entry for improving patient safety (5.45±2.20 versus 8.09±2.21, p<0.001) and the ease of using the computerized physician order entry (3.83±1.88 versus 6.44±2.31, p<0.001). The characteristics independently associated with satisfaction were the system's user-friendliness, accuracy, capacity to provide clear information, and fast response time.
Six months after its implementation, healthcare workers were satisfied, albeit not entirely, with the computerized physician order entry. The overall users' satisfaction with computerized physician order entry was lower among physicians compared to other healthcare professionals. The factors associated with satisfaction included the belief that digitalization decreased the workload and contributed to the intensive care unit quality with a user-friendly and accurate system and that digitalization provided concise information within a reasonable time frame.