Abstract
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
Abstract
Rev Bras Ter Intensiva. 2021;33(1):38-47
DOI 10.5935/0103-507X.20210004
Triage for intensive care unit admission is a frequent event and is associated to worse clinical outcomes. The process of triage is variable and may be influenced by biases and prejudices, which could lead to potentially unfair decisions. The Brazilian Federal Council of Medicine (Conselho Federal de Medicina) has recently released a guideline for intensive care unit admission and discharge. The aim of this paper is to evaluate the ethical dilemmas related to the implementation of this guideline, through the accountability for reasonabless approach, known as A4R, as elaborated by Norman Daniels. We conclude that the guideline contemplates A4R conditions, but we observe that there is a need for indication of A4R-concordant criteria to operationalize the guidelines.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):166-196
DOI 10.5935/0103-507X.20200039
Different therapies are currently used, considered, or proposed for the treatment of COVID-19; for many of those therapies, no appropriate assessment of effectiveness and safety was performed. This document aims to provide scientifically available evidence-based information in a transparent interpretation, to subsidize decisions related to the pharmacological therapy of COVID-19 in Brazil.
A group of 27 experts and methodologists integrated a task-force formed by professionals from the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB), the Brazilian Society of Infectious Diseases (Sociedad Brasileira de Infectologia - SBI) and the Brazilian Society of Pulmonology and Tisiology (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT). Rapid systematic reviews, updated on April 28, 2020, were conducted. The assessment of the quality of evidence and the development of recommendations followed the GRADE system. The recommendations were written on May 5, 8, and 13, 2020.
Eleven recommendations were issued based on low or very-low level evidence. We do not recommend the routine use of hydroxychloroquine, chloroquine, azithromycin, lopinavir/ritonavir, corticosteroids, or tocilizumab for the treatment of COVID-19. Prophylactic heparin should be used in hospitalized patients, however, no anticoagulation should be provided for patients without a specific clinical indication. Antibiotics and oseltamivir should only be considered for patients with suspected bacterial or influenza coinfection, respectively.
So far no pharmacological intervention was proven effective and safe to warrant its use in the routine treatment of COVID-19 patients; therefore such patients should ideally be treated in the context of clinical trials. The recommendations herein provided will be revised continuously aiming to capture newly generated evidence.
Abstract
Rev Bras Ter Intensiva. 2020;32(1):2-10
DOI 10.5935/0103-507X.20200002
Current COVID-19 epidemics was declared on December 31, 2019 at the Wuhan city seafood market, rapidly spreading throughout China, and later reaching several countries (mainly South Korea, Japan, Italy and Iran) and, since March 1, reaching Portugal. Most of the infected patients present with mild symptoms, not requiring hospitalization. Among those admitted to the hospital, 6% to 10% require admission to the intensive care unit. These recommendations are aimed to support the organization of intensive care services to respond COVID-19, providing optimized care to the patient and protection for healthcare professionals.
Abstract
Rev Bras Ter Intensiva. 2019;31(4):434-443
DOI 10.5935/0103-507X.20190084
Immobility can cause several complications, including skeletal muscle atrophy and weakness, that influence the recovery of critically ill patients. This effect can be mitigated by early mobilization. Six key questions guided this research: Is early mobilization safe? Which patients are candidates for early mobilization? What are the contraindications? What is the appropriate dose, and how should it be defined? What results are obtained? What are the prognostic indicators for the use of early mobilization? The objective of this guideline was to produce a document that would provide evidence-based recommendations and suggestions regarding the early mobilization of critically ill adult patients, with the aim of improving understanding of the topic and making a positive impact on patient care. This guideline was based on a systematic review of articles conducted using the PICO search strategy, as recommended by the Guidelines Project of the Associação Médica Brasileira. Randomized clinical trials, prognostic cohort studies, and systematic reviews with or without meta-analysis were selected, and the evidence was classified according to the Oxford Center for Evidence-based Medicine Levels of Evidence. For all the questions addressed, enough evidence was found to support safe and well-defined early mobilization, with prognostic indicators that support and recommend the technique. Early mobilization is associated with better functional outcomes and should be performed whenever indicated. Early mobilization is safe and should be the goal of the entire multidisciplinary team.
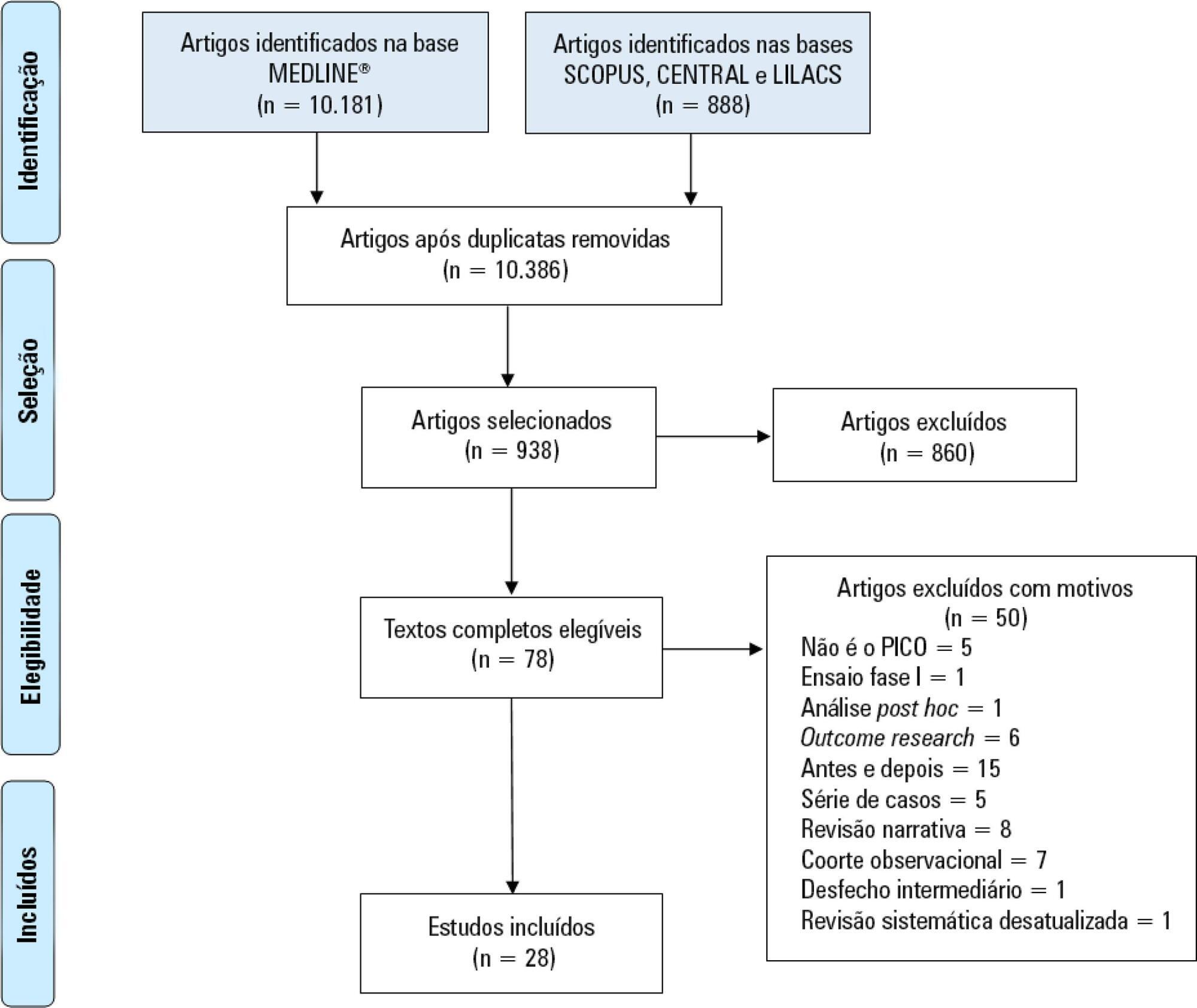
Abstract
Rev Bras Ter Intensiva. 2019;31(1):5-14
DOI 10.5935/0103-507X.20190002
Critically ill patients are at risk of developing stress ulcers in the upper digestive tract. Agents that suppress gastric acid are commonly prescribed to reduce the incidence of clinically important stress ulcer-related gastrointestinal bleeding. However, the indiscriminate use of stress ulcer prophylaxis in all patients admitted to the intensive care unit is not warranted and can have potential adverse clinical effects and cost implications. The present guidelines from the Sociedade Portuguesa de Cuidados Intensivos summarizes the current evidence and gives six clinical statements and an algorithm aiming to provide a standardized prescribing policy for the use of stress ulcer prophylaxis in the intensive care unit.
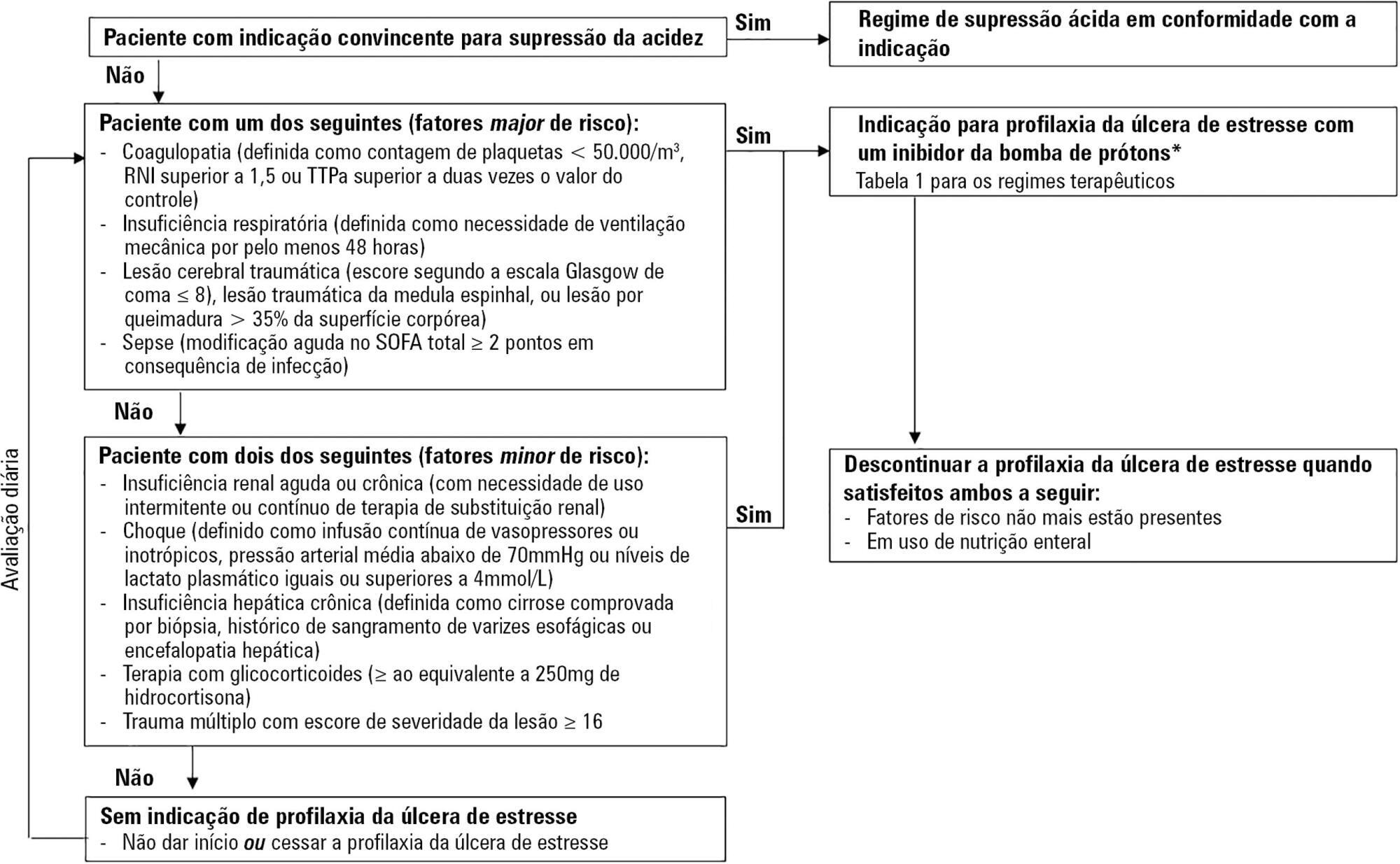
Abstract
Rev Bras Ter Intensiva. 2017;29(2):131-141
DOI 10.5935/0103-507X.20170023
To construct and implement an instrument (checklist) to improve safety when performing the prone maneuver.
This was an applied, qualitative and descriptive study. The instrument was developed based on a broad review of the literature pertaining to the construction of a care protocol using the main electronic databases (MEDLINE, LILACS and Cochrane).
We describe the construction of a patient safety tool with numerous modifications and adaptations based on the observations of the multidisciplinary team regarding its use in daily practice.
The use of the checklist when performing the prone maneuver increased the safety and reliability of the procedure. The team's understanding of the tool's importance to patient safety and training in its use are necessary for its success.
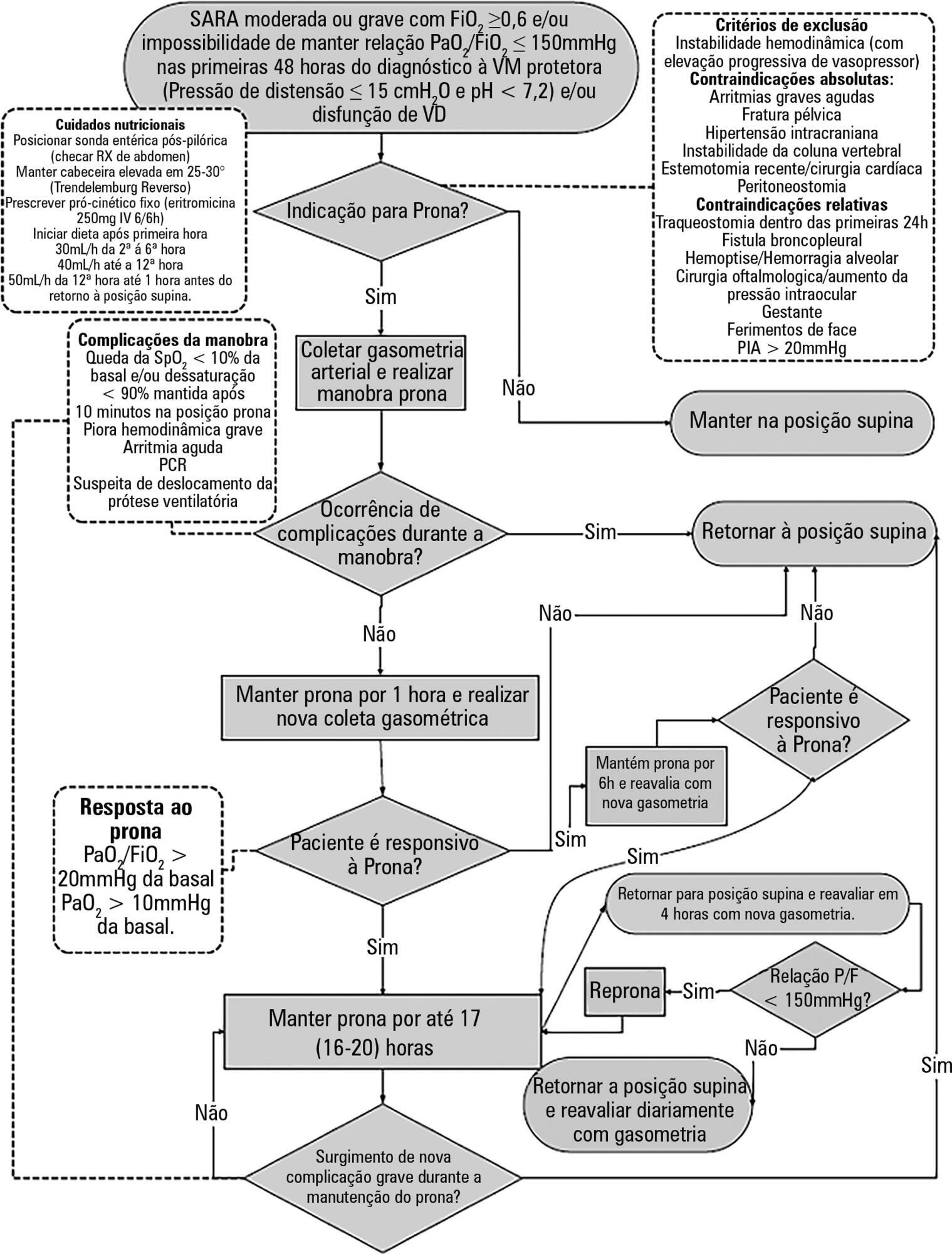
Abstract
Rev Bras Ter Intensiva. 2014;26(2):89-121
DOI 10.5935/0103-507X.20140017
Perspectives on invasive and noninvasive ventilatory support for critically ill patients are evolving, as much evidence indicates that ventilation may have positive effects on patient survival and the quality of the care provided in intensive care units in Brazil. For those reasons, the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB) and the Brazilian Thoracic Society (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT), represented by the Mechanical Ventilation Committee and the Commission of Intensive Therapy, respectively, decided to review the literature and draft recommendations for mechanical ventilation with the goal of creating a document for bedside guidance as to the best practices on mechanical ventilation available to their members. The document was based on the available evidence regarding 29 subtopics selected as the most relevant for the subject of interest. The project was developed in several stages, during which the selected topics were distributed among experts recommended by both societies with recent publications on the subject of interest and/or significant teaching and research activity in the field of mechanical ventilation in Brazil. The experts were divided into pairs that were charged with performing a thorough review of the international literature on each topic. All the experts met at the Forum on Mechanical Ventilation, which was held at the headquarters of AMIB in São Paulo on August 3 and 4, 2013, to collaboratively draft the final text corresponding to each sub-topic, which was presented to, appraised, discussed and approved in a plenary session that included all 58 participants and aimed to create the final document.