You searched for:"Flávia Ribeiro Machado"
We found (44) results for your search.Abstract
Rev Bras Ter Intensiva. 2022;34(4):410-417
DOI 10.5935/0103-507X.20220261-en
To describe the effects of balanced solution use on the short-term outcomes of patients with traumatic brain injury enrolled in BaSICS trial.
Patients were randomized to receive either 0.9% saline or balanced solution during their intensive care unit stay. The primary endpoint was 90-day mortality, and the secondary outcomes were days alive and free of intensive care unit stay at 28 days. The primary endpoint was assessed using Bayesian logistic regression. The secondary endpoint was assessed using a Bayesian zero-inflated beta binomial regression.
We included 483 patients (236 in the 0.9% saline arm and 247 in the balanced solution arm). A total of 338 patients (70%) with a Glasgow coma scale score ≤ 12 were enrolled. The overall probability that balanced solutions were associated with higher 90-day mortality was 0.98 (OR 1.48; 95%CrI 1.04 - 2.09); this mortality increment was particularly noticeable in patients with a Glasgow coma scale score below 6 at enrollment (probability of harm of 0.99). Balanced solutions were associated with -1.64 days alive and free of intensive care unit at 28 days (95%CrI -3.32 - 0.00) with a probability of harm of 0.97.
There was a high probability that balanced solutions were associated with high 90-day mortality and fewer days alive and free of intensive care units at 28 days.
Abstract
Rev Bras Ter Intensiva. 2016;28(4):413-419
DOI 10.5935/0103-507X.20160074
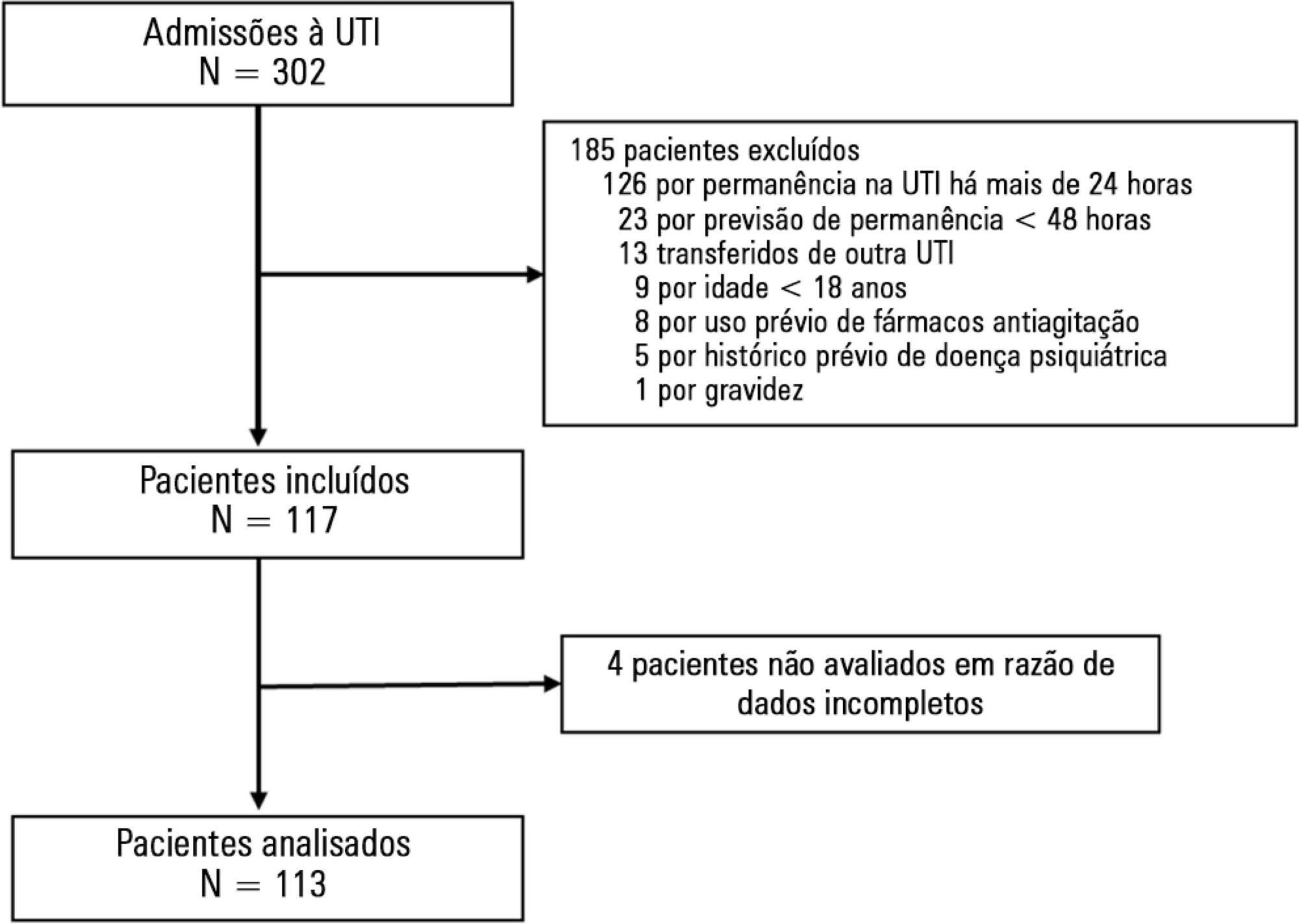
To evaluate the incidence of agitation in the first 7 days after intensive care unit admission, its risk factors and its associations with clinical outcomes.
This single-center prospective cohort study included all patients older than 18 years with a predicted stay > 48 hours within the first 24 hours of intensive care unit admission. Agitation was defined as a Richmond Agitation Sedation Scale score ≥ +2, an episode of agitation or the use of a specific medication recorded in patient charts.
Agitation occurred in 31.8% of the 113 patients. Multivariate analysis showed that delirium [OR = 24.14; CI95% 5.15 - 113.14; p < 0.001], moderate or severe pain [OR = 5.74; CI95% 1.73 - 19.10; p = 0.004], mechanical ventilation [OR = 10.14; CI95% 2.93 - 35.10; p < 0.001], and smoking habits [OR = 4.49; CI95% 1.33 - 15.17; p = 0.015] were independent factors for agitation, while hyperlactatemia was associated with a lower risk [OR = 0.169; CI95% 0.04 - 0.77; p = 0.021]. Agitated patients had fewer mechanical ventilation-free days at day 7 (p = 0.003).
The incidence of agitation in the first 7 days after admission to the intensive care unit was high. Delirium, moderate/severe pain, mechanical ventilation, and smoking habits were independent risk factors. Agitated patients had fewer ventilator-free days in the first 7 days.
Abstract
Rev Bras Ter Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
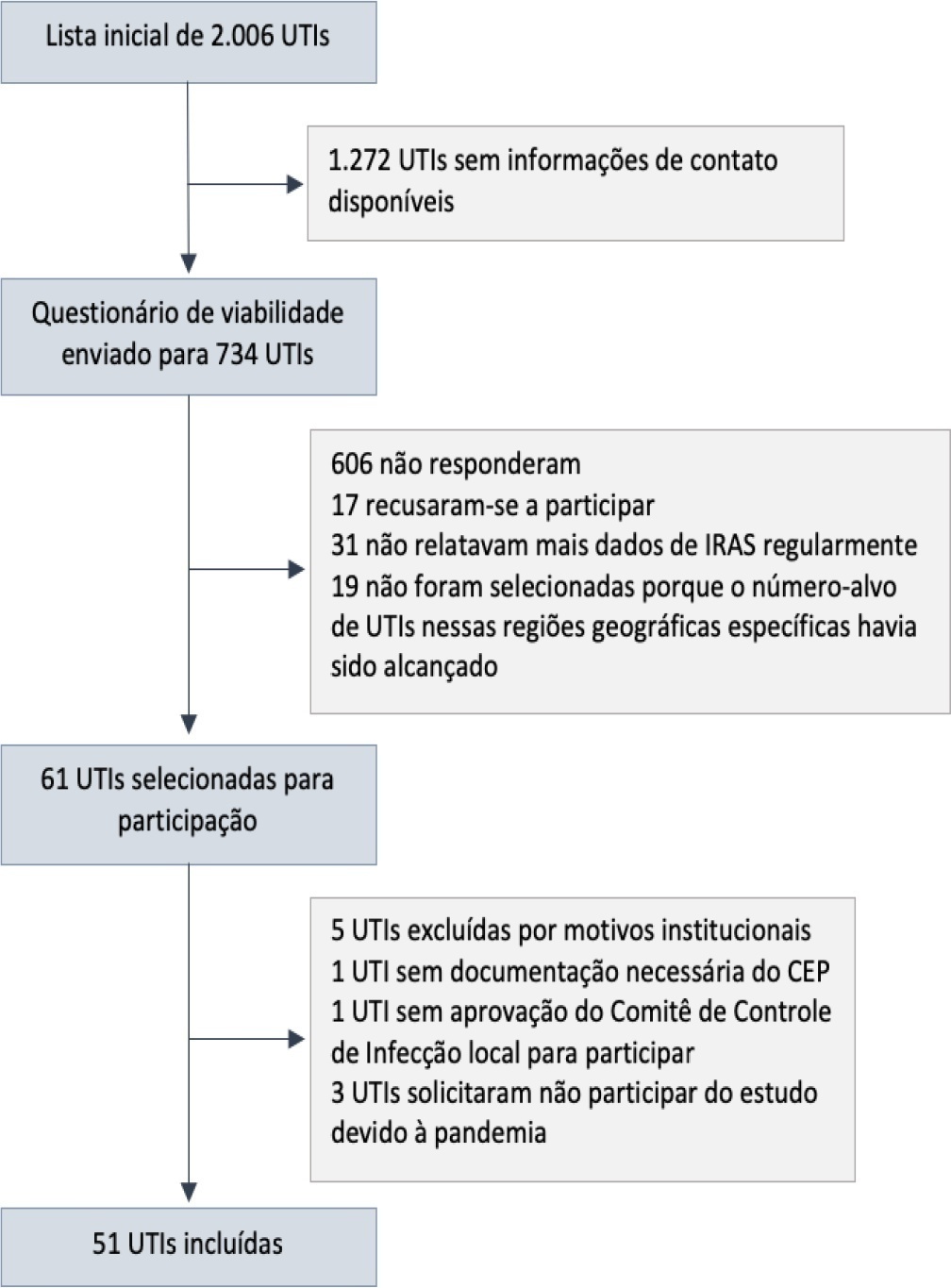
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.
Abstract
Rev Bras Ter Intensiva. 2011;23(4):426-433
DOI 10.1590/S0103-507X2011000400006
OBJECTIVE: To investigate the correlation of organ dysfunction and its progression with inflammatory response during the early phases of septic shock by assessing baseline cytokine concentrations. METHODS: This study included patients over 18 years old with septic shock within the first 48 hours after the onset of organ dysfunction. Interleukin 6 (IL-6), interleukin 8 (IL-8), interleukin 10 (IL-10) and C-reactive protein levels were assessed at inclusion and after 24 hours, and the differences between these values were calculated. The progression of organ dysfunction was assessed using the Sequential Organ Failure Assessment (SOFA) score upon admission and 24 hours later for a delta-SOFA determination and were categorized as either worsened or improved. The results were expressed as means + standard deviation or median (25-75% percentiles). Values with descriptive p values of 0.05 or less were considered significant. RESULTS: Overall, we included 41 patients with median SOFA scores of 8.0 (6.5-10.0) upon admission (T0) and 8.0 (6.0-10.0) 24 hours later (T1). Worsened, improved or unchanged SOFA scores were observed in 11 (Group 1), 17 (Group 2) and 13 (Group 3) patients, respectively. For Group 1, the baseline IL-6, IL-8 and IL-10 values were higher, and a significant increase of IL-8 levels was found after 24 hours. The change in the SOFA score after 24 hours was significantly, although weakly, correlated with baseline IL-6 and IL-8 concentrations. CONCLUSIONS: Higher baseline IL-6, IL-8 and IL-10 levels are associated with unfavorable organ dysfunction outcomes. Increased IL-8 levels within the first 24 hours are correlated with a worsening dysfunction.
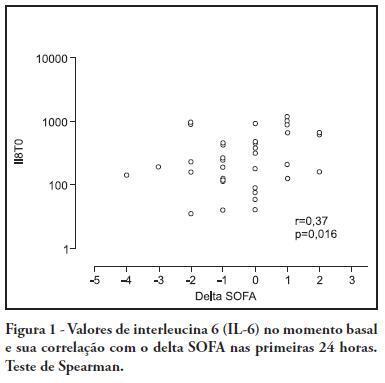
Abstract
Rev Bras Ter Intensiva. 2017;29(4):436-443
DOI 10.5935/0103-507X.20170070
To define the frequency of cytomegalovirus disease among kidney transplant patients in an intensive care unit in which this complication was suspected and to identify predisposing factors and their possible impact on clinical outcome.
Retrospective observational study in which kidney transplant patients over the age of 18 years were hospitalized for any reason in an intensive care unit with at least one collection of samples to test for the presence of antigenemia or cytomegalovirus via polymerase chain reaction during hospitalization. Cytomegalovirus disease was defined as positive antigenemia or polymerase chain reaction above 500 copies/mL in the presence of symptoms and in the appropriate clinical setting, as judged by the attending physician.
A total of 99 patients were included (age: 53.4 ± 12.8 years, 71.6% male). Cytomegalovirus disease was diagnosed in 39 patients (39.4%). Respiratory symptoms (51%), non-specific clinical worsening (20%) or gastrointestinal symptoms (14%) were the main reasons for exam collection. Transplant time was lower in those with cytomegalovirus disease than in those without this diagnosis (6.5 months and 31.2 months, p = 0.001), along with pulse therapy in the last 6 months (41% and 16.9%, p = 0.008) and previous use of thymoglobulin in the last year (35.9% and 6.8%, p < 0.001). In the logistic regression model, only the transplant time and the use of thymoglobulin were associated with a higher frequency of cytomegalovirus. There was no difference in clinical evolution between patients with and without cytomegalovirus disease.
In kidney transplant patients suspected of cytomegalovirus disease, the prevalence was high. Transplant time less than 6 months, and the use of thymoglobulin in the last year should increase the intensivist's suspicion for this complication.
Abstract
Rev Bras Ter Intensiva. 2006;18(1):45-51
DOI 10.1590/S0103-507X2006000100009
BACKGROUND AND OBJECTIVES: This study aims to describe the view of family members about the quality of care given in a general intensive care unit of a university hospital. METHODS: A questionnaire to evaluate the level of satisfaction with the care was elaborated. The study included family members of patients with a length of stay more than 48h who already had visited the patient one time or more during the period. The exclusion criterion was family of admitted patients with less than 48h of ICU stay, family members who had not visited the patient at all or family members who did not desire to answer the questionnaire for any personal reason. RESULTS: There were 100 relatives interviewed face to face. The most frequent complaint that had generated the greatest concern was the general status of the patient, present in 28% of the interviews. A total of 96% considered the quality of the medical team as excellent or good. However, 15% declared to be unsatisfied with the medical information given and the other 5%, although admitting satisfaction, complained about having to talk with different doctors each day. CONCLUSIONS: Imperfections in the communication appears as the main predictor of unsatisfactory quality of the service in the view of the family members. Although one cannot directly compare the degree of satisfaction between distinct studies due to different methodologies, we considered that in the presented sample the recognition of the most frequent factors of dissatisfaction can point out areas for improvement in the quality of care offered in the ICU.
Abstract
Rev Bras Ter Intensiva. 2020;32(4):493-505
DOI 10.5935/0103-507X.20200081
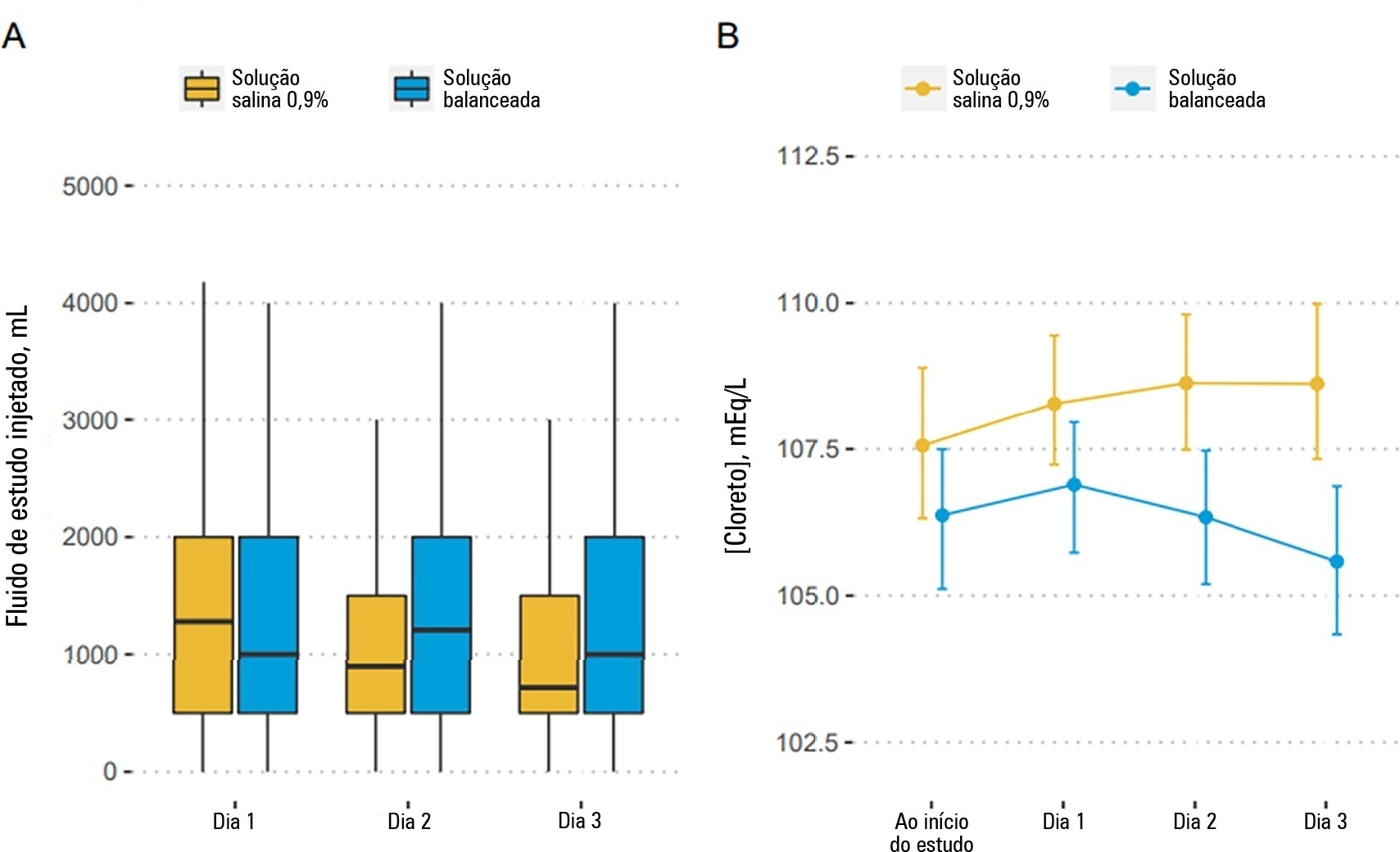
To report the statistical analysis plan (first version) for the Balanced Solutions versus Saline in Intensive Care Study (BaSICS).
BaSICS is a multicenter factorial randomized controlled trial that will assess the effects of Plasma-Lyte 148 versus 0.9% saline as the fluid of choice in critically ill patients, as well as the effects of a slow (333mL/h) versus rapid (999mL/h) infusion speed during fluid challenges, on important patient outcomes. The fluid type will be blinded for investigators, patients and the analyses. No blinding will be possible for the infusion speed for the investigators, but all analyses will be kept blinded during the analysis procedure.
BaSICS will have 90-day mortality as its primary endpoint, which will be tested using mixed-effects Cox proportional hazard models, considering sites as a random variable (frailty models) adjusted for age, organ dysfunction and admission type. Important secondary endpoints include renal replacement therapy up to 90 days, acute renal failure, organ dysfunction at days 3 and 7, and mechanical ventilation-free days within 28 days.
This manuscript provides details on the first version of the statistical analysis plan for the BaSICS trial and will guide the study’s analysis when follow-up is finished.