Abstract
Crit Care Sci. 2024;36:e20240005en
DOI 10.62675/2965-2774.20240005-en
To investigate the factors influencing carbon dioxide transfer in a system that integrates an oxygenation membrane in series with high-bicarbonate continuous veno-venous hemodialysis in hypercapnic animals.
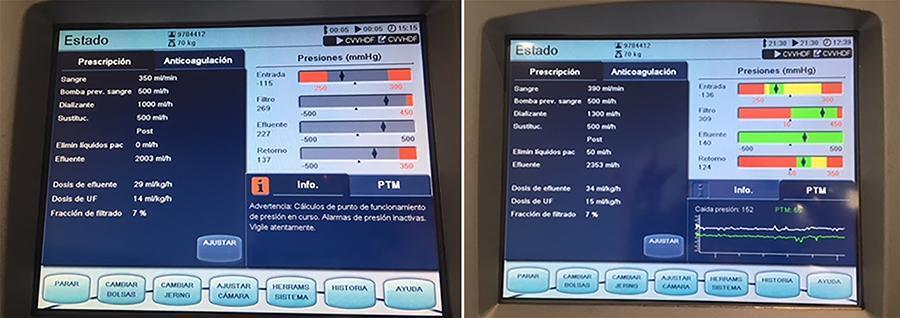
In an experimental setting, we induced severe acute kidney injury and hypercapnia in five female Landrace pigs. Subsequently, we initiated high (40mEq/L) bicarbonate continuous veno-venous hemodialysis with an oxygenation membrane in series to maintain a pH above 7.25. At intervals of 1 hour, 6 hours, and 12 hours following the initiation of continuous veno-venous hemodialysis, we performed standardized sweep gas flow titration to quantify carbon dioxide transfer. We evaluated factors associated with carbon dioxide transfer through the membrane lung with a mixed linear model.
A total of 20 sweep gas flow titration procedures were conducted, yielding 84 measurements of carbon dioxide transfer. Multivariate analysis revealed associations among the following (coefficients ± standard errors): core temperature (+7.8 ± 1.6 °C, p < 0.001), premembrane partial pressure of carbon dioxide (+0.2 ± 0.1/mmHg, p < 0.001), hemoglobin level (+3.5 ± 0.6/g/dL, p < 0.001), sweep gas flow (+6.2 ± 0.2/L/minute, p < 0.001), and arterial oxygen saturation (-0.5 ± 0.2%, p = 0.019). Among these variables, and within the physiological ranges evaluated, sweep gas flow was the primary modifiable factor influencing the efficacy of low-blood-flow carbon dioxide removal.
Sweep gas flow is the main carbon dioxide removal-related variable during continuous veno-venous hemodialysis with a high bicarbonate level coupled with an oxygenator. Other carbon dioxide transfer modulating variables included the hemoglobin level, arterial oxygen saturation, partial pressure of carbon dioxide and core temperature. These results should be interpreted as exploratory to inform other well-designed experimental or clinical studies.
Abstract
Rev Bras Ter Intensiva. 2020;32(1):115-122
DOI 10.5935/0103-507X.20200017
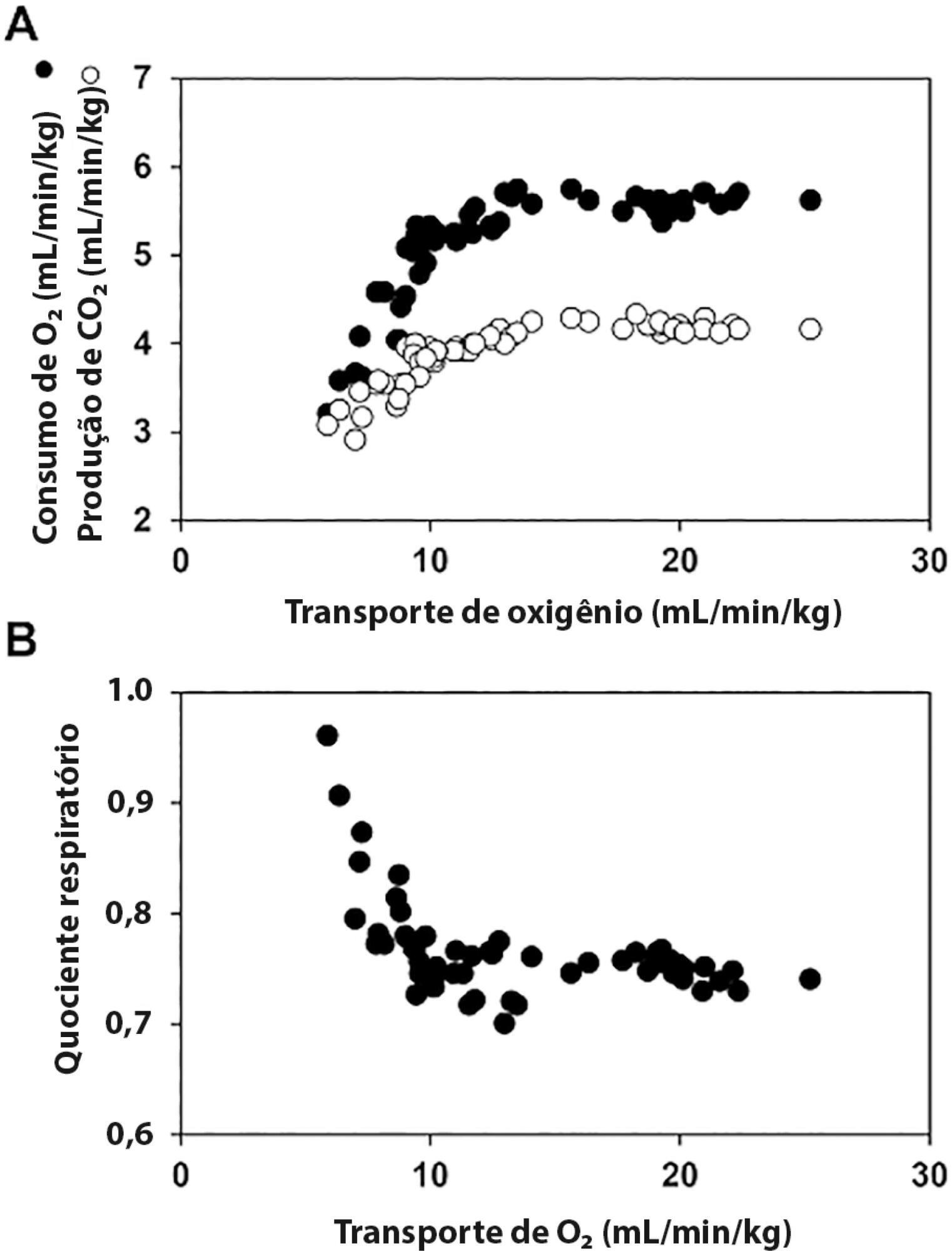
The central venous minus arterial carbon dioxide pressure to arterial minus central venous oxygen content ratio (Pcv-aCO2/Ca-cvO2) has been proposed as a surrogate for respiratory quotient and an indicator of tissue oxygenation. Some small observational studies have found that a Pcv-aCO2/Ca-cvO2 > 1.4 was associated with hyperlactatemia, oxygen supply dependency, and increased mortality. Moreover, Pcv-aCO2/Ca-cvO2 has been incorporated into algorithms for tissue oxygenation evaluation and resuscitation. However, the evidence for these recommendations is quite limited and of low quality. The goal of this narrative review was to analyze the methodological bases, the pathophysiologic foundations, and the experimental and clinical evidence supporting the use of Pcv-aCO2/Ca-cvO2 as a surrogate for respiratory quotient. Physiologically, the increase in respiratory quotient secondary to critical reductions in oxygen transport is a life-threatening and dramatic event. Nevertheless, this event is easily noticeable and probably does not require further monitoring. Since the beginning of anaerobic metabolism is indicated by the sudden increase in respiratory quotient and the normal range of respiratory quotient is wide, the use of a defined cutoff of 1.4 for Pcv-aCO2/Ca-cvO2 is meaningless. Experimental studies have shown that Pcv-aCO2/Ca-cvO2 is more dependent on factors that modify the dissociation of carbon dioxide from hemoglobin than on respiratory quotient and that respiratory quotient and Pcv-aCO2/Ca-cvO2 may have distinct behaviors. Studies performed in critically ill patients have shown controversial results regarding the ability of Pcv-aCO2/Ca-cvO2 to predict outcome, hyperlactatemia, microvascular abnormalities, and oxygen supply dependency. A randomized controlled trial also showed that Pcv-aCO2/Ca-cvO2 is useless as a goal of resuscitation. Pcv-aCO2/Ca-cvO2 should be carefully interpreted in critically ill patients.
Abstract
Rev Bras Ter Intensiva. 2020;32(1):143-148
DOI 10.5935/0103-507X.20200020
In recent years and due, in part, to technological advances, the use of extracorporeal carbon dioxide removal systems paired with the use of extracorporeal membrane oxygenation has resurfaced. However, studies are lacking that establish its indications and evidence to support its use. These systems efficiently eliminate carbon dioxide in patients with hypercapnic respiratory failure using small-bore cannula, usually double-lumen cannula with a small membrane lung surface area. Currently, we have several systems with different types of membranes and sizes. Pump-driven veno-venous systems generate fewer complications than do arteriovenous systems. Both require systemic anticoagulation. The “lung-kidney” support system, by combining a removal system with hemofiltration, simultaneously eliminates carbon dioxide and performs continuous extrarenal replacement. We describe our initial experience with a combined system for extracorporeal carbon dioxide removal-continuous extrarenal replacement in a lung transplant patients with hypercapnic respiratory failure, barotrauma and associated acute renal failure. The most important technical aspects, the effectiveness of the system for the elimination of carbon dioxide and a review of the literature are described.