You searched for:"Cintia Magalhães Carvalho Grion"
We found (14) results for your search.Abstract
Revista Brasileira de Terapia Intensiva. 2006;18(1):38-44
DOI 10.1590/S0103-507X2006000100008
BACKGROUND AND OBJECTIVES: The ventilator-associated pneumonia (VAP) is a severe infection that presents multiple causes which can vary depending on the type of intensive care unit, type of patients, emphasizing the needs for vigilance measures with local data. The aim of this study is to describe the incidence, risk factors and mortality of VAP in patients in a surgical ICU. METHODS: Prospective cohort conducted from January 2004 to January 2005. It was included all the patients in mechanical ventilation, followed daily to collect data about demographics, diagnostic, APACHE II and TISS 28 scores, duration of mechanical ventilation, length of stay, incidence of VAP and mortality. RESULTS: 462 patients were studied; age 57.2 ± 16.6 years, 55% men. The mean APACHE II score was 18.3 and the incidence of VAP was 18.8%. The TISS score at admission OR = 1.050 (IC 95%: 1.003-1.050) and the enteral nutrition OR = 5.609 (IC 3.351-9.388) were factors associated with VAP and the prophylactic use of antibiotics was a factor of protection OR = 0.399 (IC95%: 0.177-0.902). The patients with VAP had longer length of stay in ICU (10.3 ± 10.7 vs 4.9 ± 3.3 days), higher median of duration of mechanical ventilation (4 vs 1 days), higher mean of TISS 28 (24.4 ± 4.6 vs 22.8 ± 4.5), and higher crude mortality (46 vs 28.8%) when compared with the patients without VAP. CONCLUSIONS: VAP was a frequent infection in surgical patients in mechanical ventilation. Enteral nutrition and admission TISS were risk factors and the previous use of antibiotics was protection factor to develop VAP. In our sample the results demonstrate that VAP is associated with higher duration in mechanical ventilation, longer length of stay and higher mortality.
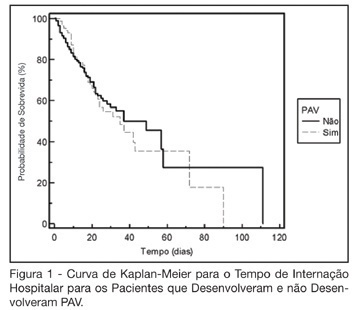
Abstract
Revista Brasileira de Terapia Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.

Abstract
Revista Brasileira de Terapia Intensiva. 2022;34(4):484-491
DOI 10.5935/0103-507X.20220264-en
To obtain data on bed refusal in intensive care units in Brazil and to evaluate the use of triage systems by professionals.
A cross-sectional survey. Using the Delphi methodology, a questionnaire was created contemplating the objectives of the study. Physicians and nurses enrolled in the research network of the Associação de Medicina Intensiva Brasileira (AMIBnet) were invited to participate. A web platform (SurveyMonkey®) was used to distribute the questionnaire. The variables in this study were measured in categories and expressed as proportions. The chi-square test or Fisher’s exact test was used to verify associations. The significance level was set at 5%.
In total, 231 professionals answered the questionnaire, representing all regions of the country. The national intensive care units had an occupancy rate of more than 90% always or frequently for 90.8% of the participants. Among the participants, 84.4% had already refused admitting patients to the intensive care unit due to the capacity of the unit. Half of the Brazilian institutions (49.7%) did not have triage protocols for admission to intensive beds.
Bed refusal due to high occupancy rates is common in Brazilian intensive care units. Even so, half of the services in Brazil do not adopt protocols for triage of beds.
Abstract
Revista Brasileira de Terapia Intensiva. 2020;32(4):493-505
DOI 10.5935/0103-507X.20200081
To report the statistical analysis plan (first version) for the Balanced Solutions versus Saline in Intensive Care Study (BaSICS).
BaSICS is a multicenter factorial randomized controlled trial that will assess the effects of Plasma-Lyte 148 versus 0.9% saline as the fluid of choice in critically ill patients, as well as the effects of a slow (333mL/h) versus rapid (999mL/h) infusion speed during fluid challenges, on important patient outcomes. The fluid type will be blinded for investigators, patients and the analyses. No blinding will be possible for the infusion speed for the investigators, but all analyses will be kept blinded during the analysis procedure.
BaSICS will have 90-day mortality as its primary endpoint, which will be tested using mixed-effects Cox proportional hazard models, considering sites as a random variable (frailty models) adjusted for age, organ dysfunction and admission type. Important secondary endpoints include renal replacement therapy up to 90 days, acute renal failure, organ dysfunction at days 3 and 7, and mechanical ventilation-free days within 28 days.
This manuscript provides details on the first version of the statistical analysis plan for the BaSICS trial and will guide the study’s analysis when follow-up is finished.
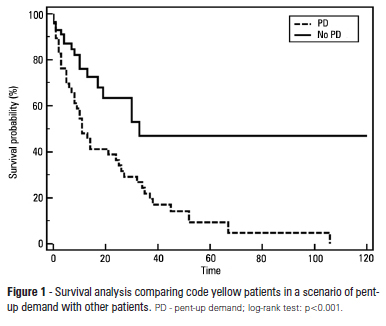
Abstract
Revista Brasileira de Terapia Intensiva. 2013;25(2):99-105
DOI 10.5935/0103-507X.20130020
OBJECTIVE: To describe the epidemiological data of the clinical instability events in patients attended to by the rapid response team and to identify prognostic factors. METHODS: This was a longitudinal study, performed from January to July 2010, with an adult inpatient population in a hospital environment. The data collected regarding the code yellow service included the criteria of the clinical instability, the drug and non-drug therapies administered and the activities and procedures performed. The outcomes evaluated were the need for intensive care unit admission and the hospital mortality rates. A level of p=0.05 was considered to be significant. RESULTS: A total of 150 code yellow events that occurred in 104 patients were evaluated. The most common causes were related to acute respiratory insufficiency with hypoxia or a change in the respiratory rate and a concern of the team about the patient's clinical condition. It was necessary to request a transfer to the intensive care unit in 80 of the 150 cases (53.3%). It was necessary to perform 42 procedures. The most frequent procedures were orotracheal intubation and the insertion of a central venous catheter. The patients who were in critical condition and had to wait for an intensive care unit bed had a higher risk of death compared to the other patients (hazard ratio: 3.12; 95% CI: 1.80-5.40; p<0.001). CONCLUSIONS: There are patients in critical condition that require expert intensive care in the regular ward unit hospital beds. The events that most frequently led to the code yellow activation were related to hemodynamic and respiratory support. The interventions performed indicate the need for a physician on the team. The situation of pent-up demand is associated with a higher mortality rate.