-
Special Article
The II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
Abstract
Special ArticleThe II Brazilian Guidelines for the pharmacological treatment of patients hospitalized with COVID-19 Joint Guidelines of the Associação Brasileira de Medicina de Emergência, Associação de Medicina Intensiva Brasileira, Associação Médica Brasileira, Sociedade Brasileira de Angiologia e Cirurgia Vascular, Sociedade Brasileira de Infectologia, Sociedade Brasileira de Pneumologia e Tisiologia and Sociedade Brasileira de Reumatologia
Crit Care Sci. 2023;35(3):243-255
DOI 10.5935/2965-2774.20230136-pt
Views22ABSTRACT
Objective:
To update the recommendations to support decisions regarding the pharmacological treatment of patients hospitalized with COVID-19 in Brazil.
Methods:
Experts, including representatives of the Ministry of Health and methodologists, created this guideline. The method used for the rapid development of guidelines was based on the adoption and/or adaptation of existing international guidelines (GRADE ADOLOPMENT) and supported by the e-COVID-19 RecMap platform. The quality of the evidence and the preparation of the recommendations followed the GRADE method.
Results:
Twenty-one recommendations were generated, including strong recommendations for the use of corticosteroids in patients using supplemental oxygen and conditional recommendations for the use of tocilizumab and baricitinib for patients on supplemental oxygen or on noninvasive ventilation and anticoagulants to prevent thromboembolism. Due to suspension of use authorization, it was not possible to make recommendations regarding the use of casirivimab + imdevimab. Strong recommendations against the use of azithromycin in patients without suspected bacterial infection, hydroxychloroquine, convalescent plasma, colchicine, and lopinavir + ritonavir and conditional recommendations against the use of ivermectin and remdesivir were made.
Conclusion:
New recommendations for the treatment of hospitalized patients with COVID-19 were generated, such as those for tocilizumab and baricitinib. Corticosteroids and prophylaxis for thromboembolism are still recommended, the latter with conditional recommendation. Several drugs were considered ineffective and should not be used to provide the best treatment according to the principles of evidence-based medicine and to promote resource economy.
Keywords:BrazilCoronavirus infectionsCOVID-19COVID-19/drug therapyHealth planning guidelinesSARS-CoV-2See more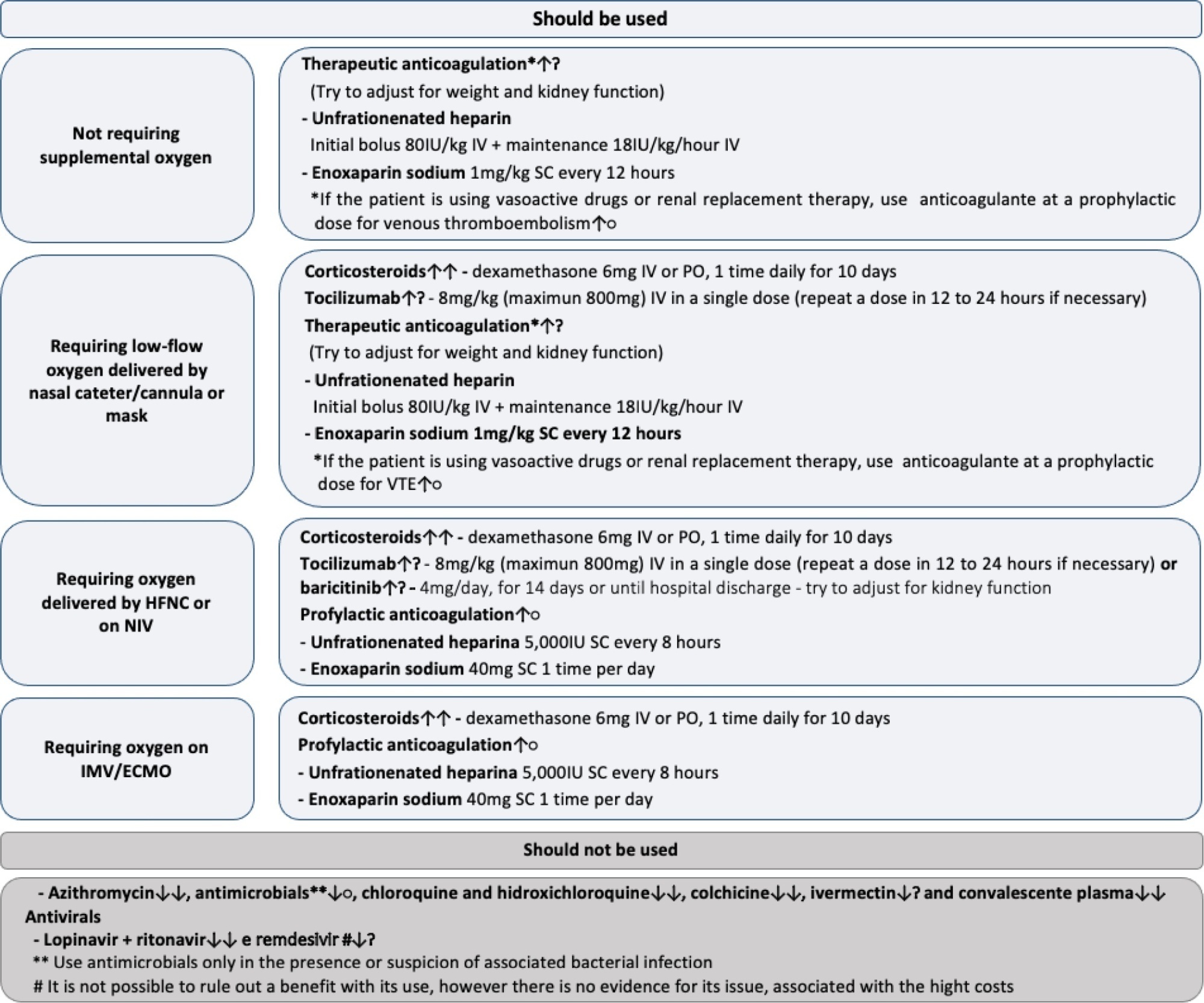
-
Special Article
Dapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
Abstract
Special ArticleDapagliflozin in patients with critical illness: rationale and design of the DEFENDER study
Crit Care Sci. 2023;35(3):256-265
DOI 10.5935/2965-2774.20230129-pt
Views9ABSTRACT
Background:
Critical illness is a major ongoing health care burden worldwide and is associated with high mortality rates. Sodium-glucose cotransporter-2 inhibitors have consistently shown benefits in cardiovascular and renal outcomes. The effects of sodium-glucose cotransporter-2 inhibitors in acute illness have not been properly investigated.
Methods:
DEFENDER is an investigator-initiated, multicenter, randomized, open-label trial designed to evaluate the efficacy and safety of dapagliflozin in 500 adult participants with acute organ dysfunction who are hospitalized in the intensive care unit. Eligible participants will be randomized 1:1 to receive dapagliflozin 10mg plus standard of care for up to 14 days or standard of care alone. The primary outcome is a hierarchical composite of hospital mortality, initiation of kidney replacement therapy, and intensive care unit length of stay, up to 28 days. Safety will be strictly monitored throughout the study.
Conclusion:
DEFENDER is the first study designed to investigate the use of a sodium-glucose cotransporter-2 inhibitor in general intensive care unit patients with acute organ dysfunction. It will provide relevant information on the use of drugs of this promising class in critically ill patients.
ClinicalTrials.gov registry:
NCT05558098
Keywords:Critical care outcomesCritical illnessOrgan dysfunctionSodium-glucose transporter 2 inhibitorsSee more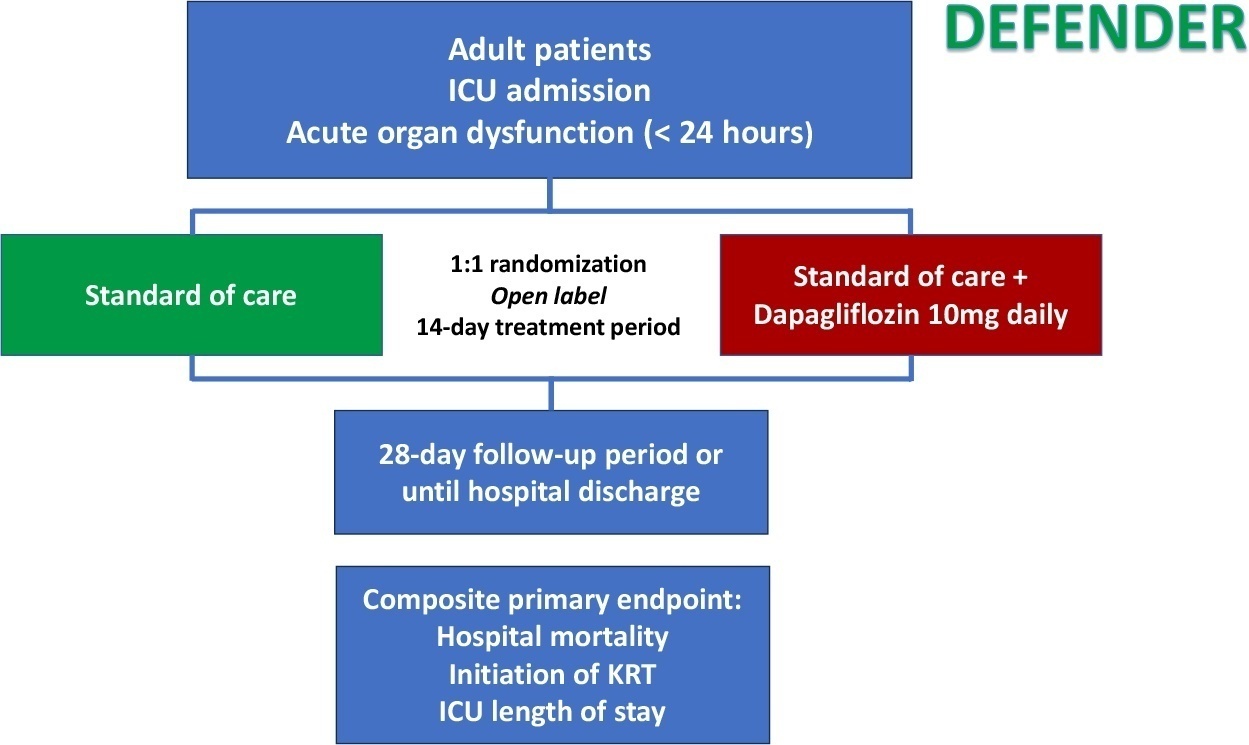
-
Special Article
Impact of Telemedicine use on clinical care indicators of pediatric intensive care units: protocol for a cluster randomized clinical trial
Crit Care Sci. 2023;35(3):266-272
Abstract
Special ArticleImpact of Telemedicine use on clinical care indicators of pediatric intensive care units: protocol for a cluster randomized clinical trial
Crit Care Sci. 2023;35(3):266-272
DOI 10.5935/2965-2774.20230223-pt
Views6ABSTRACT
The objective of this study is to present the protocol of a cluster randomized clinical trial to be conducted through the TeleICU project – Qualification of Intensive Care by Telemedicine. The study will consist of a cluster randomized clinical trial, open label, in pediatric intensive care units, with an allocation ratio of 1:1, to compare the intervention group (support of Telemedicine for patients admitted to the pediatric intensive care unit) with a control group (pediatric intensive care unit usual care). The study proposed to select 16 pediatric intensive care units, including 100 participants per site, with a total of 1,600 participants. The intervention group will receive telerounds from Monday to Friday and will have specialists and continuing education activities available. The primary outcome measure will be the length of stay in the pediatric intensive care unit, defined as the difference between the date of discharge of the participant and the date of admission to the intensive care unit. The secondary outcomes will be mortality rate, invasive mechanical ventilation-free days, days using antibiotics, days using vasoactive drugs and days using sedoanalgesia. This study will be conducted in accordance with Resolution 466/12 of the National Health Council, with approval by the Research Ethics Committee of the institutions involved. The present study has the potential to reproduce studies on Telemedicine in intensive care and may make important contributions to care in intensive care units in Brazil and other settings. If Telemedicine shows positive clinical care results compared to conventional treatment, more pediatric patients may benefit.
ClinicalTrials.gov registry: NCT05260710
Keywords:Clinical trial protocolIntensive careIntensive care unitspediatricRandomized controlled trialTelemedicineSee more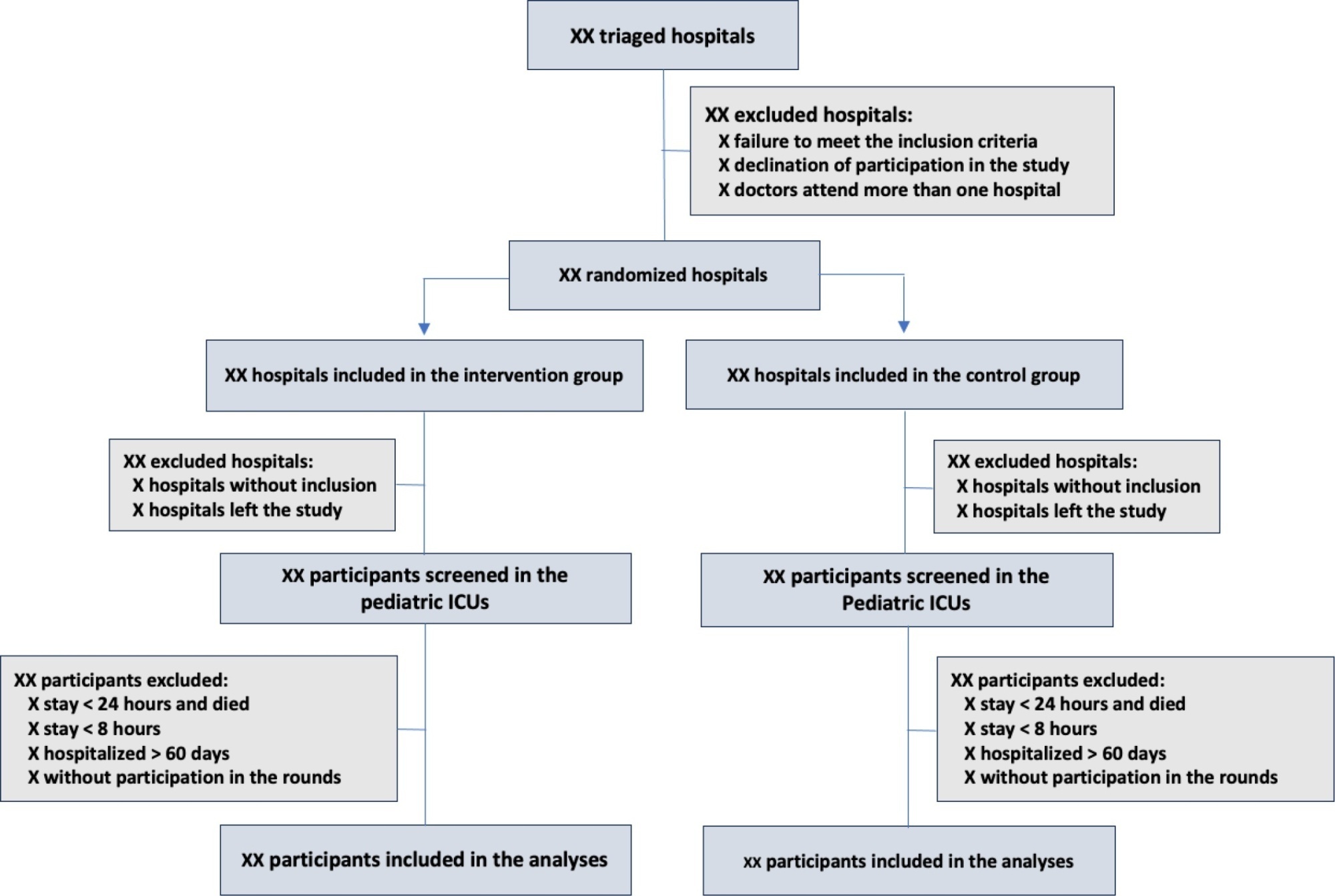
-
Original Article
Performance and labor conditions of physiotherapists in Brazilian intensive care units during the COVID-19 pandemic. What did we learn?
Crit Care Sci. 2023;35(3):273-280
Abstract
Original ArticlePerformance and labor conditions of physiotherapists in Brazilian intensive care units during the COVID-19 pandemic. What did we learn?
Crit Care Sci. 2023;35(3):273-280
DOI 10.5935/2965-2774.20230359-pt
Views5ABSTRACT
Objective:
To describe the role of physiotherapists in assisting patients suspected to have or diagnosed with COVID-19 hospitalized in intensive care units in Brazil regarding technical training, working time, care practice, labor conditions and remuneration.
Methods:
An analytical cross-sectional survey was carried out through an electronic questionnaire distributed to physiotherapists who worked in the care of patients with COVID-19 in Brazilian intensive care units.
Results:
A total of 657 questionnaires were completed by physiotherapists from the five regions of the country, with 85.3% working in adult, 5.4% in neonatal, 5.3% in pediatric and 3.8% in mixed intensive care units (pediatric and neonatal). In intensive care units with a physiotherapists available 24 hours/day, physiotherapists worked more frequently (90.6%) in the assembly, titration, and monitoring of noninvasive ventilation (p = 0.001). Most intensive care units with 12-hour/day physiotherapists (25.8%) did not apply any protocol compared to intensive care units with 18-hour/day physiotherapy (9.9%) versus 24 hours/day (10.2%) (p = 0.032). Most of the respondents (51.0%) received remuneration 2 or 3 times the minimum wage, and only 25.1% received an additional payment for working with patients suspected to have or diagnosed with COVID-19; 85.7% of them did not experience a lack of personal protective equipment.
Conclusion:
Intensive care units with 24-hour/day physiotherapists had higher percentages of protocols and noninvasive ventilation for patients with COVID-19. The use of specific resources varied between the types of intensive care units and hospitals and in relation to the physiotherapists’ labor conditions. This study showed that most professionals had little experience in intensive care and low wages.
Keywords:COVID-19Intensive care unitsOccupational risksPhysiotherapistsProfessional trainingRemunerationSurvey and questionnairesWork hoursSee more -
Original Article
Physical rehabilitation in Brazilian pediatric intensive care units: a multicenter point prevalence study
Crit Care Sci. 2023;35(3):290-301
Abstract
Original ArticlePhysical rehabilitation in Brazilian pediatric intensive care units: a multicenter point prevalence study
Crit Care Sci. 2023;35(3):290-301
DOI 10.5935/2965-2774.20230388-pt
Views5ABSTRACT
Objective:
To determine the prevalence and factors associated with the physical rehabilitation of critically ill children in Brazilian pediatric intensive care units.
Methods:
A 2-day, cross-sectional, multicenter point prevalence study comprising 27 pediatric intensive care units (out of 738) was conducted in Brazil in April and June 2019. This Brazilian study was part of a large multinational study called Prevalence of Acute Rehabilitation for Kids in the PICU (PARK-PICU). The primary outcome was the prevalence of mobility provided by physical therapy or occupational therapy. Clinical data on patient mobility, potential mobility safety events, and mobilization barriers were prospectively collected in patients admitted for ≥ 72 hours.
Results:
Children under the age of 3 years comprised 68% of the patient population. The prevalence of therapist-provided mobility was 74%, or 277 out of the 375 patient-days. Out-of-bed mobility was most positively associated with family presence (adjusted odds ratios 3.31;95%CI 1.70 – 6.43) and most negatively associated with arterial lines (adjusted odds ratios 0.16; 95%CI 0.05 – 0.57). Barriers to mobilization were reported on 27% of patient-days, the most common being lack of physician order (n = 18). Potential safety events occurred in 3% of all mobilization events.
Conclusion:
Therapist-provided mobility in Brazilian pediatric intensive care units is frequent. Family presence was high and positively associated with out-of-bed mobility. The presence of physiotherapists 24 hours a day in Brazilian pediatric intensive care units may have a substantial impact on the mobilization of critically ill children.
Keywords:Critical careIntensive care unitsOccupational therapypediatricPhysical therapy modalitiesRehabilitationSee more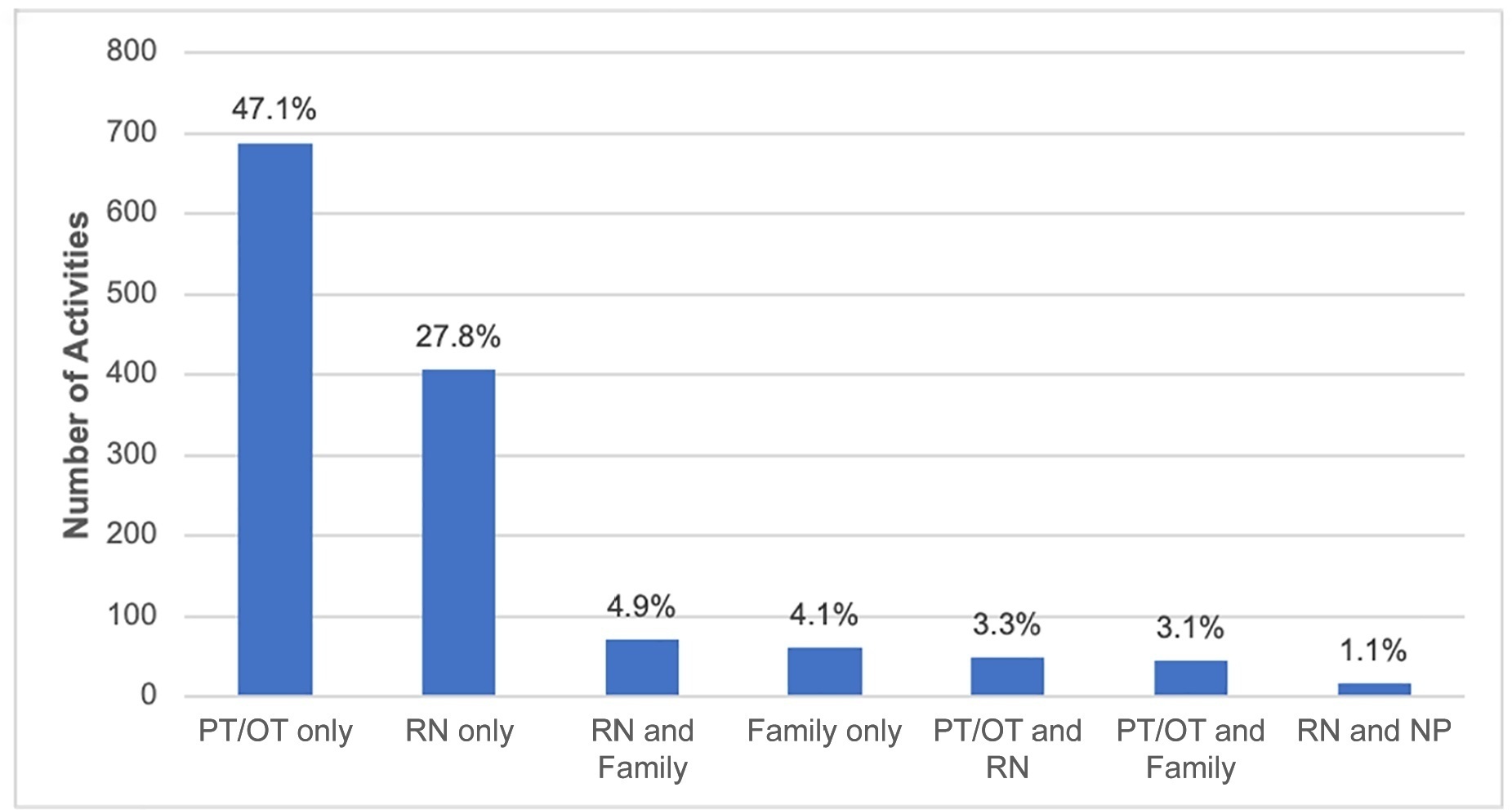
-
Original Article
Accuracy of the persistent AKI risk index in predicting acute kidney injury in patients admitted to the intensive care unit for acute respiratory failure
Crit Care Sci. 2023;35(3):302-310
Abstract
Original ArticleAccuracy of the persistent AKI risk index in predicting acute kidney injury in patients admitted to the intensive care unit for acute respiratory failure
Crit Care Sci. 2023;35(3):302-310
DOI 10.5935/2965-2774.20230141-pt
Views5ABSTRACT
Objective:
To evaluate the accuracy of the persistent AKI risk index (PARI) in predicting acute kidney injury within 72 hours after admission to the intensive care unit, persistent acute kidney injury, renal replacement therapy, and death within 7 days in patients hospitalized due to acute respiratory failure.
Methods:
This study was done in a cohort of diagnoses of consecutive adult patients admitted to the intensive care unit of eight hospitals in Curitiba, Brazil, between March and September 2020 due to acute respiratory failure secondary to suspected COVID-19. The COVID-19 diagnosis was confirmed or refuted by RT-PCR for the detection of SARS-CoV-2. The ability of PARI to predict acute kidney injury at 72 hours, persistent acute kidney injury, renal replacement therapy, and death within 7 days was analyzed by ROC curves in comparison to delta creatinine, SOFA, and APACHE II.
Results:
Of the 1,001 patients in the cohort, 538 were included in the analysis. The mean age was 62 ± 17 years, 54.8% were men, and the median APACHE II score was 12. At admission, the median SOFA score was 3, and 83.3% had no renal dysfunction. After admission to the intensive care unit, 17.1% had acute kidney injury within 72 hours, and through 7 days, 19.5% had persistent acute kidney injury, 5% underwent renal replacement therapy, and 17.1% died. The PARI had an area under the ROC curve of 0.75 (0.696 – 0.807) for the prediction of acute kidney injury at 72 hours, 0.71 (0.613 – 0.807) for renal replacement therapy, and 0.64 (0.565 – 0.710) for death.
Conclusion:
The PARI has acceptable accuracy in predicting acute kidney injury within 72 hours and renal replacement therapy within 7 days of admission to the intensive care unit, but it is not significantly better than the other scores.
Keywords:Acute kidney injuryCoronavirus infectionsCOVID-19DeathIntensive care unitsMortalityPrognosisRenal replacement therapyRespiratory insufficiencySARS-CoV-2See more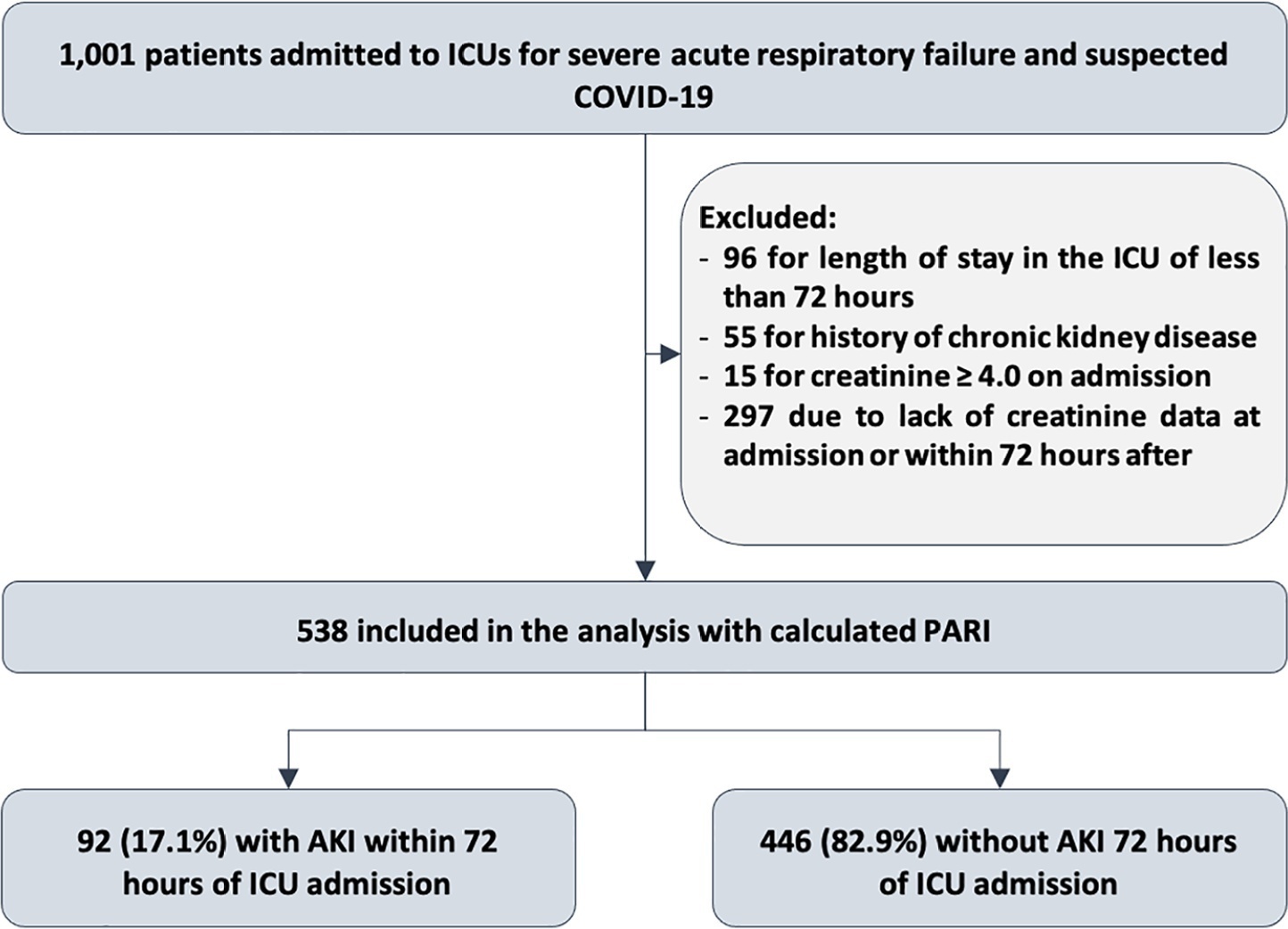
-
Original Article
Translation and cross-cultural adaptation of the anchor points of the Cornell Assessment of Pediatric Delirium scale into Portuguese
Crit Care Sci. 2023;35(3):320-327
Abstract
Original ArticleTranslation and cross-cultural adaptation of the anchor points of the Cornell Assessment of Pediatric Delirium scale into Portuguese
Crit Care Sci. 2023;35(3):320-327
DOI 10.5935/2965-2774.20230165-pt
Views6See moreABSTRACT
Objective:
To translate and cross-culturally adapt the Cornell Assessment of Pediatric Delirium anchor points from English to Brazilian Portuguese.
Methods:
For the translation and cross-cultural adaptation of the anchor points, all steps recommended internationally were followed after authorization for use by the lead author. The stages were as follows: translation of the original version into Portuguese by two bilingual translators who were native speakers of the target language, synthesis of the versions, reverse translation by two translators who were native speakers of the source language, review and synthesis of the back-translation, review by a committee of experts and preparation of the final version.
Results:
The translation and cross-cultural adaptation of the anchor points was conducted in accordance with recommendations. The linguistic and semantic issues that arose were discussed by a committee of judges, with 91.8% agreement, as determined using a Likert scale, after changes by consensus. After reanalysis by the authors, there were no changes, resulting in the final version, which was easy to understand and administer.
Conclusion:
The translation and cross-cultural adaptation of the anchor points of the Cornell Assessment of Pediatric Delirium scale into Portuguese spoken in Brazil were successful, maintaining the linguistic and semantic properties of the original instrument. The table of anchor points is easy to understand and will be helpful during the assessment of children younger than 24 months using the Cornell Assessment of Pediatric Delirium scale.
-
Letter to the Editor
Trauma induced coagulopathy and fibrinogen levels: why do we need to measure them, and what are the supplementation strategies?
Crit Care Sci. 2023;35(3):328-330
Abstract
Letter to the EditorTrauma induced coagulopathy and fibrinogen levels: why do we need to measure them, and what are the supplementation strategies?
Crit Care Sci. 2023;35(3):328-330
DOI 10.5935/2965-2774.20230132-pt
Views8INTRODUCTIONFibrinogen is a large glycoprotein produced in the liver. With a normal plasma concentration of 1.5 – 3.5g/L, fibrinogen is the most abundant blood coagulation factor. The final stage of blood clot formation is the conversion of soluble fibrinogen to insoluble fibrin, leading to a stable clot.() In cases of severe bleeding, fibrinogen reaches critically […]See more -
Letter to the Editor
To: The Respiratory Rate-Oxygenation Index predicts failure of post-extubation high-flow nasal cannula therapy in intensive care unit patients: a retrospective cohort study
Crit Care Sci. 2023;35(3):331-332
Abstract
Letter to the EditorTo: The Respiratory Rate-Oxygenation Index predicts failure of post-extubation high-flow nasal cannula therapy in intensive care unit patients: a retrospective cohort study
Crit Care Sci. 2023;35(3):331-332
DOI 10.5935/2965-2774.20230366-pt
Views7To the editor,We have read with interest the study entitled “The respiratory rate-oxygenation index predicts failure of post extubation high-flow nasal cannula therapy in intensive care unit patients: a retrospective cohort study” published by Fuentes et al. in this journal.() The authors proposed the respiratory rate-oxygenation (ROX) index as a predictor of post extubation high-flow […]See more -
Letter to the Editor
To: Critical COVID-19 and neurological dysfunction – a direct comparative analysis between SARS-CoV-2 and other infectious pathogens
Crit Care Sci. 2023;35(3):335-336
Abstract
Letter to the EditorTo: Critical COVID-19 and neurological dysfunction – a direct comparative analysis between SARS-CoV-2 and other infectious pathogens
Crit Care Sci. 2023;35(3):335-336
DOI 10.5935/2965-2774.20230383-pt
Views5To the Editor,Teixeira-Vaz et al. deserve applause for staging a prospective analysis of neurological dysfunction ensuing after infection from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) versus other pathogens.() While the authors show that critically ill coronavirus disease 2019 (COVID-19) patients are prone to neurological complications, there is a need to consider additional factors in […]See more -
Letter to the Editor
To: Critical COVID-19 and neurological dysfunction – a direct comparative analysis between SARS-CoV-2 and other infectious pathogens
Crit Care Sci. 2023;35(3):338-339
Abstract
Letter to the EditorTo: Critical COVID-19 and neurological dysfunction – a direct comparative analysis between SARS-CoV-2 and other infectious pathogens
Crit Care Sci. 2023;35(3):338-339
DOI 10.5935/2965-2774.20230104-pt
Views6To the EditorWith interest, we read the article by Teixeira-Vaz et al. on a prospective, single-center cohort study among 27 coronavirus disease 2019 (COVID-19) patients requiring mechanical ventilation > 48 hours for acute respiratory distress syndrome (ARDS).() Compared to a disease control cohort, COVID-19 ARDS patients had an increased risk of neurological complications and corticospinal […]See more