You searched for:"Alvaro Avezum"
We found (7) results for your search.-
Original Article
Clinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
Abstract
Original ArticleClinical outcomes and lung mechanics characteristics between COVID-19 and non-COVID-19-associated acute respiratory distress syndrome: a propensity score analysis of two major randomized trials
Rev Bras Ter Intensiva. 2022;34(3):335-341
DOI 10.5935/0103-507X.20220040-en
Views3ABSTRACT
Objective:
To compare the lung mechanics and outcomes between COVID-19-associated acute respiratory distress syndrome and non-COVID-19-associated acute respiratory distress syndrome.
Methods:
We combined data from two randomized trials in acute respiratory distress syndrome, one including only COVID-19 patients and the other including only patients without COVID-19, to determine whether COVID-19-associated acute respiratory distress syndrome is associated with higher 28-day mortality than non-COVID-19 acute respiratory distress syndrome and to examine the differences in lung mechanics between these two types of acute respiratory distress syndrome.
Results:
A total of 299 patients with COVID-19-associated acute respiratory distress syndrome and 1,010 patients with non-COVID-19-associated acute respiratory distress syndrome were included in the main analysis. The results showed that non-COVID-19 patients used higher positive end-expiratory pressure (12.5cmH2O; SD 3.2 versus 11.7cmH2O SD 2.8; p < 0.001), were ventilated with lower tidal volumes (5.8mL/kg; SD 1.0 versus 6.5mL/kg; SD 1.2; p < 0.001) and had lower static respiratory compliance adjusted for ideal body weight (0.5mL/cmH2O/kg; SD 0.3 versus 0.6mL/cmH2O/kg; SD 0.3; p = 0.01). There was no difference between groups in 28-day mortality (52.3% versus 58.9%; p = 0.52) or mechanical ventilation duration in the first 28 days among survivors (13 [IQR 5 – 22] versus 12 [IQR 6 – 26], p = 0.46).
Conclusion:
This analysis showed that patients with non-COVID-19-associated acute respiratory distress syndrome have different lung mechanics but similar outcomes to COVID-19-associated acute respiratory distress syndrome patients. After propensity score matching, there was no difference in lung mechanics or outcomes between groups.
Keywords:Coronavirus infectionsCOVID-19Critical careCritical care outcomesRespiratory distress syndromeRespiratory mechanicsSee more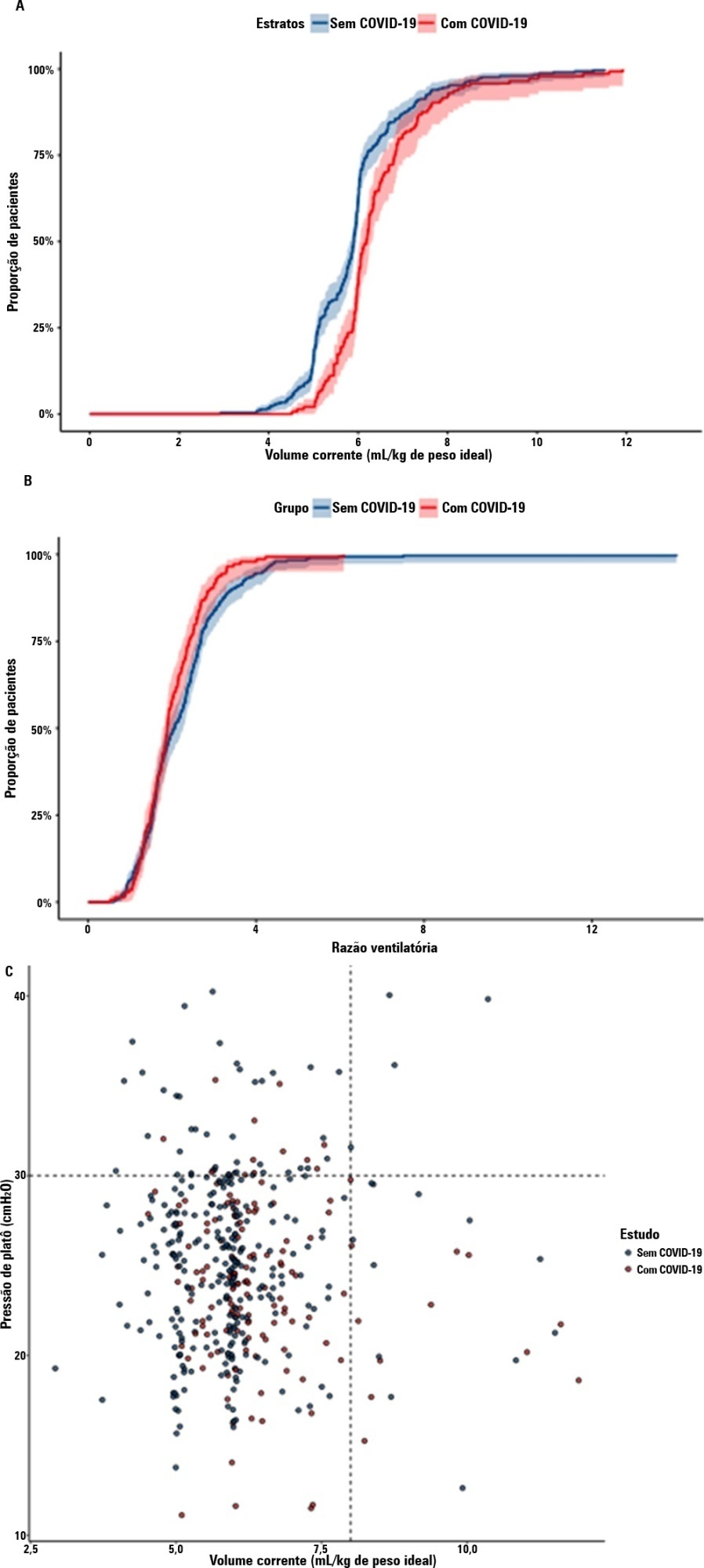
-
Special Article
Quality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
Abstract
Special ArticleQuality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
Views0See moreAbstract
Introduction:
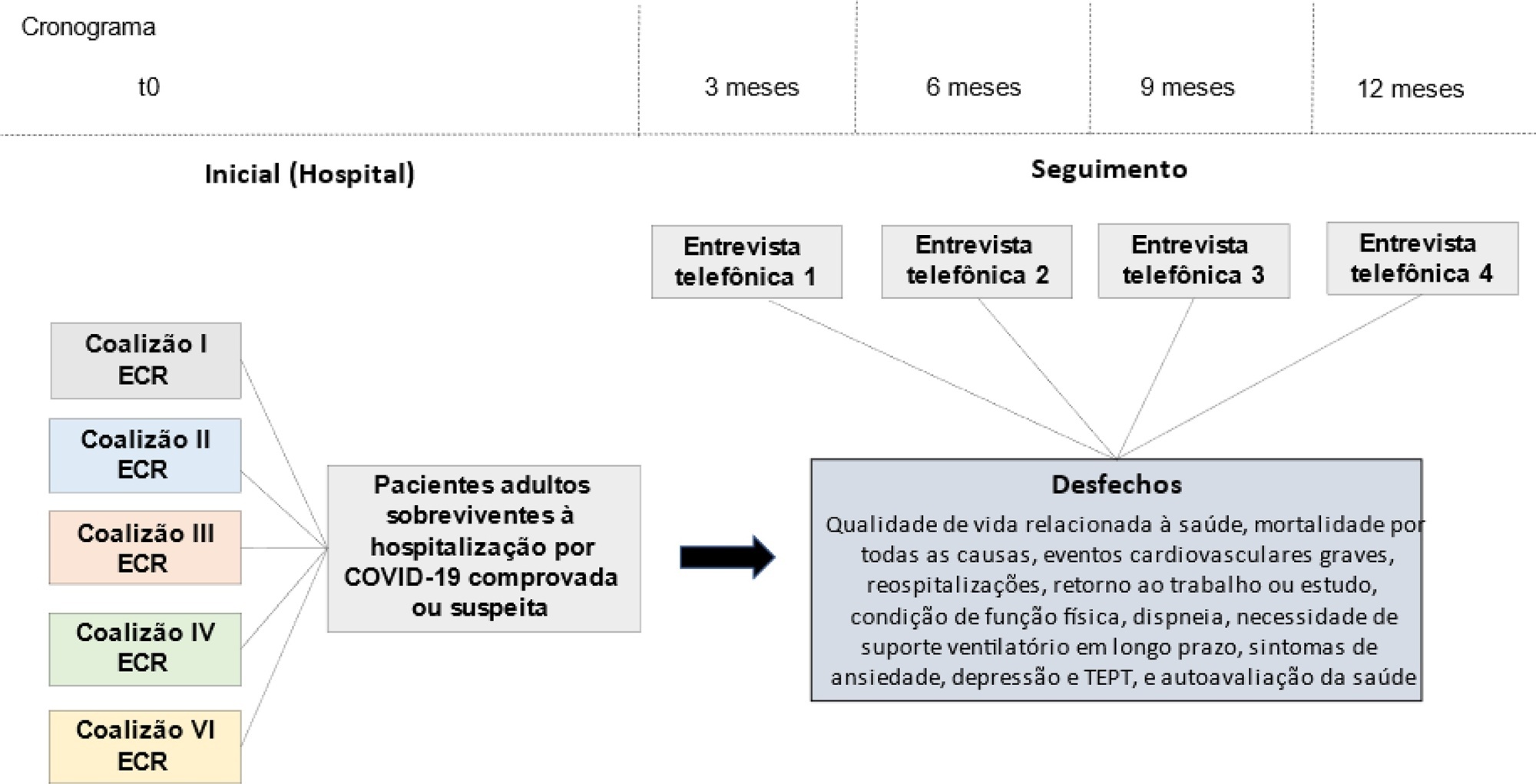
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
Methods:
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
Ethics and dissemination:
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
-
Original Article
COVID-19-associated ARDS treated with DEXamethasone (CoDEX): study design and rationale for a randomized trial
Rev Bras Ter Intensiva. 2020;32(3):354-362
Abstract
Original ArticleCOVID-19-associated ARDS treated with DEXamethasone (CoDEX): study design and rationale for a randomized trial
Rev Bras Ter Intensiva. 2020;32(3):354-362
DOI 10.5935/0103-507X.20200063
Views0Abstract
Objective:
The infection caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spreads worldwide and is considered a pandemic. The most common manifestation of SARS-CoV-2 infection (coronavirus disease 2019 – COVID-19) is viral pneumonia with varying degrees of respiratory compromise and up to 40% of hospitalized patients might develop acute respiratory distress syndrome. Several clinical trials evaluated the role of corticosteroids in non-COVID-19 acute respiratory distress syndrome with conflicting results. We designed a trial to evaluate the effectiveness of early intravenous dexamethasone administration on the number of days alive and free of mechanical ventilation within 28 days after randomization in adult patients with moderate or severe acute respiratory distress syndrome due to confirmed or probable COVID-19.
Methods:
This is a pragmatic, prospective, randomized, stratified, multicenter, open-label, controlled trial including 350 patients with early-onset (less than 48 hours before randomization) moderate or severe acute respiratory distress syndrome, defined by the Berlin criteria, due to COVID-19. Eligible patients will be randomly allocated to either standard treatment plus dexamethasone (Intervention Group) or standard treatment without dexamethasone (Control Group). Patients in the intervention group will receive dexamethasone 20mg intravenous once daily for 5 days, followed by dexamethasone 10mg IV once daily for additional 5 days or until intensive care unit discharge, whichever occurs first. The primary outcome is ventilator-free days within 28 days after randomization, defined as days alive and free from invasive mechanical ventilation. Secondary outcomes are all-cause mortality rates at day 28, evaluation of the clinical status at day 15 assessed with a 6-level ordinal scale, mechanical ventilation duration from randomization to day 28, Sequential Organ Failure Assessment Score evaluation at 48 hours, 72 hours and 7 days and intensive care unit -free days within 28.
Keywords:Adrenal cortex hormonesCoronavirusCOVID-19Critical careDexamethasoneRespiratory distress syndrome, adultSee more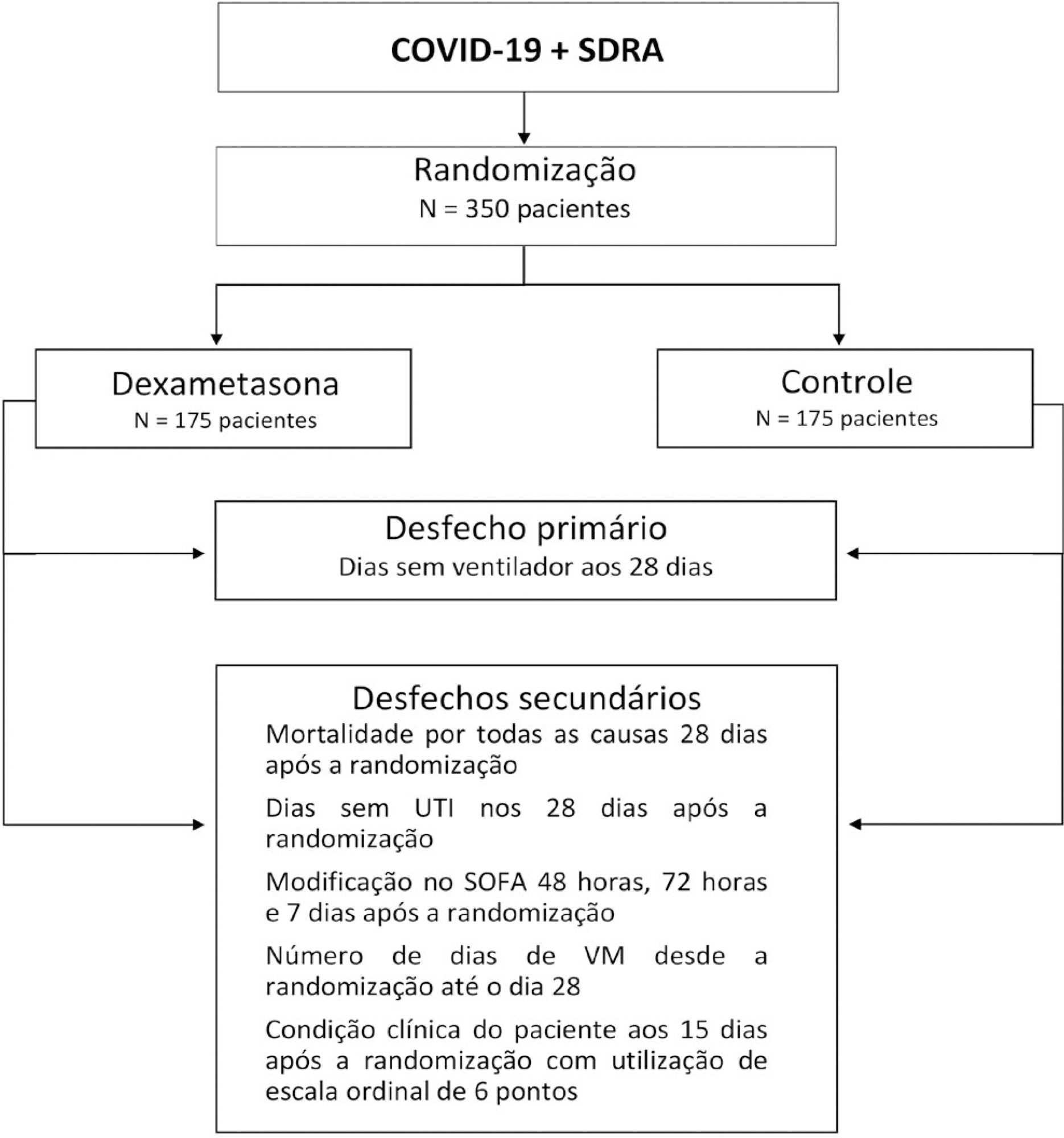
-
Original Article
Rationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
Abstract
Original ArticleRationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Views0See moreABSTRACT
Introduction:
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
Methods and analysis:
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
Ethics and dissemination:
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

-
Review Articles
Antithrombotics in acute coronary syndromes: actual guidelines and new evidences
Rev Bras Ter Intensiva. 2008;20(2):165-172
Abstract
Review ArticlesAntithrombotics in acute coronary syndromes: actual guidelines and new evidences
Rev Bras Ter Intensiva. 2008;20(2):165-172
DOI 10.1590/S0103-507X2008000200009
Views0See moreBACKGROUND AND OBJECTIVES: Acute coronary syndromes (ACS) are one of the most common causes of ICU admissions. New drugs have been developed for management of ACS. These drugs reduced morbidity and mortality; however their adverse effects or their incorrect use may cause excessive bleeding. The objective of this review is to present the principal peculiarities, doses, and indications of these drugs in ACS settings. METHODS: Original articles were retrieved crossing the terms acute coronary syndromes and antithrombotic therapy in the MedLine database as well as search for Brazilian and international guidelines in http://sumsearch.uthscsa.edu. RESULTS: In the treatment of acute coronary syndromes with non-ST-segment elevation enoxaparin was as efficient as UFH, but with a simpler management (SYNERGY and A to Z studies). In this same setting, fondaparinux was non inferior to enoxaparin and had lesser bleedings (OASIS 5), bivalirudin, combined or not with GPIIbIIIa blockers, was not inferior when compared with other heparins (ACUITY). In ST-segment elevation ACS, enoxaparin was superior to HNF in patients treated with fibrinolysis (EXTRACT TIMI 25); in OASIS 6 fondaparinux was superior to UFH in patients treated with thrombolytic therapy and not submitted to reperfusion. CONCLUSIONS: The correct management and individual combination of antithrombotic drugs are mandatory for decreased mortality and of major cardiovascular events, reducing the undesirable risk of additional bleeding.
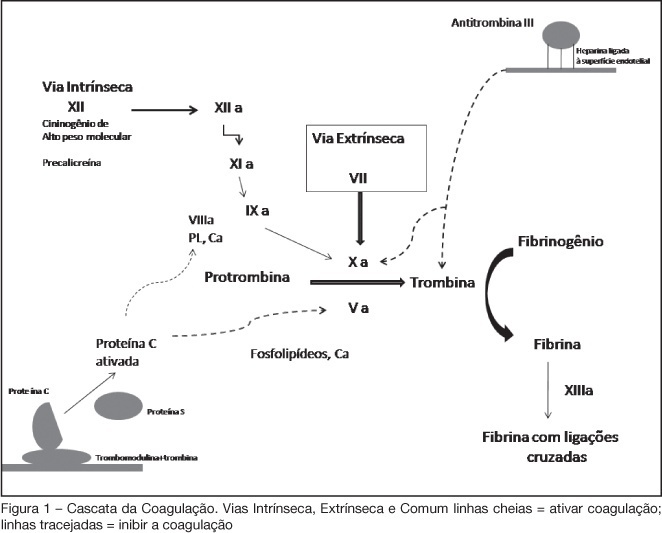
-
Artigos de Revisão
Statins on sepsis: a new therapeutic gun in intensive care medicine?
Rev Bras Ter Intensiva. 2006;18(4):402-406
Abstract
Artigos de RevisãoStatins on sepsis: a new therapeutic gun in intensive care medicine?
Rev Bras Ter Intensiva. 2006;18(4):402-406
DOI 10.1590/S0103-507X2006000400014
Views0See moreBACKGROUND AND OBJECTIVES: Sepsis is a syndrome with an important inflammatory component in its pathophysiology. Some drugs that act on the activation of inflammatory cascade have been tested in septic patients. Statins, beyond hypolipemic effects, have anti-inflammatory capacities, known as pleiotropic effects. That action may be of value on sepsis treatment. Review of publications who discuss the use of statins. This article has the objective to review the mainly papers about statins and sepses. METHODS: Original published articles were searched using Medline database crossing the keys words “sepsis and statins” between 1990 and 2006 RESULTS: Experimental and retrospective studies has been investigated the use of statins in septic patients, until this moment, most of then show benefits on morbidity and mortality. However, there is not prospective, randomized, placebo controlled trials. Or metanalyses, witch denotes lack strong and robust that could be indicated this action. So, since the benefit is still unable, it is necessary randomized clinical trials witch can prove this theory. CONCLUSIONS: Several experimental and retrospective studies have investigated the use of statins in septic patients, but despite literature demonstrating a promising role for its use in these patients, these studies are experimental or retrospective. Therefore, we must wait larger, prospective, randomized trials before we may be able to understand its role and possibly recommend these drugs in the treatment of septic patients.
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis