You searched for:"Alexandre Biasi Cavalcanti"
We found (31) results for your search.-
Original Articles
Statistical analysis plan for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART). A randomized controlled trial
Rev Bras Ter Intensiva. 2017;29(2):142-153
Abstract
Original ArticlesStatistical analysis plan for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART). A randomized controlled trial
Rev Bras Ter Intensiva. 2017;29(2):142-153
DOI 10.5935/0103-507X.20170024
Views0See moreABSTRACT
Background:
The Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) is an international multicenter randomized pragmatic controlled trial with allocation concealment involving 120 intensive care units in Brazil, Argentina, Colombia, Italy, Poland, Portugal, Malaysia, Spain, and Uruguay. The primary objective of ART is to determine whether maximum stepwise alveolar recruitment associated with PEEP titration, adjusted according to the static compliance of the respiratory system (ART strategy), is able to increase 28-day survival in patients with acute respiratory distress syndrome compared to conventional treatment (ARDSNet strategy).
Objective:
To describe the data management process and statistical analysis plan.
Methods:
The statistical analysis plan was designed by the trial executive committee and reviewed and approved by the trial steering committee. We provide an overview of the trial design with a special focus on describing the primary (28-day survival) and secondary outcomes. We describe our data management process, data monitoring committee, interim analyses, and sample size calculation. We describe our planned statistical analyses for primary and secondary outcomes as well as pre-specified subgroup analyses. We also provide details for presenting results, including mock tables for baseline characteristics, adherence to the protocol and effect on clinical outcomes.
Conclusion:
According to best trial practice, we report our statistical analysis plan and data management plan prior to locking the database and beginning analyses. We anticipate that this document will prevent analysis bias and enhance the utility of the reported results.
Trial registration:
ClinicalTrials.gov number, NCT01374022.
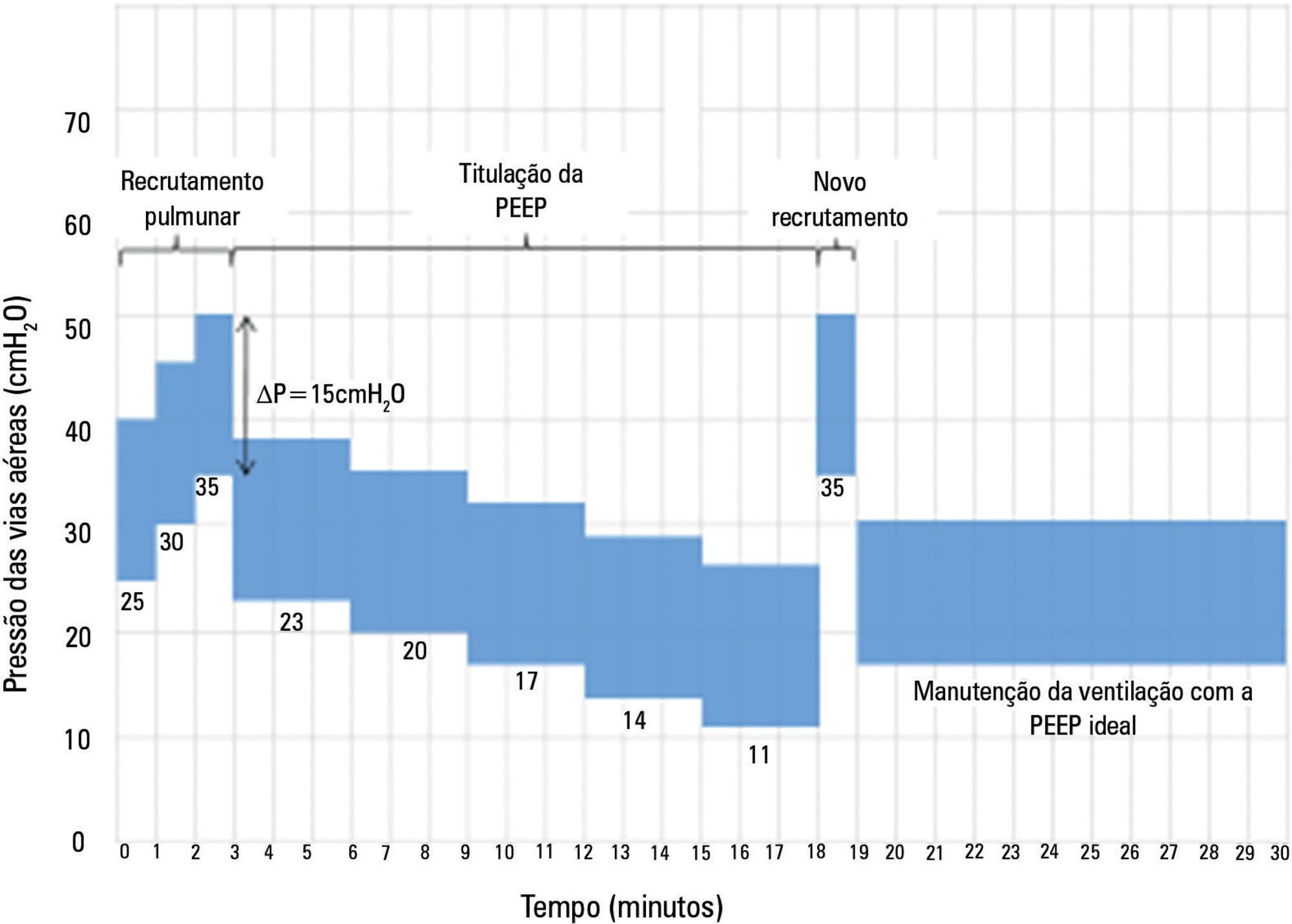
-
Commentaries
Getting a consensus: advantages and disadvantages of Sepsis 3 in the context of middle-income settings
Rev Bras Ter Intensiva. 2016;28(4):361-365
Abstract
CommentariesGetting a consensus: advantages and disadvantages of Sepsis 3 in the context of middle-income settings
Rev Bras Ter Intensiva. 2016;28(4):361-365
DOI 10.5935/0103-507X.20160068
Views0What is new on the Sepsis 3 definitions?Recently the Society of Critical Care Medicine (SCCM) and the European Society of Critical Care Medicine (ESICM) promoted a new consensus conference and published the new sepsis definitions, known as Sepsis 3.()Briefly, the broad definition of sepsis is now “a life-threatening organ dysfunction caused by dysregulated host response […]See more -
Editorials
Brazilian guidelines in critical care: let’s face this challenge…
Rev Bras Ter Intensiva. 2016;28(3):213-214
Abstract
EditorialsBrazilian guidelines in critical care: let’s face this challenge…
Rev Bras Ter Intensiva. 2016;28(3):213-214
DOI 10.5935/0103-507X.20160046
Views0Clinical practice guidelines are useful tools to improve delivery of the best care, based on the best available evidence, for our patients. They help practitioners to make clinical decisions and might help to ensure the proper allocation of resources in public health policy.() Therefore, health care guidelines and their appropriate implementation are of interest to […]See more -
Review Articles
Balanced crystalloids for septic shock resuscitation
Rev Bras Ter Intensiva. 2016;28(4):463-471
Abstract
Review ArticlesBalanced crystalloids for septic shock resuscitation
Rev Bras Ter Intensiva. 2016;28(4):463-471
DOI 10.5935/0103-507X.20160079
Views0ABSTRACT
Timely fluid administration is crucial to maintain tissue perfusion in septic shock patients. However, the question concerning which fluid should be used for septic shock resuscitation remains a matter of debate. A growing body of evidence suggests that the type, amount and timing of fluid administration during the course of sepsis may affect patient outcomes. Crystalloids have been recommended as the first-line fluids for septic shock resuscitation. Nevertheless, given the inconclusive nature of the available literature, no definitive recommendations about the most appropriate crystalloid solution can be made. Resuscitation of septic and non-septic critically ill patients with unbalanced crystalloids, mainly 0.9% saline, has been associated with a higher incidence of acid-base balance and electrolyte disorders and might be associated with a higher incidence of acute kidney injury. This can result in greater demand for renal replacement therapy and increased mortality. Balanced crystalloids have been proposed as an alternative to unbalanced solutions in order to mitigate their detrimental effects. Nevertheless, the safety and effectiveness of balanced crystalloids for septic shock resuscitation need to be further addressed in a well-designed, multicenter, pragmatic, randomized controlled trial.
Keywords:Critical care/methodsCritical care/trendsFluid therapy/methodsIsotonic solutions/administration & dosageRehydration solutions/administration & dosageResuscitation/methodsShock, septicSee more -
Special Article
Brazilian recommendations of mechanical ventilation 2013. Part I
Rev Bras Ter Intensiva. 2014;26(2):89-121
Abstract
Special ArticleBrazilian recommendations of mechanical ventilation 2013. Part I
Rev Bras Ter Intensiva. 2014;26(2):89-121
DOI 10.5935/0103-507X.20140017
Views6See morePerspectives on invasive and noninvasive ventilatory support for critically ill patients are evolving, as much evidence indicates that ventilation may have positive effects on patient survival and the quality of the care provided in intensive care units in Brazil. For those reasons, the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira – AMIB) and the Brazilian Thoracic Society (Sociedade Brasileira de Pneumologia e Tisiologia – SBPT), represented by the Mechanical Ventilation Committee and the Commission of Intensive Therapy, respectively, decided to review the literature and draft recommendations for mechanical ventilation with the goal of creating a document for bedside guidance as to the best practices on mechanical ventilation available to their members. The document was based on the available evidence regarding 29 subtopics selected as the most relevant for the subject of interest. The project was developed in several stages, during which the selected topics were distributed among experts recommended by both societies with recent publications on the subject of interest and/or significant teaching and research activity in the field of mechanical ventilation in Brazil. The experts were divided into pairs that were charged with performing a thorough review of the international literature on each topic. All the experts met at the Forum on Mechanical Ventilation, which was held at the headquarters of AMIB in São Paulo on August 3 and 4, 2013, to collaboratively draft the final text corresponding to each sub-topic, which was presented to, appraised, discussed and approved in a plenary session that included all 58 participants and aimed to create the final document.
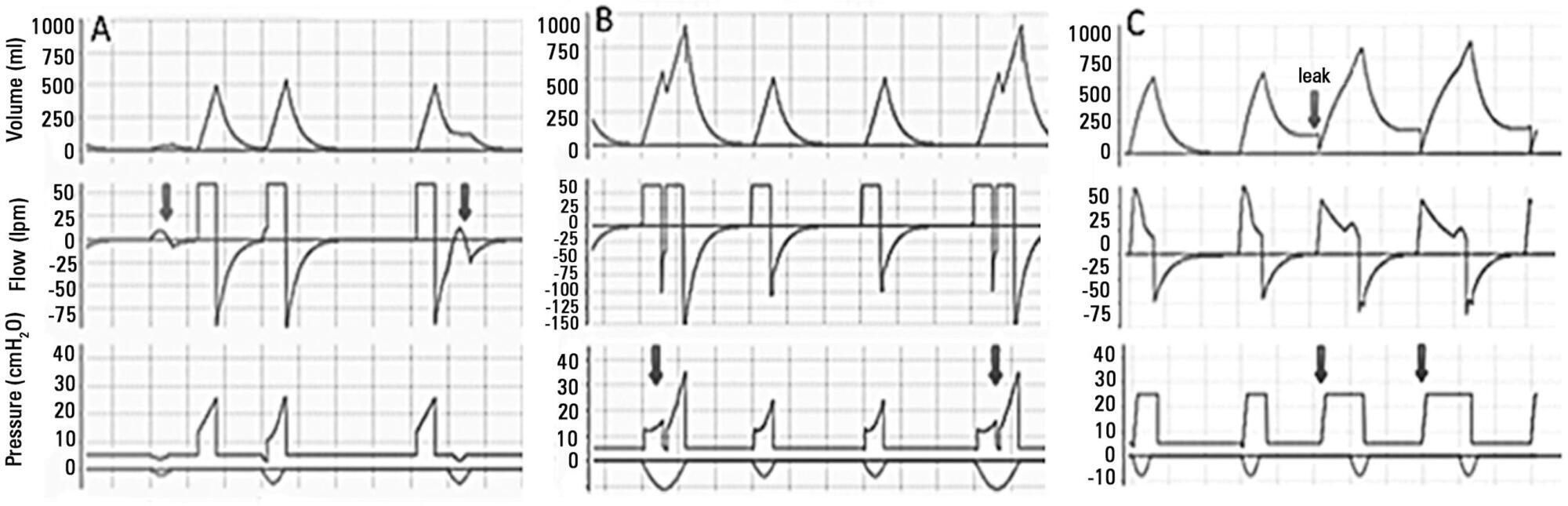
-
Special Article
Brazilian recommendations of mechanical ventilation 2013. Part 2
Rev Bras Ter Intensiva. 2014;26(3):215-239
Abstract
Special ArticleBrazilian recommendations of mechanical ventilation 2013. Part 2
Rev Bras Ter Intensiva. 2014;26(3):215-239
DOI 10.5935/0103-507X.20140034
Views2See morePerspectives on invasive and noninvasive ventilatory support for critically ill patients are evolving, as much evidence indicates that ventilation may have positive effects on patient survival and the quality of the care provided in intensive care units in Brazil. For those reasons, the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira – AMIB) and the Brazilian Thoracic Society (Sociedade Brasileira de Pneumologia e Tisiologia – SBPT), represented by the Mechanical Ventilation Committee and the Commission of Intensive Therapy, respectively, decided to review the literature and draft recommendations for mechanical ventilation with the goal of creating a document for bedside guidance as to the best practices on mechanical ventilation available to their members. The document was based on the available evidence regarding 29 subtopics selected as the most relevant for the subject of interest. The project was developed in several stages, during which the selected topics were distributed among experts recommended by both societies with recent publications on the subject of interest and/or significant teaching and research activity in the field of mechanical ventilation in Brazil. The experts were divided into pairs that were charged with performing a thorough review of the international literature on each topic. All the experts met at the Forum on Mechanical Ventilation, which was held at the headquarters of AMIB in São Paulo on August 3 and 4, 2013, to collaboratively draft the final text corresponding to each sub-topic, which was presented to, appraised, discussed and approved in a plenary session that included all 58 participants and aimed to create the final document.
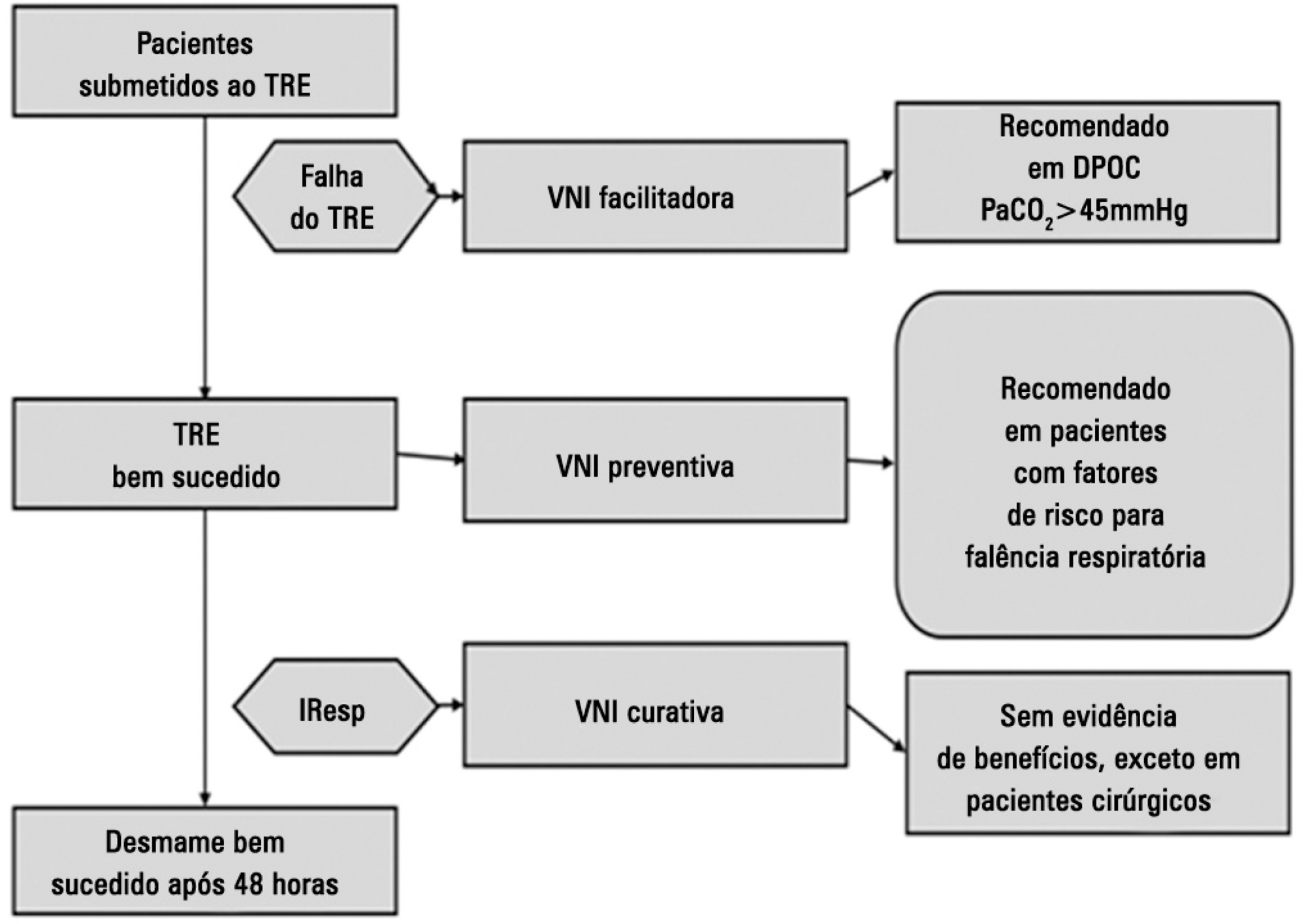
-
How to assess intensive care randomized trials
Rev Bras Ter Intensiva. 2009;21(2):219-225
Abstract
How to assess intensive care randomized trials
Rev Bras Ter Intensiva. 2009;21(2):219-225
DOI 10.1590/S0103-507X2009000200016
Views0See moreRandomized controlled trials are scientific investigations considered as the gold-standard to evaluate therapeutic interventions. Randomized controlled trials may examine the safety and efficacy of new drugs and therapeutic procedures or compare the effects of two or more drugs or any other intervention. In this article, we present the essential features of these studies, as well as, factors which may bias randomized controlled trials. We also present criteria to critically appraise articles reporting randomized controlled trials, explain how to interpret the results and how to apply them to clinical practice.

Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




