Abstract
Rev Bras Ter Intensiva. 2021;33(1):68-74
DOI 10.5935/0103-507X.20210006
To analyze whether changes in medical care due to the application of COVID-19 protocols affected clinical outcomes in patients without COVID-19 during the pandemic.
This was a retrospective, observational cohort study carried out in a thirty-eight-bed surgical and medical intensive care unit of a high complexity private hospital. Patients with respiratory failure admitted to the intensive care unit during March and April 2020 and the same months in 2019 were selected. We compared interventions and outcomes of patients without COVID-19 during the pandemic with patients admitted in 2019. The main variables analyzed were intensive care unit respiratory management, number of chest tomography scans and bronchoalveolar lavages, intensive care unit complications, and status at hospital discharge.
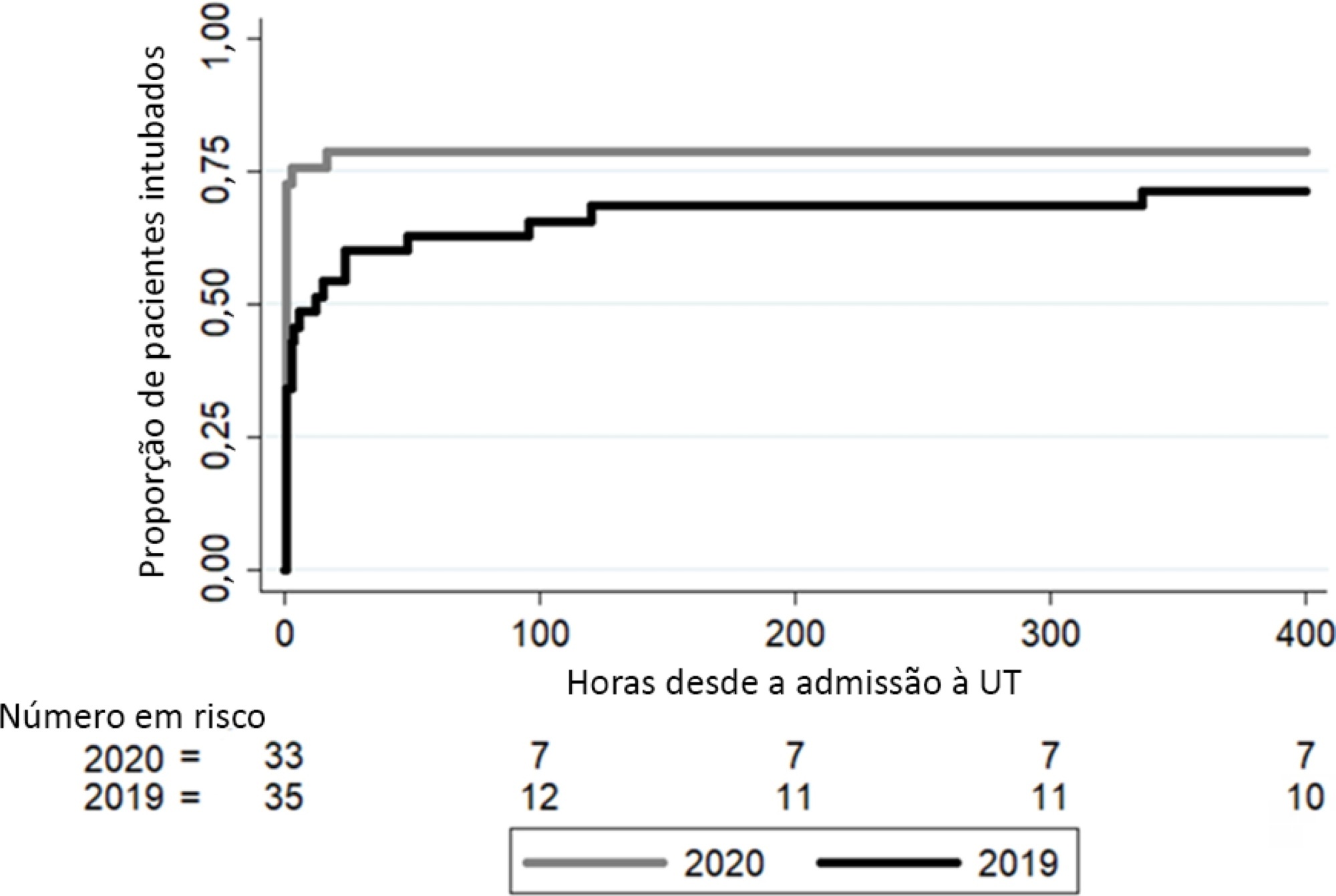
In 2020, a significant reduction in the use of a high-flow nasal cannula was observed: 14 (42%) in 2019 compared to 1 (3%) in 2020. Additionally, in 2020, a significant increase was observed in the number of patients under mechanical ventilation admitted to the intensive care unit from the emergency department, 23 (69%) compared to 11 (31%) in 2019. Nevertheless, the number of patients with mechanical ventilation after 5 days of admission was similar in both years: 24 (69%) in 2019 and 26 (79%) in 2020.
Intensive care unit protocols based on international recommendations for the COVID-19 pandemic have produced a change in non-COVID-19 patient management. We observed a reduction in the use of a high-flow nasal cannula and an increased number of tracheal intubations in the emergency department. However, no changes in the percentage of intubated patients in the intensive care unit, the number of mechanical ventilation days or the length of stay in intensive care unit.
Abstract
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
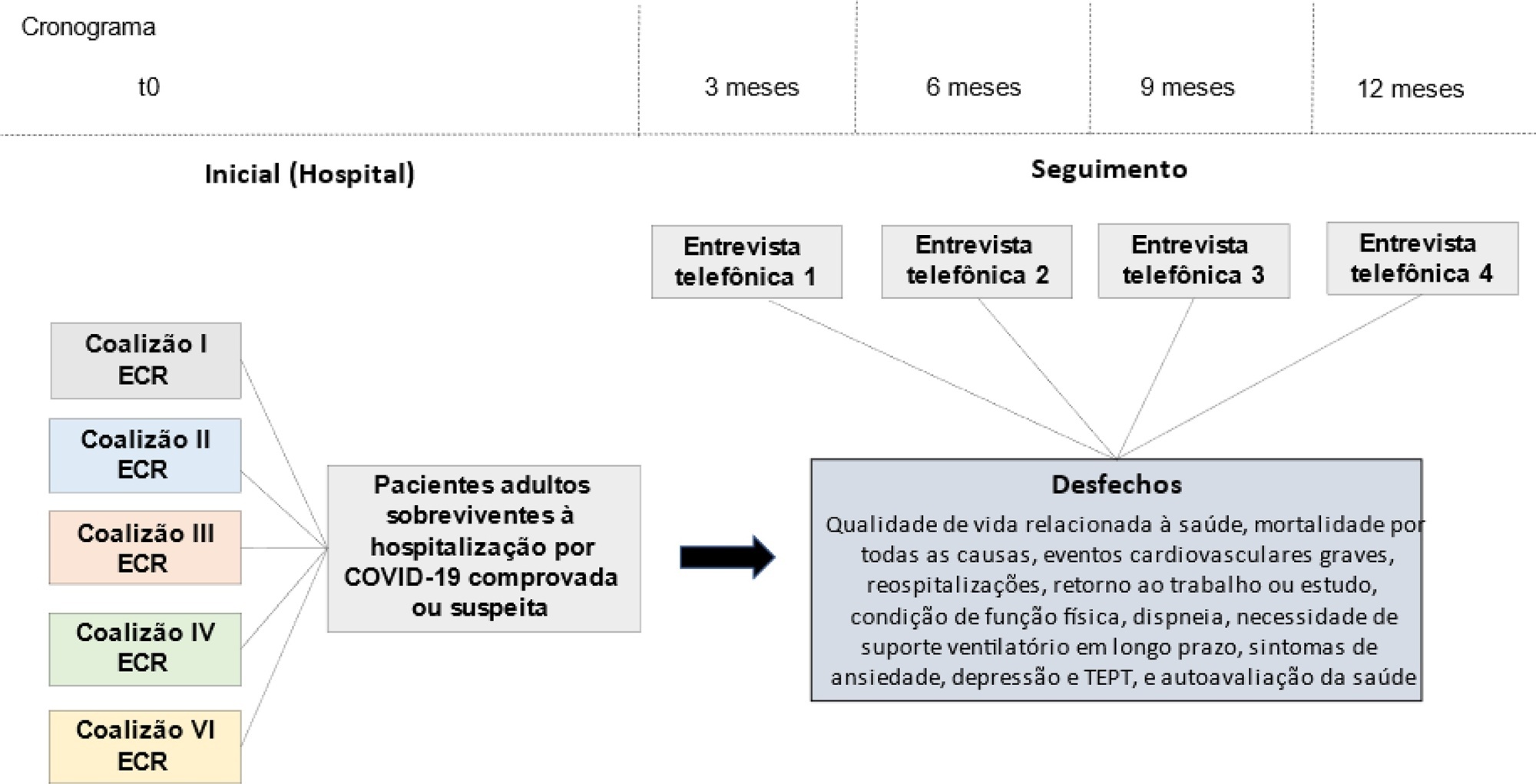
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
Abstract
Rev Bras Ter Intensiva. 2020;32(4):603-605
DOI 10.5935/0103-507X.20200098
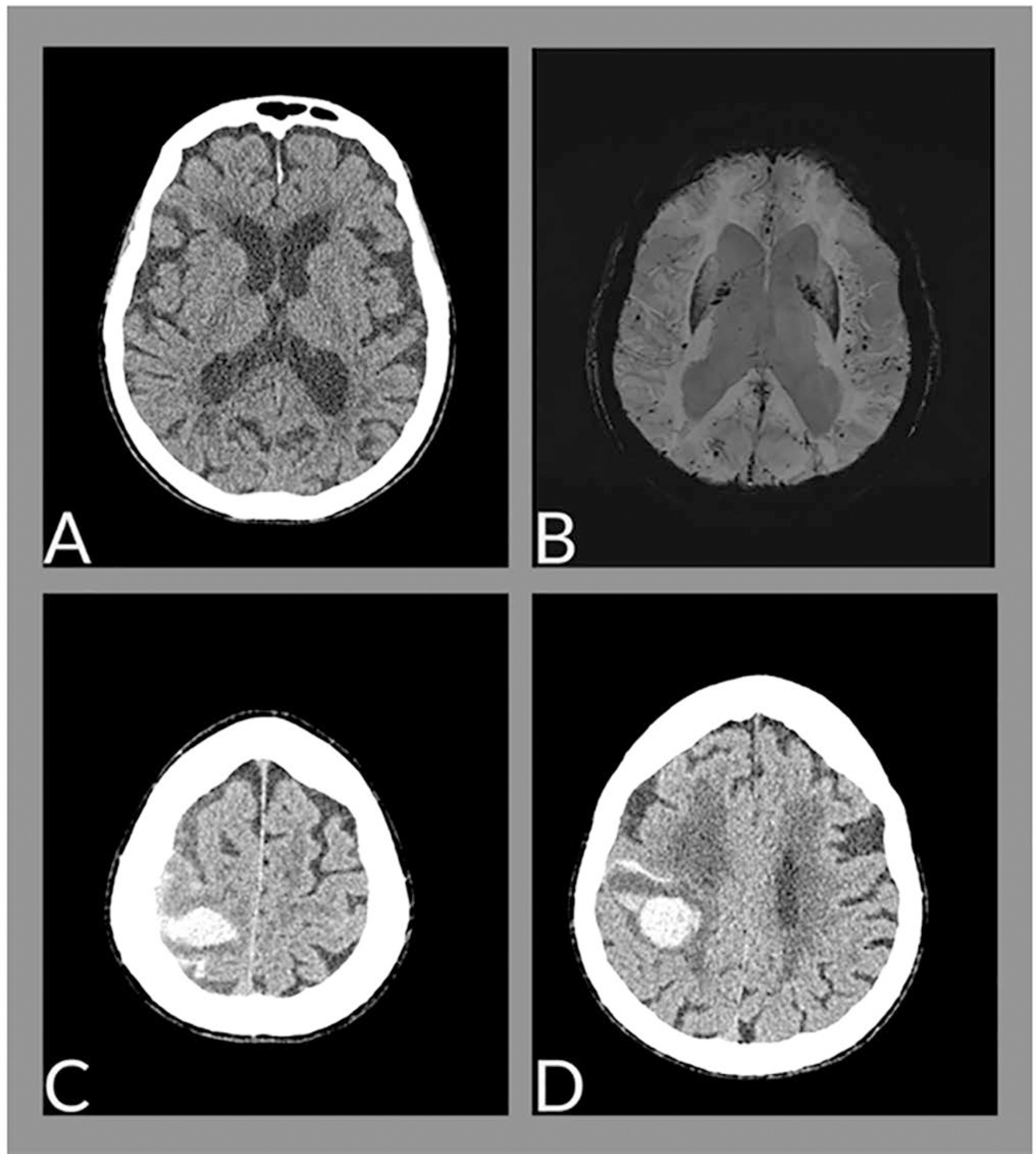
The neurological changes associated with COVID-19 have been frequently described, especially in cases of greater severity, and are related to multifactorial causes, such as endothelial dysfunction, inflammatory mediator release (cytokine storm), endothelial dysfunction and hypoxemia. We report the case of a female patient, 88 years old, with cerebral hemorrhage associated with amyloid angiopathy in the context of SARS-CoV-2 infection.
Abstract
Rev Bras Ter Intensiva. 2020;32(4):487-492
DOI 10.5935/0103-507X.20200082
This study aims to describe the clinical characteristics and predictors of mechanical ventilation of adult inpatients with COVID-19 in a single center.
A retrospective cohort study was performed and included adult inpatients hospitalized from March 17th to May 3rd, 2020, who were diagnosed with SARS-CoV-2 infection. Clinical and demographic characteristics were extracted from electronic medical records.
Overall, 88 consecutive patients were included in this study. The median age of the patients was 63 years (IQR 49 - 71); 59 (67%) were male, 65 (86%) had a college degree and 67 (76%) had at least one comorbidity. Twenty-nine (33%) patients were admitted to the intensive care unit, 18 (20%) patients needed mechanical ventilation, and 9 (10.2%) died during hospitalization. The median length of stay in the intensive care unit and the median duration of mechanical ventilation was 23 and 29.5 days, respectively. An age ≥ 65 years was an independent risk factor for mechanical ventilation (OR 8.4 95%CI 1.3 - 55.6 p = 0.02).
Our findings describe the first wave of Brazilian patients hospitalized for COVID-19. Age was the strongest predictor of respiratory insufficiency and the need for mechanical ventilation in our population.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

Abstract
Rev Bras Ter Intensiva. 2020;32(2):166-196
DOI 10.5935/0103-507X.20200039
Different therapies are currently used, considered, or proposed for the treatment of COVID-19; for many of those therapies, no appropriate assessment of effectiveness and safety was performed. This document aims to provide scientifically available evidence-based information in a transparent interpretation, to subsidize decisions related to the pharmacological therapy of COVID-19 in Brazil.
A group of 27 experts and methodologists integrated a task-force formed by professionals from the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB), the Brazilian Society of Infectious Diseases (Sociedad Brasileira de Infectologia - SBI) and the Brazilian Society of Pulmonology and Tisiology (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT). Rapid systematic reviews, updated on April 28, 2020, were conducted. The assessment of the quality of evidence and the development of recommendations followed the GRADE system. The recommendations were written on May 5, 8, and 13, 2020.
Eleven recommendations were issued based on low or very-low level evidence. We do not recommend the routine use of hydroxychloroquine, chloroquine, azithromycin, lopinavir/ritonavir, corticosteroids, or tocilizumab for the treatment of COVID-19. Prophylactic heparin should be used in hospitalized patients, however, no anticoagulation should be provided for patients without a specific clinical indication. Antibiotics and oseltamivir should only be considered for patients with suspected bacterial or influenza coinfection, respectively.
So far no pharmacological intervention was proven effective and safe to warrant its use in the routine treatment of COVID-19 patients; therefore such patients should ideally be treated in the context of clinical trials. The recommendations herein provided will be revised continuously aiming to capture newly generated evidence.