Abstract
Rev Bras Ter Intensiva. 2020;32(4):487-492
DOI 10.5935/0103-507X.20200082
This study aims to describe the clinical characteristics and predictors of mechanical ventilation of adult inpatients with COVID-19 in a single center.
A retrospective cohort study was performed and included adult inpatients hospitalized from March 17th to May 3rd, 2020, who were diagnosed with SARS-CoV-2 infection. Clinical and demographic characteristics were extracted from electronic medical records.
Overall, 88 consecutive patients were included in this study. The median age of the patients was 63 years (IQR 49 - 71); 59 (67%) were male, 65 (86%) had a college degree and 67 (76%) had at least one comorbidity. Twenty-nine (33%) patients were admitted to the intensive care unit, 18 (20%) patients needed mechanical ventilation, and 9 (10.2%) died during hospitalization. The median length of stay in the intensive care unit and the median duration of mechanical ventilation was 23 and 29.5 days, respectively. An age ≥ 65 years was an independent risk factor for mechanical ventilation (OR 8.4 95%CI 1.3 - 55.6 p = 0.02).
Our findings describe the first wave of Brazilian patients hospitalized for COVID-19. Age was the strongest predictor of respiratory insufficiency and the need for mechanical ventilation in our population.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):354-362
DOI 10.5935/0103-507X.20200063
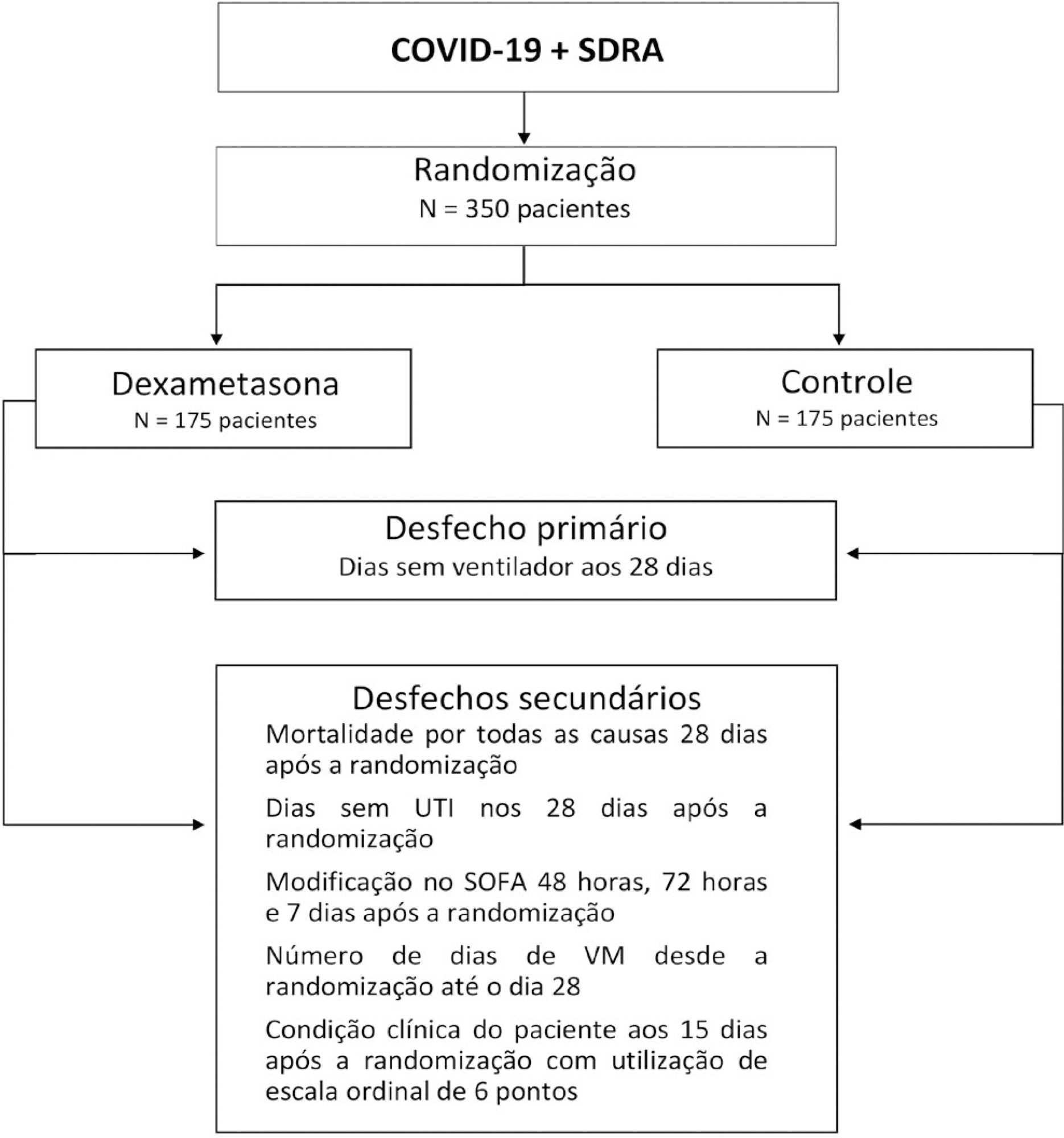
The infection caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spreads worldwide and is considered a pandemic. The most common manifestation of SARS-CoV-2 infection (coronavirus disease 2019 - COVID-19) is viral pneumonia with varying degrees of respiratory compromise and up to 40% of hospitalized patients might develop acute respiratory distress syndrome. Several clinical trials evaluated the role of corticosteroids in non-COVID-19 acute respiratory distress syndrome with conflicting results. We designed a trial to evaluate the effectiveness of early intravenous dexamethasone administration on the number of days alive and free of mechanical ventilation within 28 days after randomization in adult patients with moderate or severe acute respiratory distress syndrome due to confirmed or probable COVID-19.
This is a pragmatic, prospective, randomized, stratified, multicenter, open-label, controlled trial including 350 patients with early-onset (less than 48 hours before randomization) moderate or severe acute respiratory distress syndrome, defined by the Berlin criteria, due to COVID-19. Eligible patients will be randomly allocated to either standard treatment plus dexamethasone (Intervention Group) or standard treatment without dexamethasone (Control Group). Patients in the intervention group will receive dexamethasone 20mg intravenous once daily for 5 days, followed by dexamethasone 10mg IV once daily for additional 5 days or until intensive care unit discharge, whichever occurs first. The primary outcome is ventilator-free days within 28 days after randomization, defined as days alive and free from invasive mechanical ventilation. Secondary outcomes are all-cause mortality rates at day 28, evaluation of the clinical status at day 15 assessed with a 6-level ordinal scale, mechanical ventilation duration from randomization to day 28, Sequential Organ Failure Assessment Score evaluation at 48 hours, 72 hours and 7 days and intensive care unit -free days within 28.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):348-353
DOI 10.5935/0103-507X.20200062
A novel coronavirus emerged this year as a cause of viral pneumonia. The main characteristics of the virus are rapid transmission, high contagion capacity and potential severity. The objective of this case series study is to describe the clinical characteristics of patients with confirmed coronavirus disease (COVID-19) admitted to different intensive care units in Argentina for mechanical ventilation.
A descriptive, prospective, multicenter case series study was conducted between April 1 and May 8, 2020. Data from patients older than 18 years who were admitted to the intensive care unit for mechanical ventilation for acute respiratory failure with a positive diagnosis of COVID-19 were included.
The variables for 47 patients from 31 intensive care units were recorded: 78.7% were men (median age of 61 years), with a SAPS II score of 43 and a Charlson index score of 3. The initial ventilatory mode was volume control - continuous mandatory ventilation with a tidal volume less than 8mL/kg in 100% of cases, with a median positive end-expiratory pressure of 10.5cmH2O. At the end of the study, 29 patients died, 8 were discharged, and 10 remained hospitalized. The SAPS II score was higher among patients who died (p = 0.046). Charlson comorbidity index was associated with higher mortality (OR = 2.27, 95% CI 1.13 - 4.55, p = 0.02).
Patients with COVID-19 and on mechanical ventilation in this series presented clinical variables similar to those described to date in other international reports. Our findings provide data that may predict outcomes.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):444-457
DOI 10.5935/0103-507X.20200075
The COVID-19 pandemic has brought concerns to managers, healthcare professionals, and the general population related to the potential mechanical ventilators’ shortage for severely ill patients. In Brazil, there are several initiatives aimed at producing alternative ventilators to cover this gap. To assist the teams that work in these initiatives, we provide a discussion of some basic concepts on physiology and respiratory mechanics, commonly used mechanical ventilation terms, the differences between triggering and cycling, the basic ventilation modes and other relevant aspects, such as mechanisms of ventilator-induced lung injury, respiratory drive, airway heating and humidification, cross-contamination risks, and aerosol dissemination. After the prototype development phase, preclinical bench-tests and animal model trials are needed to determine the safety and performance of the ventilator, following the minimum technical requirements. Next, it is mandatory going through the regulatory procedures as required by the Brazilian Health Regulatory Agency (Agência Nacional de Vigilância Sanitária - ANVISA). The manufacturing company should be appropriately registered by ANVISA, which also must be notified about the conduction of clinical trials, following the research protocol approval by the Research Ethics Committee. The registration requisition of the ventilator with ANVISA should include a dossier containing the information described in this paper, which is not intended to cover all related matters but to provide guidance on the required procedures.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

Abstract
Rev Bras Ter Intensiva. 2020;32(2):166-196
DOI 10.5935/0103-507X.20200039
Different therapies are currently used, considered, or proposed for the treatment of COVID-19; for many of those therapies, no appropriate assessment of effectiveness and safety was performed. This document aims to provide scientifically available evidence-based information in a transparent interpretation, to subsidize decisions related to the pharmacological therapy of COVID-19 in Brazil.
A group of 27 experts and methodologists integrated a task-force formed by professionals from the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB), the Brazilian Society of Infectious Diseases (Sociedad Brasileira de Infectologia - SBI) and the Brazilian Society of Pulmonology and Tisiology (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT). Rapid systematic reviews, updated on April 28, 2020, were conducted. The assessment of the quality of evidence and the development of recommendations followed the GRADE system. The recommendations were written on May 5, 8, and 13, 2020.
Eleven recommendations were issued based on low or very-low level evidence. We do not recommend the routine use of hydroxychloroquine, chloroquine, azithromycin, lopinavir/ritonavir, corticosteroids, or tocilizumab for the treatment of COVID-19. Prophylactic heparin should be used in hospitalized patients, however, no anticoagulation should be provided for patients without a specific clinical indication. Antibiotics and oseltamivir should only be considered for patients with suspected bacterial or influenza coinfection, respectively.
So far no pharmacological intervention was proven effective and safe to warrant its use in the routine treatment of COVID-19 patients; therefore such patients should ideally be treated in the context of clinical trials. The recommendations herein provided will be revised continuously aiming to capture newly generated evidence.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):224-228
DOI 10.5935/0103-507X.20200030
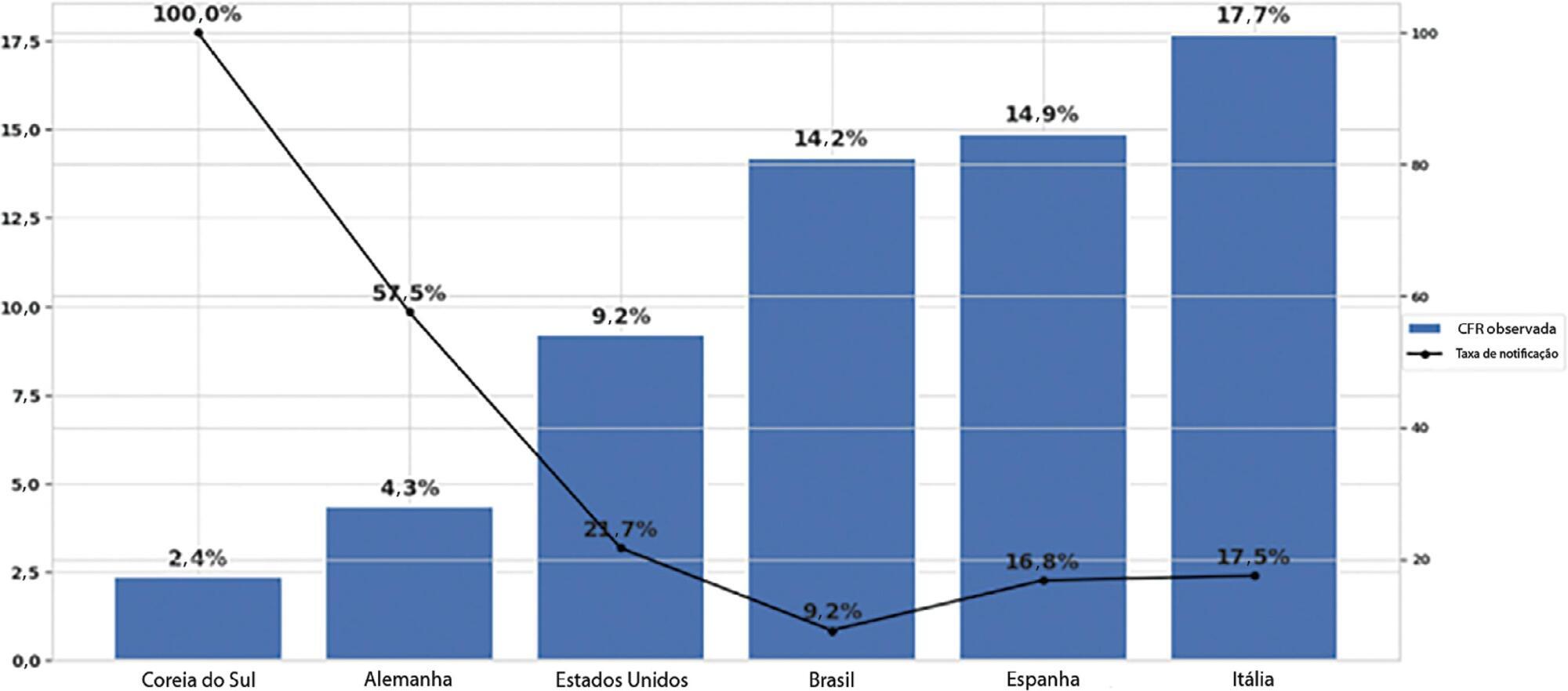
To estimate the reporting rates of coronavirus disease 2019 (COVID-19) cases for Brazil as a whole and states.
We estimated the actual number of COVID-19 cases using the reported number of deaths in Brazil and each state, and the expected case-fatality ratio from the World Health Organization. Brazil’s expected case-fatality ratio was also adjusted by the population’s age pyramid. Therefore, the notification rate can be defined as the number of confirmed cases (notified by the Ministry of Health) divided by the number of expected cases (estimated from the number of deaths).
The reporting rate for COVID-19 in Brazil was estimated at 9.2% (95%CI 8.8% - 9.5%), with all the states presenting rates below 30%. São Paulo and Rio de Janeiro, the most populated states in Brazil, showed small reporting rates (8.9% and 7.2%, respectively). The highest reporting rate occurred in Roraima (31.7%) and the lowest in Paraiba (3.4%).
The results indicated that the reporting of confirmed cases in Brazil is much lower as compared to other countries we analyzed. Therefore, decision-makers, including the government, fail to know the actual dimension of the pandemic, which may interfere with the determination of control measures.
Abstract
Rev Bras Ter Intensiva. 2020;32(2):213-223
DOI 10.5935/0103-507X.20200028
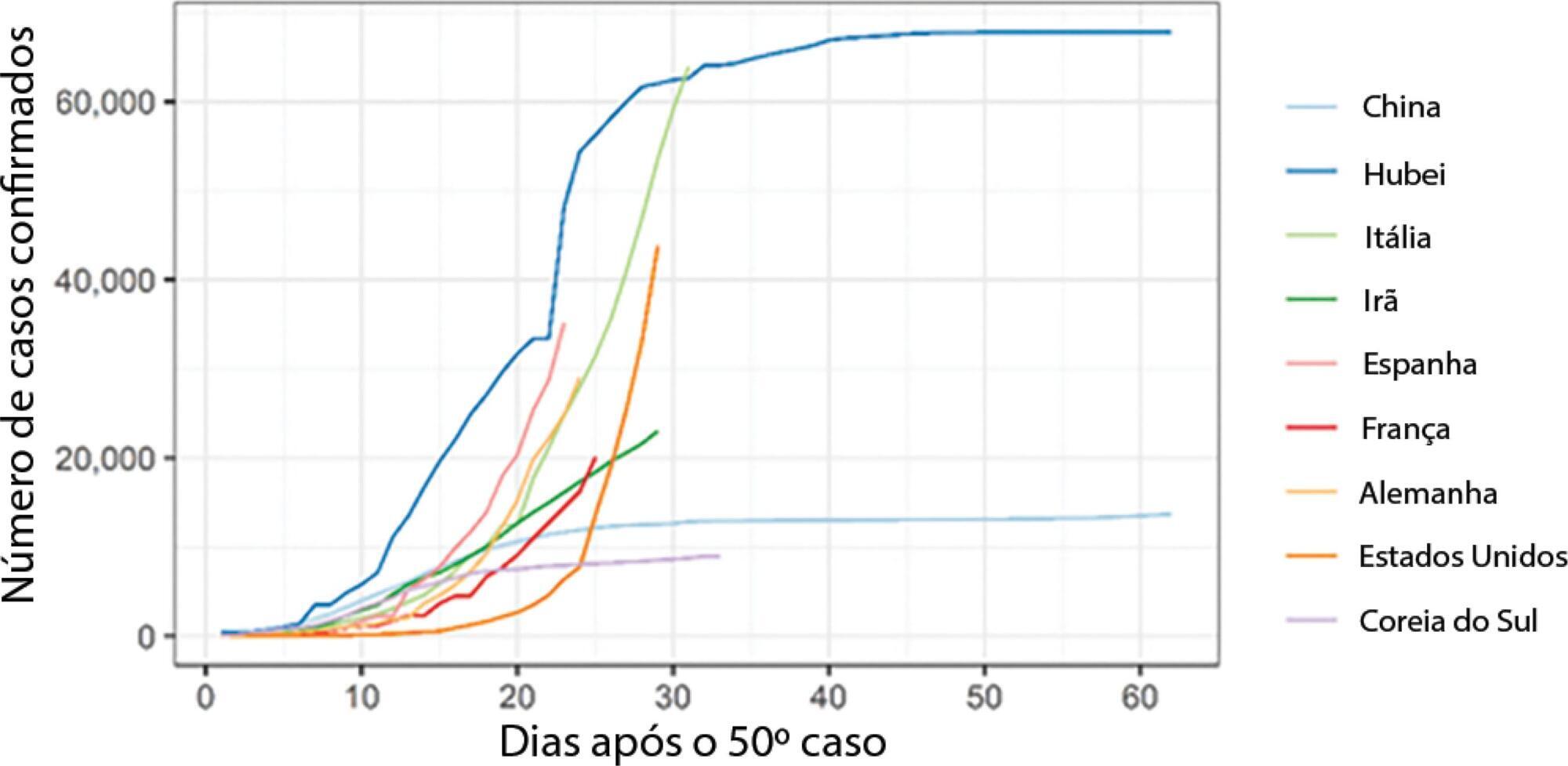
To analyse the measures adopted by countries that have shown control over the transmission of coronavirus disease 2019 (COVID-19) and how each curve of accumulated cases behaved after the implementation of those measures.
The methodology adopted for this study comprises three phases: systemizing control measures adopted by different countries, identifying structural breaks in the growth of the number of cases for those countries, and analyzing Brazilian data in particular.
We noted that China (excluding Hubei Province), Hubei Province, and South Korea have been effective in their deceleration of the growth rates of COVID-19 cases. The effectiveness of the measures taken by these countries could be seen after 1 to 2 weeks of their application. In Italy and Spain, control measures at the national level were taken at a late stage of the epidemic, which could have contributed to the high propagation of COVID-19. In Brazil, Rio de Janeiro and São Paulo adopted measures that could be effective in slowing the propagation of the virus. However, we only expect to see their effects on the growth of the curve in the coming days.
Our results may help decisionmakers in countries in relatively early stages of the epidemic, especially Brazil, understand the importance of control measures in decelerating the growth curve of confirmed cases.