You searched for:"Flávia Ribeiro Machado"
We found (44) results for your search.Abstract
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
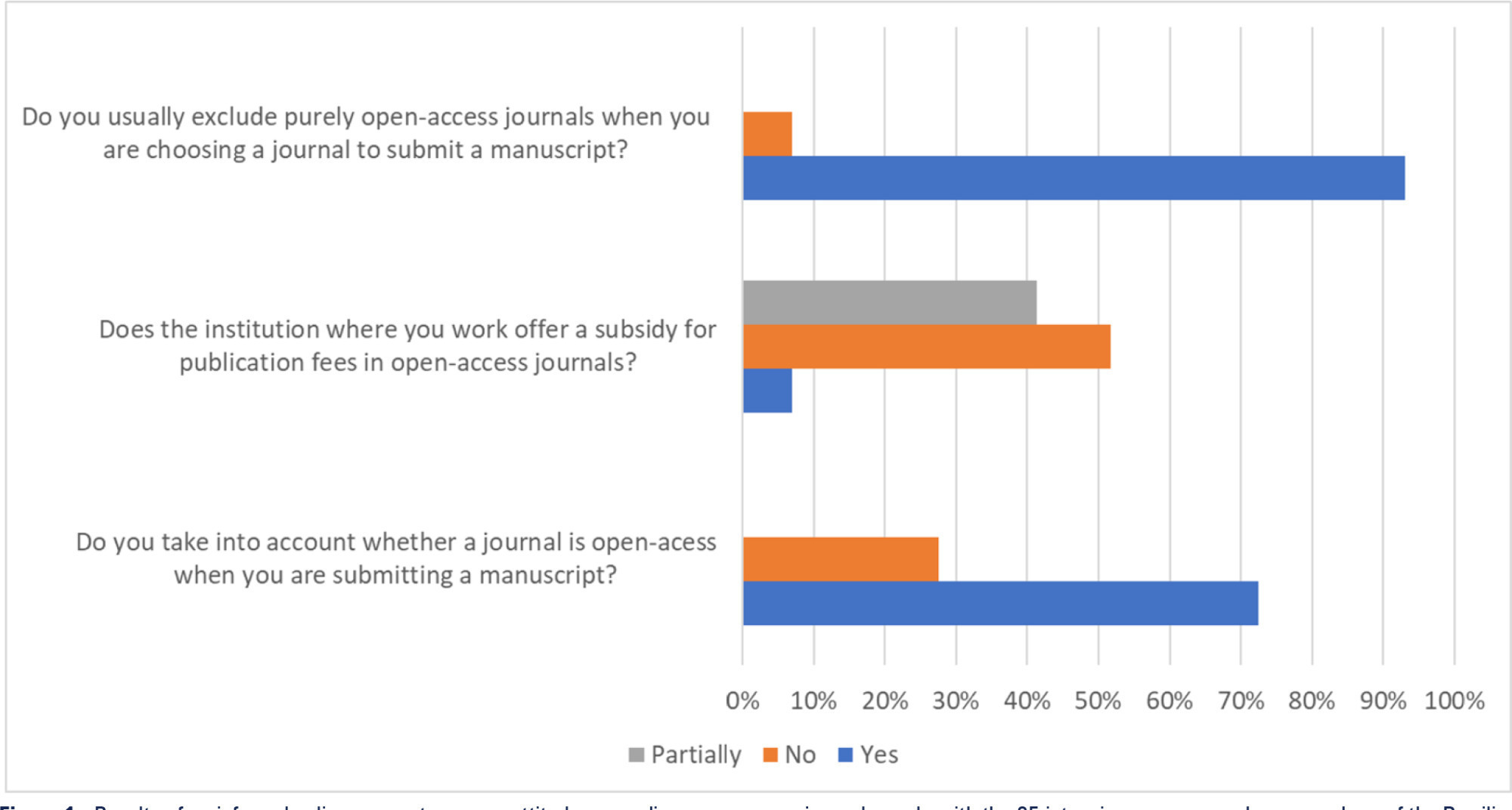
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
Abstract
Rev Bras Ter Intensiva. 2009;21(3):324-331
DOI 10.1590/S0103-507X2009000300014
Constipation is a common complication identified among critically ill patients. Its incidence is highly variable due to lack of definition of such patients. Besides the already known consequences of constipation, in recent years it was observed that this complication may also be related to worse prognosis of critically ill patients. This review endeavors to describe the main available scientific evidence showing that constipation is a prognostic marker and a clinical representation of intestinal dysfunction, in addition to eventually interfering in the prognosis with treatment. Ogilvie syndrome, a major cause of morbidity and mortality in intensive care units was also reviewed. Considering the above cases it was concluded that more attention to this disorder is required in intensive care units as well as development of protocols for diagnosis and management of critically ill patients.

Abstract
Rev Bras Ter Intensiva. 2010;22(4):327-332
DOI 10.1590/S0103-507X2010000400003
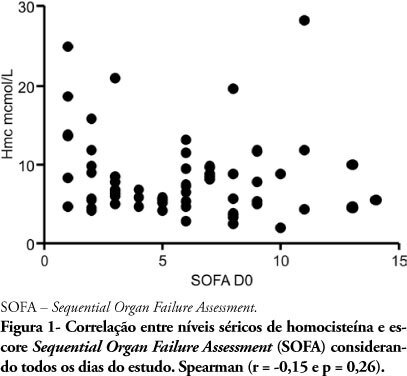
OBJECTIVE: Homocysteine and sepsis are both associated with inflammation and endothelial activation. Therefore this study was aimed to evaluate if the plasma homocystein level is related with the septic patient clinical severity. METHODS: Severe sepsis or septic shock patients, with less than 48 hours from organ dysfunction start, were admitted to this prospective observational study. Homocysteine levels were determined by the time of study admission and then on the Days 3, 7 and 14. The homocysteine association with the Sequential Organ Failure Assessment (SOFA) score was evaluated using the Sperman test, and its association with mortality using the Mann-Whitney test. A p<0.05 value was considered statistically significant. RESULTS: Twenty one patients were enrolled, and 60 blood samples were collected to measure total homocysteine [median 6.92 (5.27 - 9.74 μmol/L)]. The Sperman correlation test showed no association between homocysteine and SOFA ( r=0.15 and p=0.26). Also no correlation was found for the homocysteine level by the study admission time and the difference between the Day 3 SOFA score versus by study admission (deltaSOFA) (r=0.04 and p=0.87). Homocysteine variation between the Day 3 and the study admission (deltaHmc) and SOFA score variation in the same period were not correlated (r=-0.11 and p=0.66). Homocysteine by the study admission was not correlated with death in intensive care unit rate (p= 0.46) or in-hospital death rate (p = 0.13). This was also true for deltaHmc (p=0.12 and p=0.99, respectively). CONCLUSION: Baseline homocysteine levels and its variations within the first dysfunction days were not related with septic patients' worsened organ function parameters or mortality.
Abstract
Rev Bras Ter Intensiva. 2022;34(3):335-341
DOI 10.5935/0103-507X.20220040-en
To compare the lung mechanics and outcomes between COVID-19-associated acute respiratory distress syndrome and non-COVID-19-associated acute respiratory distress syndrome.
We combined data from two randomized trials in acute respiratory distress syndrome, one including only COVID-19 patients and the other including only patients without COVID-19, to determine whether COVID-19-associated acute respiratory distress syndrome is associated with higher 28-day mortality than non-COVID-19 acute respiratory distress syndrome and to examine the differences in lung mechanics between these two types of acute respiratory distress syndrome.
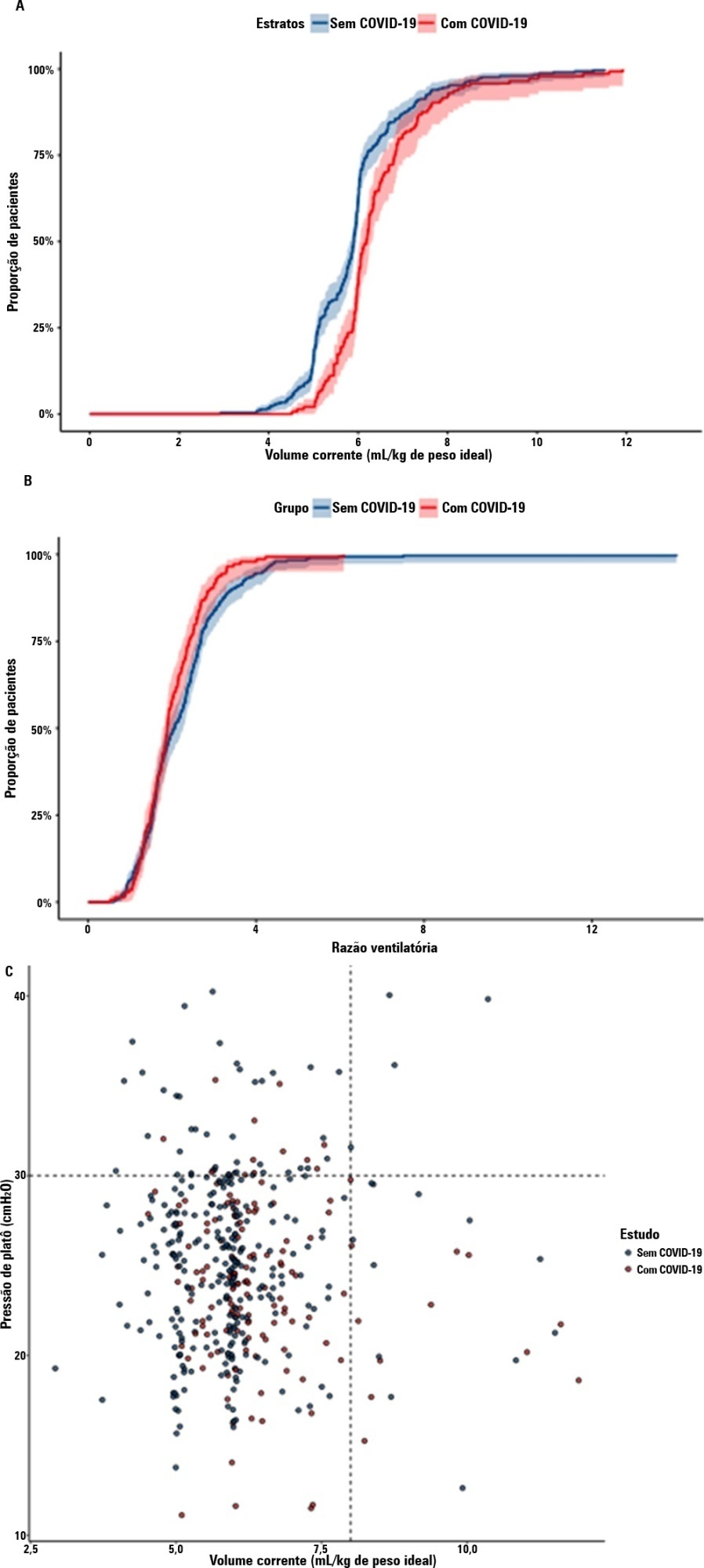
A total of 299 patients with COVID-19-associated acute respiratory distress syndrome and 1,010 patients with non-COVID-19-associated acute respiratory distress syndrome were included in the main analysis. The results showed that non-COVID-19 patients used higher positive end-expiratory pressure (12.5cmH2O; SD 3.2 versus 11.7cmH2O SD 2.8; p < 0.001), were ventilated with lower tidal volumes (5.8mL/kg; SD 1.0 versus 6.5mL/kg; SD 1.2; p < 0.001) and had lower static respiratory compliance adjusted for ideal body weight (0.5mL/cmH2O/kg; SD 0.3 versus 0.6mL/cmH2O/kg; SD 0.3; p = 0.01). There was no difference between groups in 28-day mortality (52.3% versus 58.9%; p = 0.52) or mechanical ventilation duration in the first 28 days among survivors (13 [IQR 5 - 22] versus 12 [IQR 6 - 26], p = 0.46).
This analysis showed that patients with non-COVID-19-associated acute respiratory distress syndrome have different lung mechanics but similar outcomes to COVID-19-associated acute respiratory distress syndrome patients. After propensity score matching, there was no difference in lung mechanics or outcomes between groups.
Abstract
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

Abstract
Rev Bras Ter Intensiva. 2015;27(4):340-346
DOI 10.5935/0103-507X.20150058
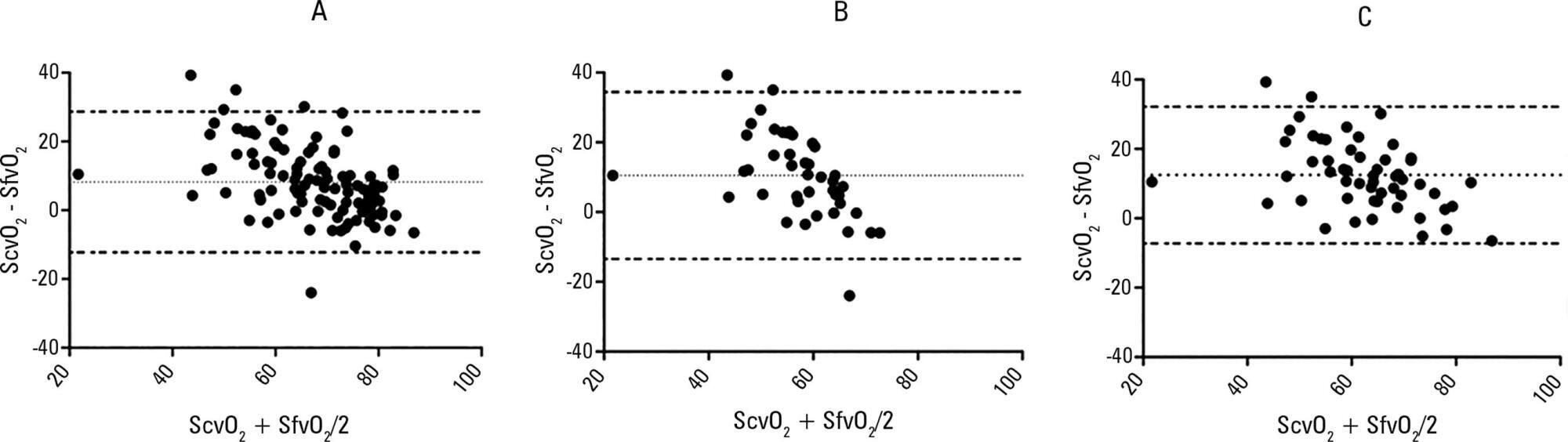
The purpose of this study was to test if venous blood drawn from femoral access can be used to estimate the central venous oxygen saturation and arterial lactate levels in critically ill patients.
Bland-Altman analysis and Spearman correlations were used to compare the femoral venous oxygen saturation and central venous oxygen saturation as well as arterial lactate levels and femoral lactate. A pre-specified subgroup analysis was conducted in patients with signs of hypoperfusion. In addition, the clinical agreement was also investigated.
Blood samples were obtained in 26 patients. In 107 paired samples, there was a moderate correlation (r = 0.686, p < 0.0001) between the central venous oxygen saturation and femoral venous oxygen saturation with a bias of 8.24 ± 10.44 (95% limits of agreement: -12.23 to 28.70). In 102 paired samples, there was a strong correlation between the arterial lactate levels and femoral lactate levels (r = 0.972, p < 0.001) with a bias of -2.71 ± 9.86 (95% limits of agreement: -22.03 to 16.61). The presence of hypoperfusion did not significantly change these results. The clinical agreement for venous saturation was inadequate, with different therapeutic decisions in 22.4% of the situation; for lactate, this was the case only in 5.2% of the situations.
Femoral venous oxygen saturation should not be used as a surrogate of central venous oxygen saturation. However, femoral lactate levels can be used in clinical practice, albeit with caution.
Abstract
Crit Care Sci. 2023;35(4):342-344
DOI 10.5935/2965-2774.20230263-pt