Abstract
Rev Bras Ter Intensiva. 2022;34(4):507-518
DOI 10.5935/0103-507X.20220145-en
To verify strategies for the prevention and treatment of abstinence syndrome in a pediatric intensive care unit.
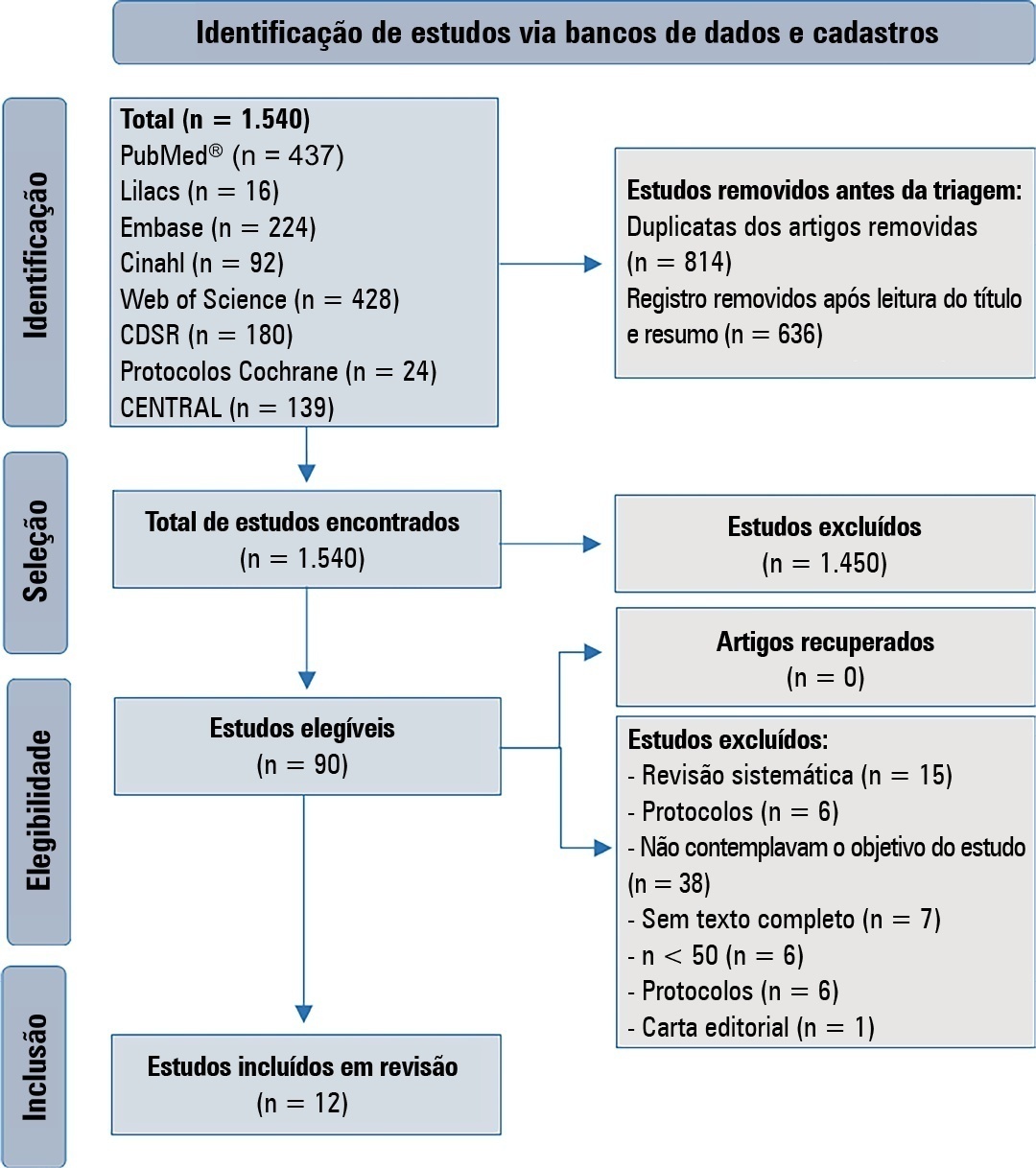
This is a systematic review in the PubMed database®, Lilacs, Embase, Web of Science, Cochrane, Cinahl, Cochrane Database Systematic Review and CENTRAL. A three-step search strategy was used for this review, and the protocol was approved in PROSPERO (CRD42021274670).
Twelve articles were included in the analysis. There was great heterogeneity among the studies included, especially regarding the therapeutic regimens used for sedation and analgesia. Midazolam doses ranged from 0.05mg/kg/hour to 0.3mg/kg/hour. Morphine also varied considerably, from 10mcg/kg/hour to 30mcg/kg/hour, between studies. Among the 12 selected studies, the most commonly used scale for the identification of withdrawal symptoms was the Sophia Observational Withdrawal Symptoms Scale. In three studies, there was a statistically significant difference in the prevention and management of the withdrawal syndrome due to the implementation of different protocols (p < 0.01 and p < 0.001).
There was great variation in the sedoanalgesia regimen used by the studies and the method of weaning and evaluation of withdrawal syndrome. More studies are needed to provide more robust evidence about the most appropriate treatment for the prevention and reduction of withdrawal signs and symptoms in critically ill children.
CRD 42021274670
Abstract
Rev Bras Ter Intensiva. 2006;18(2):186-189
DOI 10.1590/S0103-507X2006000200012
BACKGROUND AND OBJECTIVES: Critically ill patients frequently need to use sedative and analgesic drugs, as part of their treatment or during several procedures. It is a challenge for all intensive care providers to determine the best drug to be used for each patient, with less collateral effects. The objective of this study is to describe the background to give the use of remifentanil in intensive care clinical practice. CONTENTS: Remifentanil is a short acting opioid agonist little used in intensive care medicine. Several studies have been published, showing that remifentanil is a safe drug to be used for the sedation and analgesia for intensive care patients, still needing more information regardless to septic shock patients. CONCLUSIONS: Because remifentanil is a relatively new drug, it is not yet part of the routine drugs used for intensive care providers, although solid evidences of its safety and efficiency for critically ill patients.