Abstract
Rev Bras Ter Intensiva. 2022;34(3):327-334
DOI 10.5935/0103-507X.20220070-en
To describe the implementation and results of the collaborative PROADI-SUS project by the Brazilian Ministry of Health to reduce healthcare-associated infections: ventilator-associated pneumonia, primary central line-associated bloodstream infection and catheter-associated urinary tract infections.
This was a prospective observational study that investigated the implementation stages and outcomes during 18 months in five intensive care units in the city of Recife. Reductions in healthcare-associated infections in each unit were calculated using previous medians compared to those of the study period.
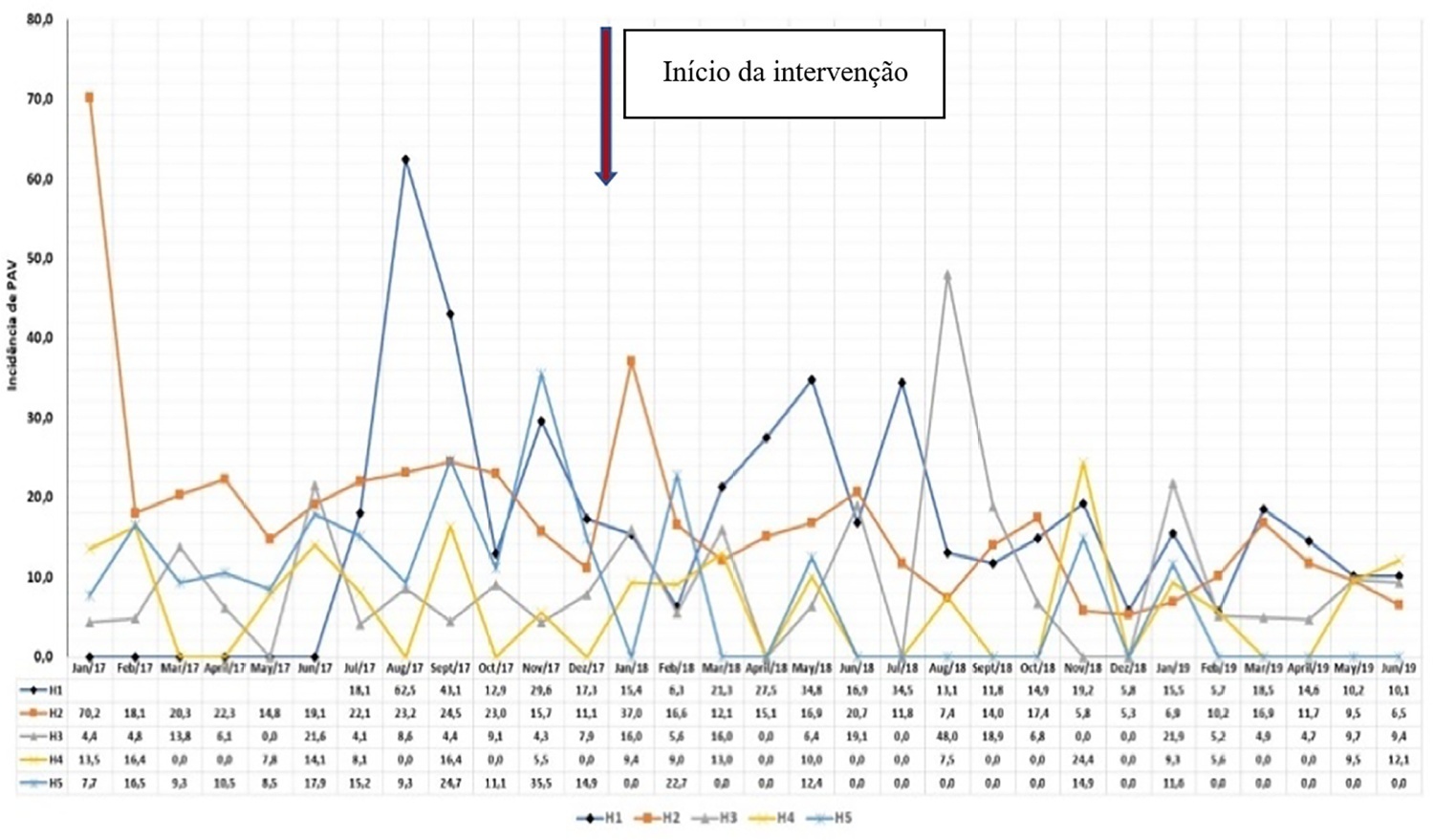
The goal of reducing the three healthcare-associated infections, i.e., 30% in 18 months, was achieved in at least one of the healthcare-associated infections and was also achieved for two healthcare-associated infections in two hospitals and three healthcare-associated infections in just one hospital; the latter reached the target of 36 months. Implementing the bundles and monitoring the results by the professionals were considered essential actions by the local management teams. In addition, the acquisition of supplies and their availability alongside the beds, signage, checklists, staff awareness, adaptation, team building, training and celebration of achievements were assessed as being relevant for reducing healthcare-associated infections.
The collaborative approach reduced healthcare-associated infections, despite partial adherence to the bundles. The hypothesis is that success is related to the project methodology and motivated multidisciplinary teams, especially nursing teams.
Abstract
Rev Bras Ter Intensiva. 2009;21(1):45-50
DOI 10.1590/S0103-507X2009000100007
OBJECTIVE: A randomized clinical trial is a prospective study that compares the effect and value of interventions in human beings, of one or more groups vs. a control group. The objective of this study was to evaluate the quality of published randomized clinical trials in Intensive care in Brazil. METHODS: All randomized clinical trials in intensive care found by manual search in Revista Brasileira de Terapia Intensiva from January 2001 to March 2008 were assessed to evaluate their description by the quality scale. Descriptive statistics and a 95 % confidence interval were used for the primary outcome. Our primary outcome was the randomized clinical trial quality. RESULTS: Our search found 185 original articles, of which 14 were randomized clinical trials. Only one original article (7.1%) showed good quality. There was no statistical significance between the collected data and the data shown in the hypothesis of this search. CONCLUSION: It can be concluded that in the sample of assessed articles 7% of the randomized clinical trials in intensive care published in a single intensive care journal in Brazil, present good methodological quality.
