Home
Abstract
Rev Bras Ter Intensiva. 2011;23(2):158-163
DOI 10.1590/S0103-507X2011000200007
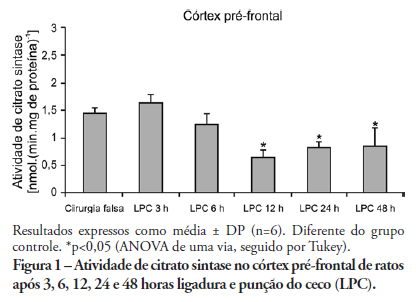
OBJECTIVE: An extensive body of evidence from experimental studies indicates that sepsis is associated with increased reactive oxygen species production, depletion of antioxidants, and accumulation of markers of oxidative stress. Moreover, mitochondrial dysfunction has been implicated in the pathogenesis of multiple organ dysfunction syndrome (MODS). Citrate synthase is an enzyme localized in the mitochondrial matrix and an important component of the Krebs cycle; consequently, citrate synthase has been used as a quantitative enzyme marker for the presence of intact mitochondria. Thus, we investigated citrate synthase activity in the brains of rats submitted to a cecal ligation puncture model of sepsis. METHODS: At several times points (3, 6, 12, 24 and 48 hours) after the cecal ligation puncture operation, six rats were killed by decapitation. Their brains were removed, and the hippocampus, striatum, cerebellum, cerebral cortex and prefrontal cortex were dissected and used to determine citrate synthase activity. RESULTS: We found that citrate synthase activity in the prefrontal cortex was inhibited 12, 24 and 48 hours after cecal ligation puncture. In the cerebral cortex, citrate synthase activity was inhibited 3, 12, 24 and 48 hours after cecal ligation puncture. Citrate synthase was not affected in the hippocampus, striatum or cerebellum up to 48 hours after cecal ligation puncture. CONCLUSION: Considering that energy impairment due to mitochondrial dysfunction in sepsis has been well described and that oxidative stress plays a crucial role in sepsis development, we believe that energy impairment may also be involved in these processes. If citrate synthase inhibition also occurs in a sepsis model, it is tempting to speculate that a reduction in brain metabolism may be related to the pathophysiology of this disease.


Abstract
Rev Bras Ter Intensiva. 2011;23(2):158-163
DOI 10.1590/S0103-507X2011000200007
OBJECTIVE: An extensive body of evidence from experimental studies indicates that sepsis is associated with increased reactive oxygen species production, depletion of antioxidants, and accumulation of markers of oxidative stress. Moreover, mitochondrial dysfunction has been implicated in the pathogenesis of multiple organ dysfunction syndrome (MODS). Citrate synthase is an enzyme localized in the mitochondrial matrix and an important component of the Krebs cycle; consequently, citrate synthase has been used as a quantitative enzyme marker for the presence of intact mitochondria. Thus, we investigated citrate synthase activity in the brains of rats submitted to a cecal ligation puncture model of sepsis. METHODS: At several times points (3, 6, 12, 24 and 48 hours) after the cecal ligation puncture operation, six rats were killed by decapitation. Their brains were removed, and the hippocampus, striatum, cerebellum, cerebral cortex and prefrontal cortex were dissected and used to determine citrate synthase activity. RESULTS: We found that citrate synthase activity in the prefrontal cortex was inhibited 12, 24 and 48 hours after cecal ligation puncture. In the cerebral cortex, citrate synthase activity was inhibited 3, 12, 24 and 48 hours after cecal ligation puncture. Citrate synthase was not affected in the hippocampus, striatum or cerebellum up to 48 hours after cecal ligation puncture. CONCLUSION: Considering that energy impairment due to mitochondrial dysfunction in sepsis has been well described and that oxidative stress plays a crucial role in sepsis development, we believe that energy impairment may also be involved in these processes. If citrate synthase inhibition also occurs in a sepsis model, it is tempting to speculate that a reduction in brain metabolism may be related to the pathophysiology of this disease.


Abstract
Rev Bras Ter Intensiva. 2011;23(1):96-103
DOI 10.1590/S0103-507X2011000100015
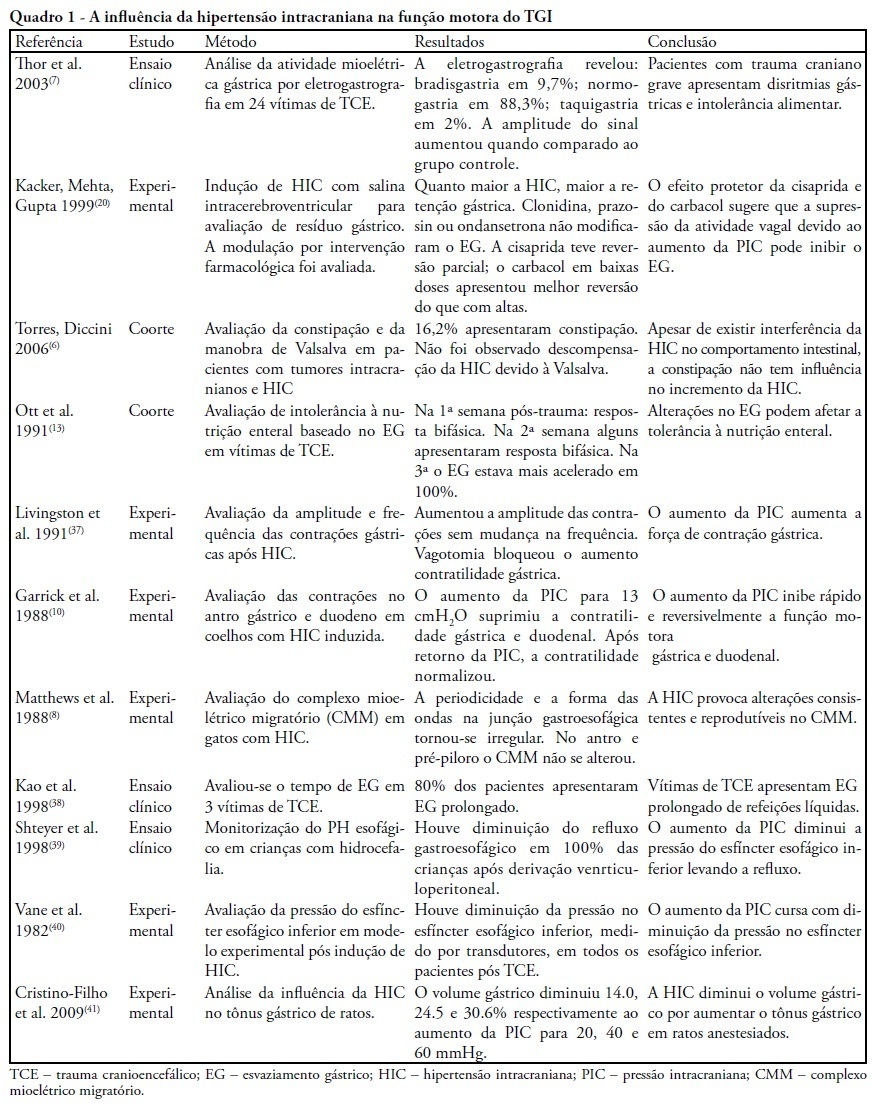
Subarachnoid hemorrhage can increase intracranial pressure, causing significant morbidity. Acute gastric dilation and delayed gastric emptying are commonly seen in patients with intracranial hypertension, and correction of these gastric abnormalities can facilitate the recovery of patients with brain injuries. We conducted a literature review of both national and international health sciences medical journals and electronic libraries spanning the last twenty-eight years and focused on the brain, gut motility and gastric emptying functional axis either in experimental animal models of brain injury or patients with acute cerebral injuries. Decreased parasympathetic tonus is a potential cause of intracranial hypertension-related food intolerance. Changes in gastrointestinal transit after a brain injury follow a biphasic pattern: an initial phase of accelerated gastric emptying and a late stage of intestinal lethargy. Changes in the physiology underlying gut motility may be essential for homeostatic stabilization in hemodynamically unstable patients. Research studies are necessary to understand the difficult management of intensive care patients with intracranial hypertension secondary to subarachnoid hemorrhages resulting from traumatic brain injuries or rupture of a cerebral aneurysm. Increased intracranial pressure induces massive increases in sympathetic activity, which is responsible for many of the peripheral systemic and gastrointestinal symptoms. Brain injuries leading to significant increases in intracranial pressure result in delayed gastrointestinal emptying due to autonomic nervous system changes.


Abstract
Rev Bras Ter Intensiva. 2011;23(1):96-103
DOI 10.1590/S0103-507X2011000100015
Subarachnoid hemorrhage can increase intracranial pressure, causing significant morbidity. Acute gastric dilation and delayed gastric emptying are commonly seen in patients with intracranial hypertension, and correction of these gastric abnormalities can facilitate the recovery of patients with brain injuries. We conducted a literature review of both national and international health sciences medical journals and electronic libraries spanning the last twenty-eight years and focused on the brain, gut motility and gastric emptying functional axis either in experimental animal models of brain injury or patients with acute cerebral injuries. Decreased parasympathetic tonus is a potential cause of intracranial hypertension-related food intolerance. Changes in gastrointestinal transit after a brain injury follow a biphasic pattern: an initial phase of accelerated gastric emptying and a late stage of intestinal lethargy. Changes in the physiology underlying gut motility may be essential for homeostatic stabilization in hemodynamically unstable patients. Research studies are necessary to understand the difficult management of intensive care patients with intracranial hypertension secondary to subarachnoid hemorrhages resulting from traumatic brain injuries or rupture of a cerebral aneurysm. Increased intracranial pressure induces massive increases in sympathetic activity, which is responsible for many of the peripheral systemic and gastrointestinal symptoms. Brain injuries leading to significant increases in intracranial pressure result in delayed gastrointestinal emptying due to autonomic nervous system changes.


Search
Search in:
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis


