You searched for:"Letícia Kawano-Dourado"
We found (2) results for your search.-
Special Article
Quality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
Abstract
Special ArticleQuality of life and long-term outcomes after hospitalization for COVID-19: Protocol for a prospective cohort study (Coalition VII)
Rev Bras Ter Intensiva. 2021;33(1):31-37
DOI 10.5935/0103-507X.20210003
Views0See moreAbstract
Introduction:
The long-term effects caused by COVID-19 are unknown. The present study aims to assess factors associated with health-related quality of life and long-term outcomes among survivors of hospitalization for COVID-19 in Brazil.
Methods:
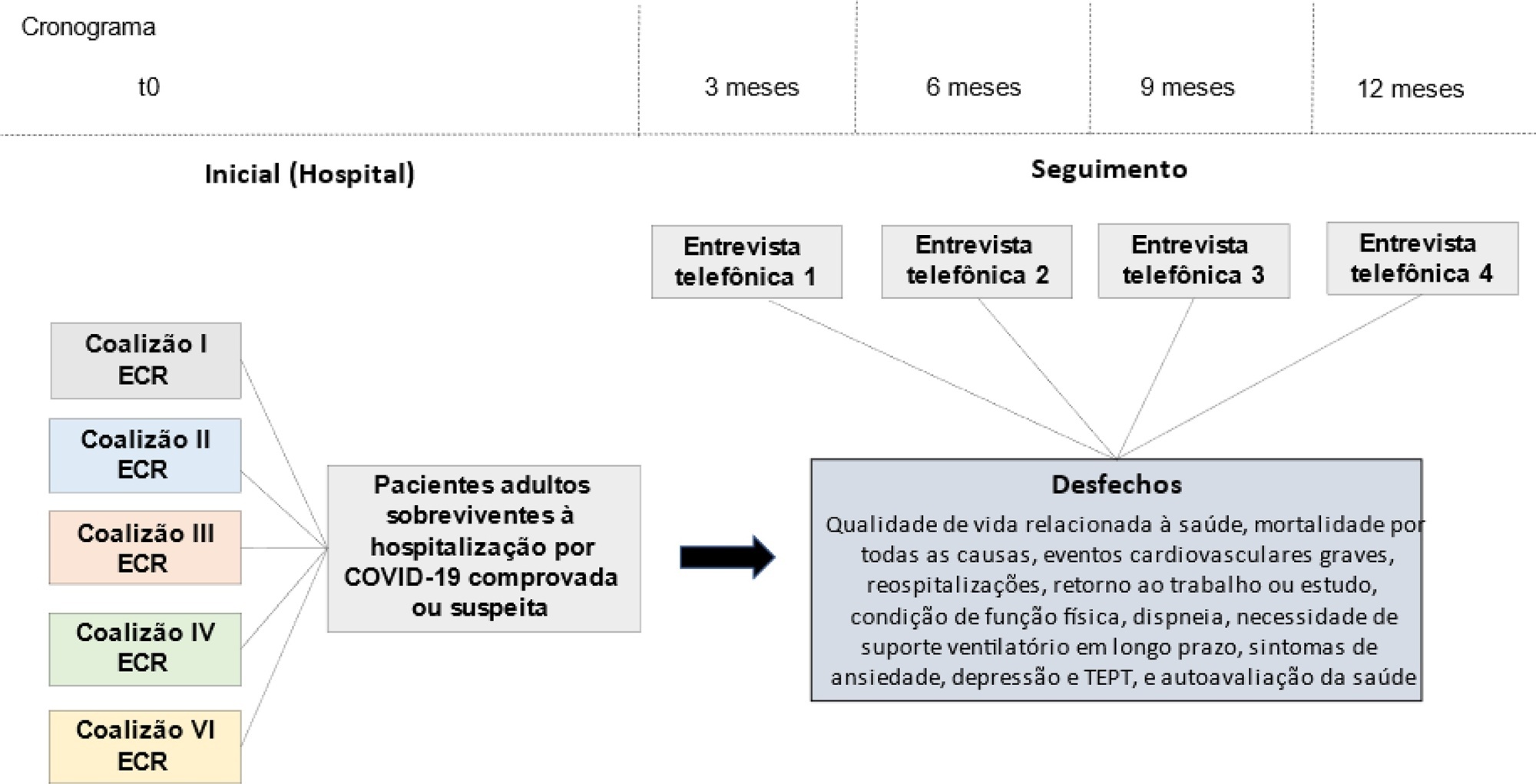
This is a multicenter prospective cohort study nested in five randomized clinical trials designed to assess the effects of specific COVID-19 treatments in over 50 centers in Brazil. Adult survivors of hospitalization due to proven or suspected SARS-CoV-2 infection will be followed-up for a period of 1 year by means of structured telephone interviews. The primary outcome is the 1-year utility score of health-related quality of life assessed by the EuroQol-5D3L. Secondary outcomes include all-cause mortality, major cardiovascular events, rehospitalizations, return to work or study, physical functional status assessed by the Lawton-Brody Instrumental Activities of Daily Living, dyspnea assessed by the modified Medical Research Council dyspnea scale, need for long-term ventilatory support, symptoms of anxiety and depression assessed by the Hospital Anxiety and Depression Scale, symptoms of posttraumatic stress disorder assessed by the Impact of Event Scale-Revised, and self-rated health assessed by the EuroQol-5D3L Visual Analog Scale. Generalized estimated equations will be performed to test the association between five sets of variables (1- demographic characteristics, 2- premorbid state of health, 3- characteristics of acute illness, 4- specific COVID-19 treatments received, and 5- time-updated postdischarge variables) and outcomes.
Ethics and dissemination:
The study protocol was approved by the Research Ethics Committee of all participant institutions. The results will be disseminated through conferences and peer-reviewed journals.
-
Original Article
Rationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
Abstract
Original ArticleRationale and design of the “Tocilizumab in patients with moderate to severe COVID-19: an open-label multicentre randomized controlled” trial (TOCIBRAS)
Rev Bras Ter Intensiva. 2020;32(3):337-347
DOI 10.5935/0103-507X.20200060
Views0See moreABSTRACT
Introduction:
Pro-inflammatory markers play a significant role in the disease severity of patients with COVID-19. Thus, anti-inflammatory therapies are attractive agents for potentially combating the uncontrolled inflammatory cascade in these patients. We designed a trial testing tocilizumab versus standard of care intending to improve the outcomes by inhibiting interleukin-6, an important inflammatory mediator in COVID-19.
Methods and analysis:
This open-label multicentre randomized controlled trial will compare clinical outcomes of tocilizumab plus standard of care versus standard of care alone in patients with moderate to severe COVID-19. Two of the following four criteria are required for protocol enrolment: D-dimer > 1,000ng/mL; C reactive protein > 5mg/dL, ferritin > 300mg/dL, and lactate dehydrogenase > upper limit of normal. The primary objective will be to compare the clinical status on day 15, as measured by a 7-point ordinal scale applied in COVID-19 trials worldwide. The primary endpoint will be assessed by an ordinal logistic regression assuming proportional odds ratios adjusted for stratification variables (age and sex).
Ethics and dissemination:
The TOCIBRAS protocol was approved by local and central (national) ethical committees in Brazil following current national and international guidelines/directives. Each participating center had the study protocol approved by their institutional review boards before initiating protocol enrolment. The data derived from this trial will be published regardless of the results. If proven active, this strategy could alleviate the consequences of the inflammatory response in COVID-19 patients and improve their clinical outcomes.

Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




