You searched for:"Cassio Luis Zandonai"
We found (2) results for your search.-
Clinical Report
Prospective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
Abstract
Clinical ReportProspective, randomized, controlled trial assessing the effects of a driving pressure–limiting strategy for patients with acute respiratory distress syndrome due to community-acquired pneumonia (STAMINA trial): protocol and statistical analysis plan
Crit Care Sci. 2024;36:e20240210en
DOI 10.62675/2965-2774.20240210-en
Views76ABSTRACT
Background:
Driving pressure has been suggested to be the main driver of ventilator-induced lung injury and mortality in observational studies of acute respiratory distress syndrome. Whether a driving pressure-limiting strategy can improve clinical outcomes is unclear.
Objective:
To describe the protocol and statistical analysis plan that will be used to test whether a driving pressure-limiting strategy including positive end-expiratory pressure titration according to the best respiratory compliance and reduction in tidal volume is superior to a standard strategy involving the use of the ARDSNet low-positive end-expiratory pressure table in terms of increasing the number of ventilator-free days in patients with acute respiratory distress syndrome due to community-acquired pneumonia.
Methods:
The ventilator STrAtegy for coMmunIty acquired pNeumoniA (STAMINA) study is a randomized, multicenter, open-label trial that compares a driving pressure-limiting strategy to the ARDSnet low-positive end-expiratory pressure table in patients with moderate-to-severe acute respiratory distress syndrome due to community-acquired pneumonia admitted to intensive care units. We expect to recruit 500 patients from 20 Brazilian and 2 Colombian intensive care units. They will be randomized to a driving pressure-limiting strategy group or to a standard strategy using the ARDSNet low-positive end-expiratory pressure table. In the driving pressure-limiting strategy group, positive end-expiratory pressure will be titrated according to the best respiratory system compliance.
Outcomes:
The primary outcome is the number of ventilator-free days within 28 days. The secondary outcomes are in-hospital and intensive care unit mortality and the need for rescue therapies such as extracorporeal life support, recruitment maneuvers and inhaled nitric oxide.
Conclusion:
STAMINA is designed to provide evidence on whether a driving pressure-limiting strategy is superior to the ARDSNet low-positive end-expiratory pressure table strategy for increasing the number of ventilator-free days within 28 days in patients with moderate-to-severe acute respiratory distress syndrome. Here, we describe the rationale, design and status of the trial.
Keywords:Extracorporeal membrane oxygenationPneumoniaPositive pressure respirationRespiration, artificialRespiratory distress syndromeVentilator-induced lung injurySee more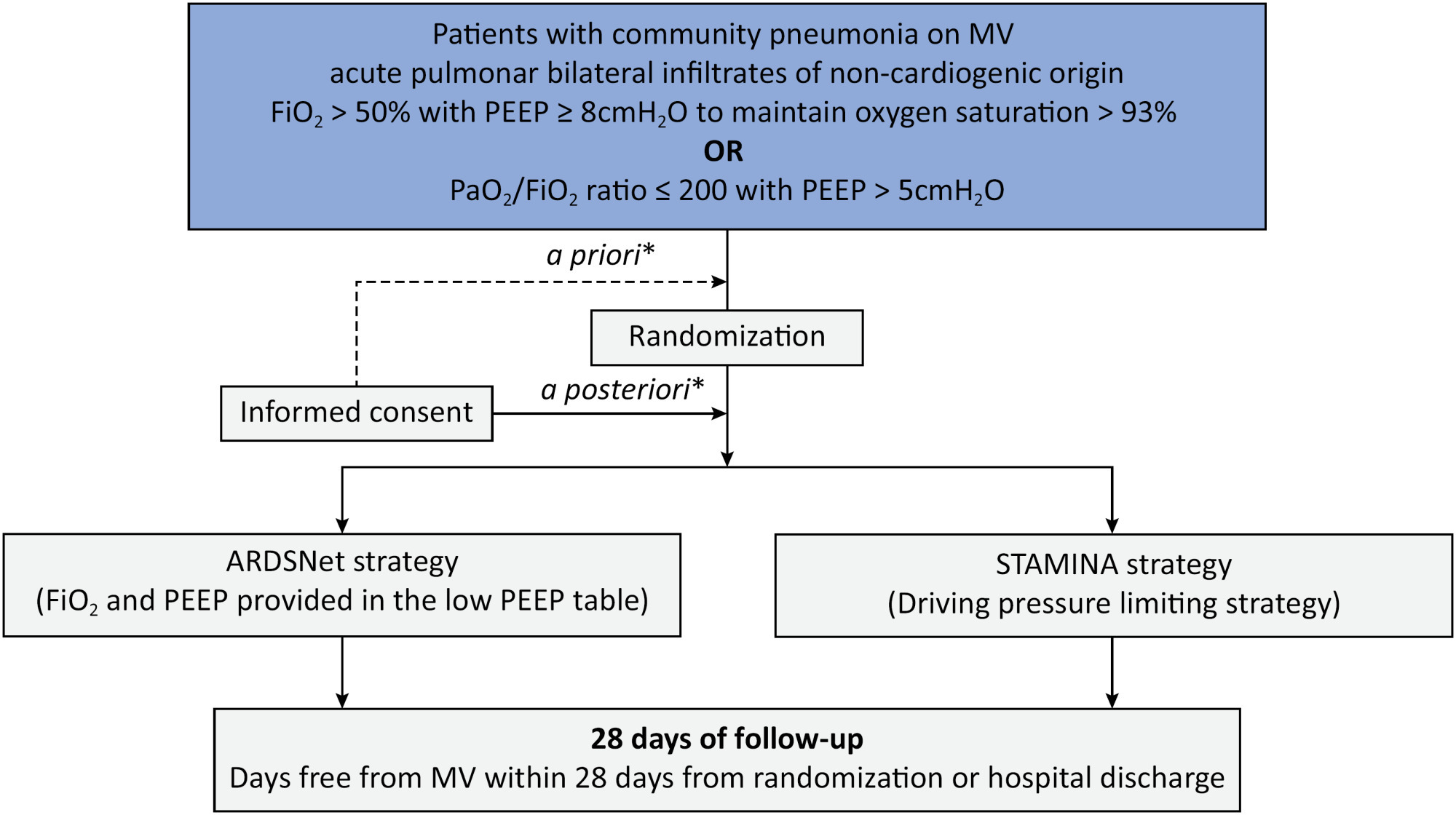
-
Original Article
IMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
Abstract
Original ArticleIMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
Views2ABSTRACT
Objective:
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
Methods:
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
Results:
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
Conclusion:
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.
Keywords:bacterialBacterial infectionsDatabaseDatabase management systemsDrug-resistanceIMPACTO-MRIntensive care unitsSoftwareSee more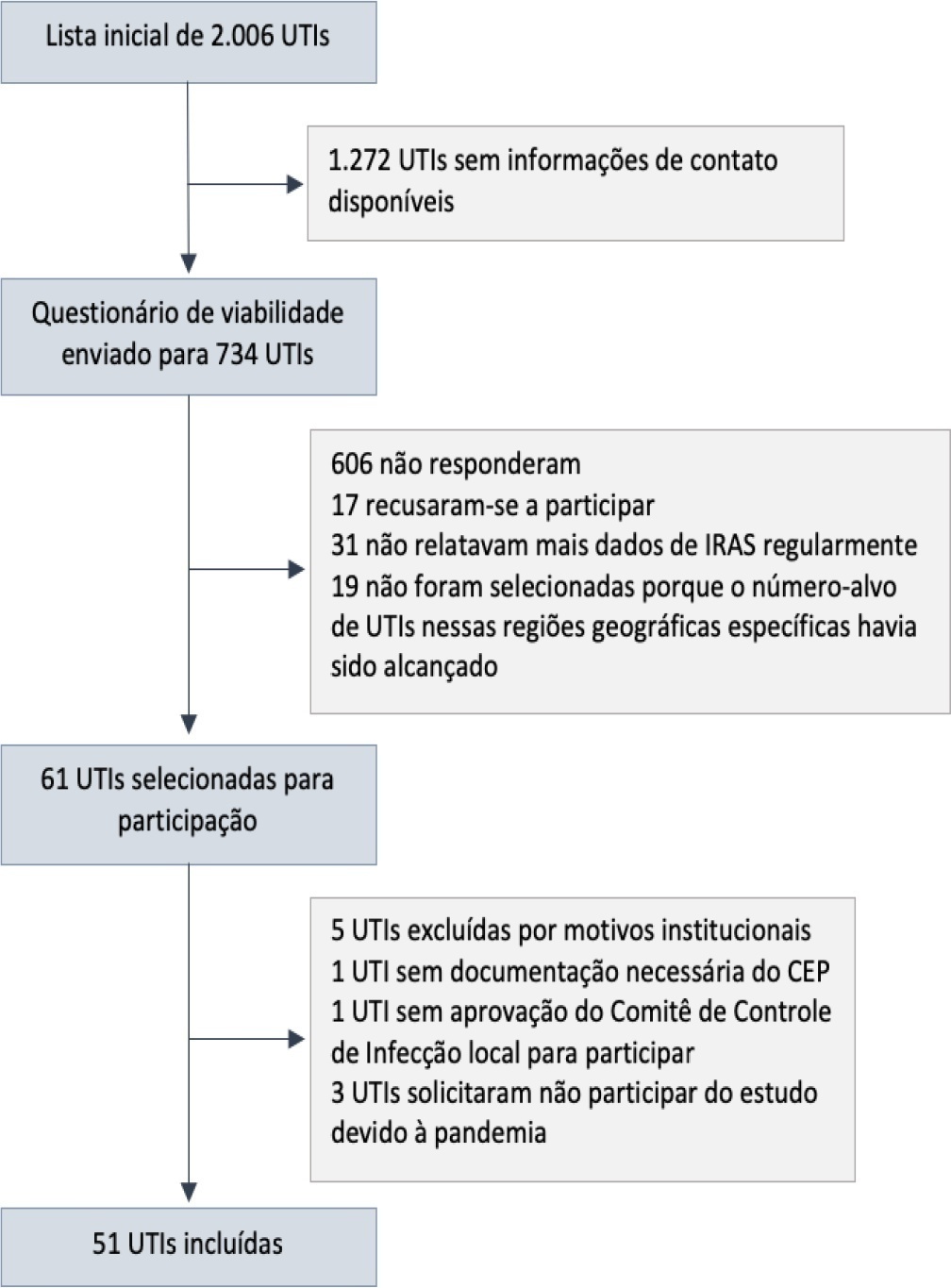
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




