Abstract
Revista Brasileira de Terapia Intensiva. 2021;33(1):48-67
DOI 10.5935/0103-507X.20210005
Proponer estrategias agile para este abordaje integral de la analgesia, sedación, delirium, implementación de movilidad temprana e inclusión familiar del paciente con síndrome de dificultad respiratoria aguda por COVID-19, considerando el alto riesgo de infección que existe entre los trabajadores de salud, el tratamiento humanitario que debemos brindar al paciente y su familia, en un contexto de falta estrategias terapéuticas específicas contra el virus globalmente disponibles a la fecha y una potencial falta de recursos sanitarios.
Se llevó a cabo una revision no sistemática de la evidencia científica en las principales bases de datos bibliográficos, sumada a la experiencia y juicio clínico nacional e internacional. Finalmente, se realizó un consenso de recomendaciones entre los integrantes del Comité de Analgesia, Sedación y Delirium de la Sociedad Argentina de Terapia Intensiva.
Se acordaron recomendaciones y se desarrollaron herramientas para asegurar un abordaje integral de analgesia, sedación, delirium, implementación de movilidad temprana e inclusión familiar del paciente adulto con síndrome de dificultad respiratoria aguda por COVID-19.
Ante el nuevo orden generado en las terapias intensivas por la progresión de la pandemia de COVID-19, proponemos no dejar atrás las buenas prácticas habituales, sino adaptarlas al contexto particular generado. Nuestro consenso está respaldado en la evidencia científica, la experiencia nacional e internacional, y será una herramienta de consulta atractiva en las terapias intensivas.
Abstract
Revista Brasileira de Terapia Intensiva. 2021;33(1):12-30
DOI 10.5935/0103-507X.20210002
To present guidelines on sensory motor stimulation for newborns and infants in the intensive care unit.
We employed a mixed methods design with a systematic review of the literature and recommendations based on scientific evidence and the opinions of physiotherapists with neonatal expertise. The research included studies published between 2010 and 2018 in the MEDLINE® and Cochrane databases that included newborns (preterm and term) and infants (between 28 days and 6 months of age) hospitalized in the intensive care unit and submitted to sensory motor stimulation methods. The studies found were classified according to the GRADE score by five physiotherapists in different regions of Brazil and presented at eight Scientific Congresses held to discuss the clinical practice guidelines.
We included 89 articles to construct the clinical practice guidelines. Auditory, gustatory and skin-to-skin stimulation stand out for enhancing vital signs, and tactile-kinesthetic massage and multisensory stimulation stand out for improving weight or sucking.
Although all modalities have good ratings for pain or stress control, it is recommended that sensory motor stimulation procedures be tailored to the infant’s specific needs and that interventions and be carried out by expert professionals.
Abstract
Revista Brasileira de Terapia Intensiva. 2021;33(1):38-47
DOI 10.5935/0103-507X.20210004
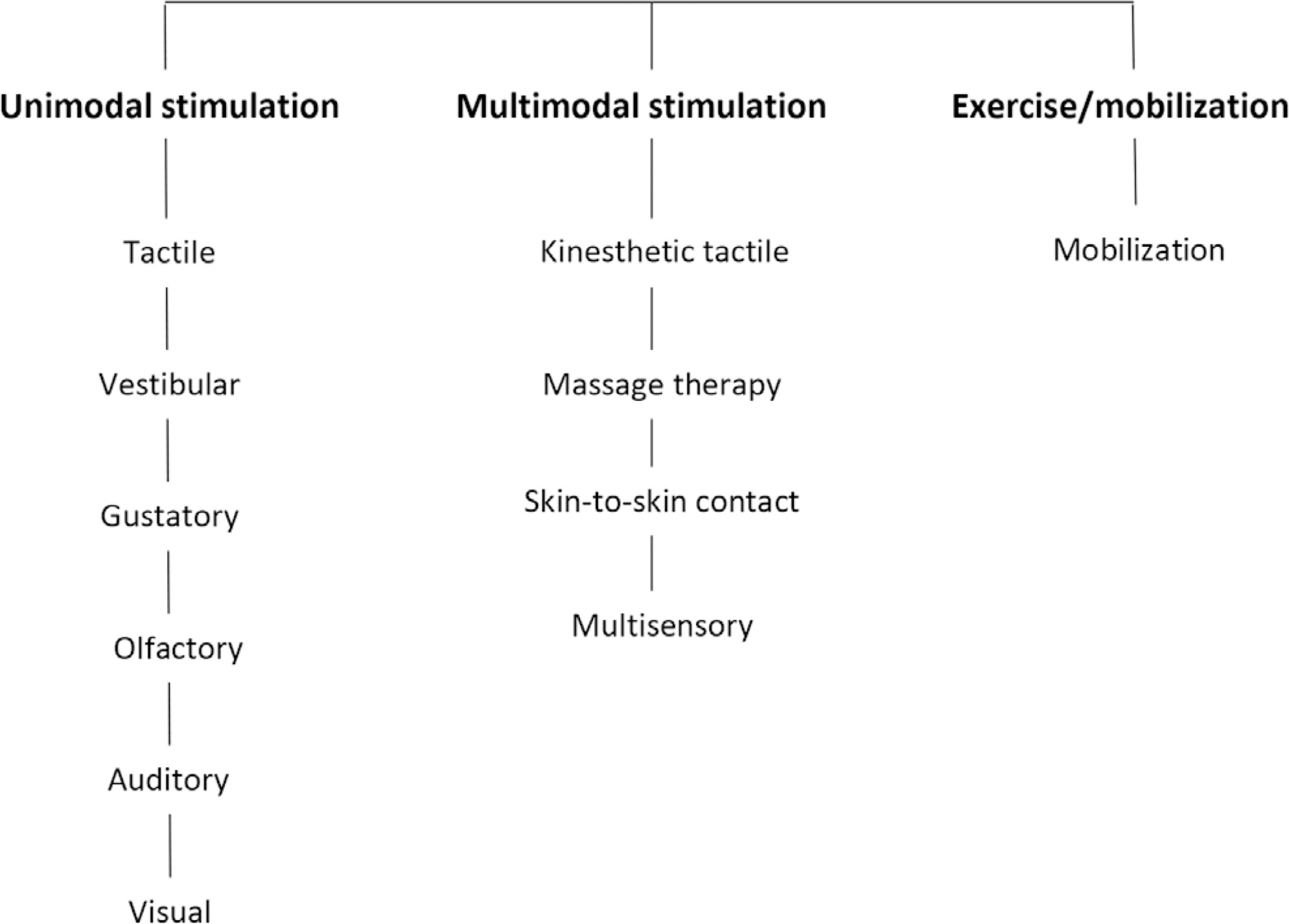
Triage for intensive care unit admission is a frequent event and is associated to worse clinical outcomes. The process of triage is variable and may be influenced by biases and prejudices, which could lead to potentially unfair decisions. The Brazilian Federal Council of Medicine (Conselho Federal de Medicina) has recently released a guideline for intensive care unit admission and discharge. The aim of this paper is to evaluate the ethical dilemmas related to the implementation of this guideline, through the accountability for reasonabless approach, known as A4R, as elaborated by Norman Daniels. We conclude that the guideline contemplates A4R conditions, but we observe that there is a need for indication of A4R-concordant criteria to operationalize the guidelines.
Abstract
Revista Brasileira de Terapia Intensiva. 2020;32(1):2-10
DOI 10.5935/0103-507X.20200002
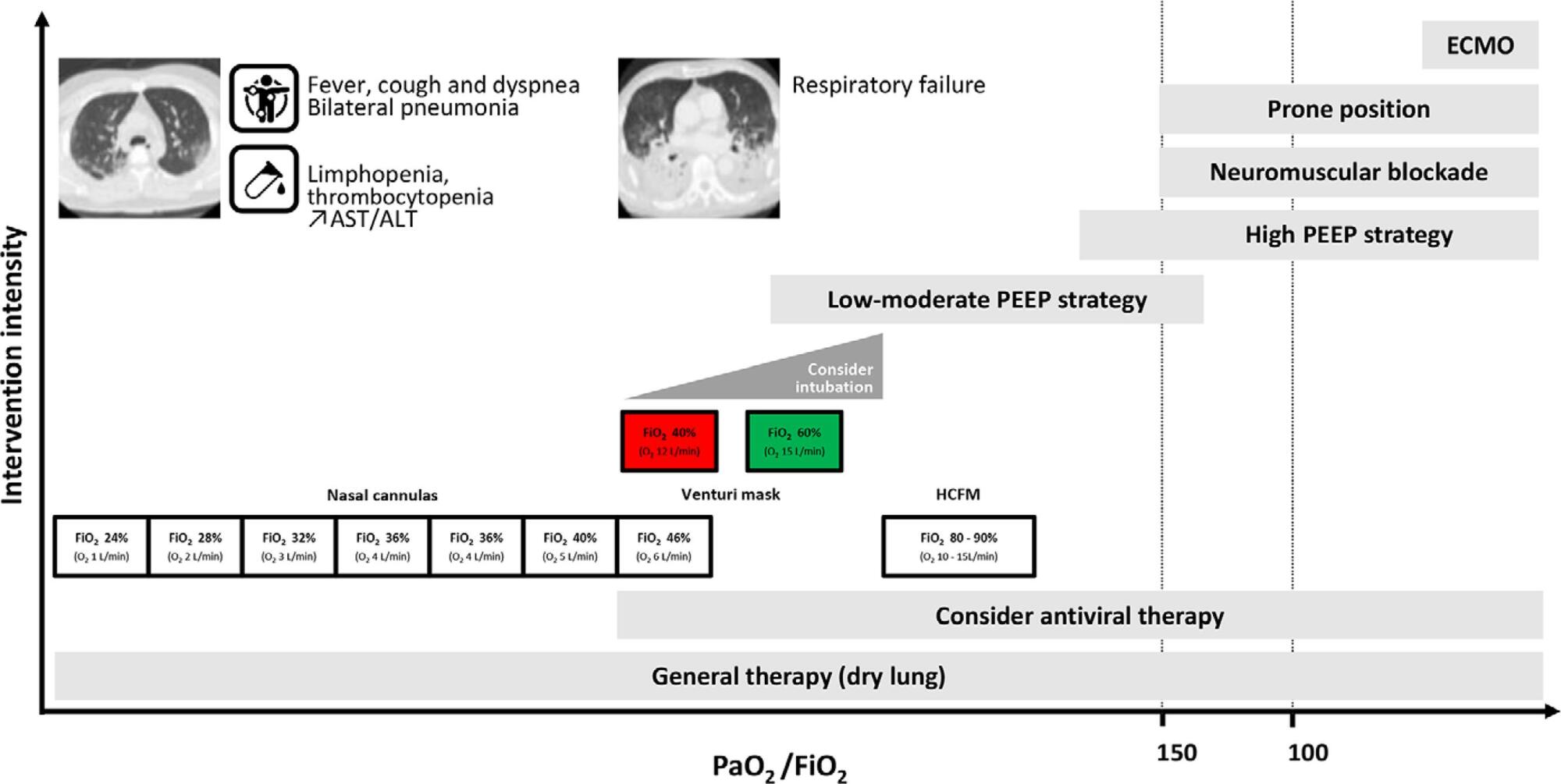
Current COVID-19 epidemics was declared on December 31, 2019 at the Wuhan city seafood market, rapidly spreading throughout China, and later reaching several countries (mainly South Korea, Japan, Italy and Iran) and, since March 1, reaching Portugal. Most of the infected patients present with mild symptoms, not requiring hospitalization. Among those admitted to the hospital, 6% to 10% require admission to the intensive care unit. These recommendations are aimed to support the organization of intensive care services to respond COVID-19, providing optimized care to the patient and protection for healthcare professionals.
Abstract
Revista Brasileira de Terapia Intensiva. 2019;31(1):5-14
DOI 10.5935/0103-507X.20190002
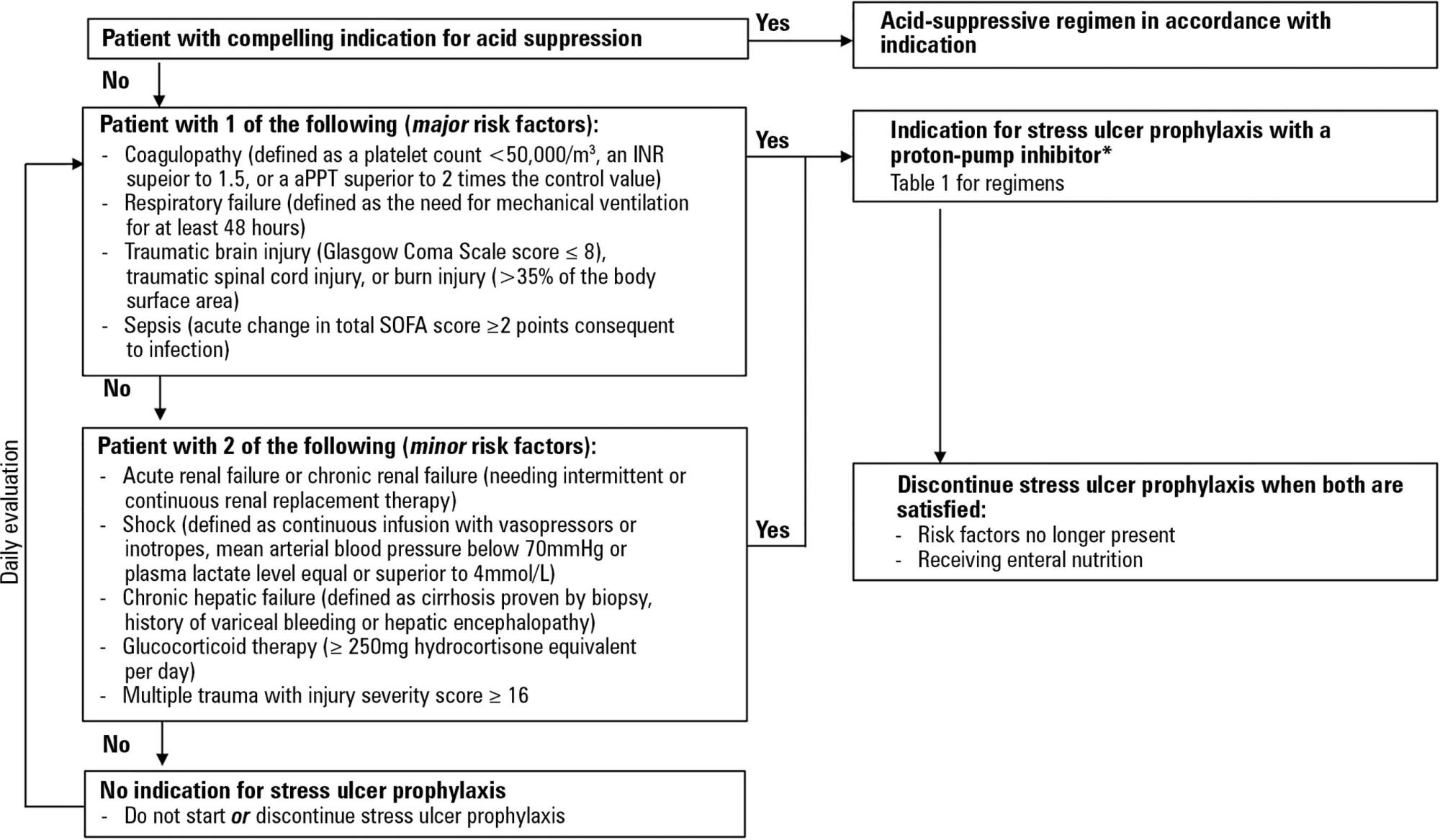
Critically ill patients are at risk of developing stress ulcers in the upper digestive tract. Agents that suppress gastric acid are commonly prescribed to reduce the incidence of clinically important stress ulcer-related gastrointestinal bleeding. However, the indiscriminate use of stress ulcer prophylaxis in all patients admitted to the intensive care unit is not warranted and can have potential adverse clinical effects and cost implications. The present guidelines from the Sociedade Portuguesa de Cuidados Intensivos summarizes the current evidence and gives six clinical statements and an algorithm aiming to provide a standardized prescribing policy for the use of stress ulcer prophylaxis in the intensive care unit.
Abstract
Revista brasileira de terapia intensiva. 2014;26(3):215-239
DOI 10.5935/0103-507X.20140034
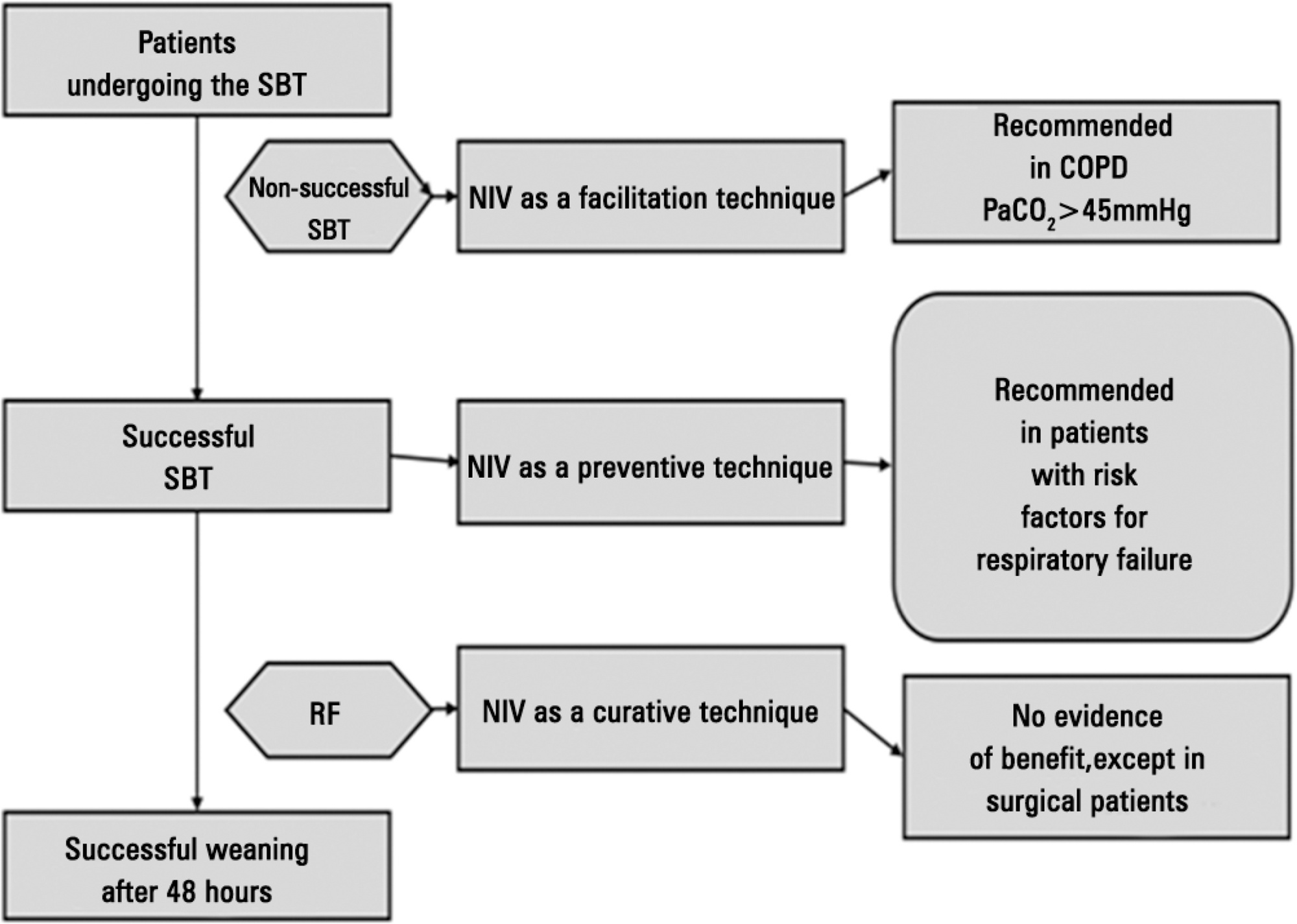
Perspectives on invasive and noninvasive ventilatory support for critically ill patients are evolving, as much evidence indicates that ventilation may have positive effects on patient survival and the quality of the care provided in intensive care units in Brazil. For those reasons, the Brazilian Association of Intensive Care Medicine (Associação de Medicina Intensiva Brasileira - AMIB) and the Brazilian Thoracic Society (Sociedade Brasileira de Pneumologia e Tisiologia - SBPT), represented by the Mechanical Ventilation Committee and the Commission of Intensive Therapy, respectively, decided to review the literature and draft recommendations for mechanical ventilation with the goal of creating a document for bedside guidance as to the best practices on mechanical ventilation available to their members. The document was based on the available evidence regarding 29 subtopics selected as the most relevant for the subject of interest. The project was developed in several stages, during which the selected topics were distributed among experts recommended by both societies with recent publications on the subject of interest and/or significant teaching and research activity in the field of mechanical ventilation in Brazil. The experts were divided into pairs that were charged with performing a thorough review of the international literature on each topic. All the experts met at the Forum on Mechanical Ventilation, which was held at the headquarters of AMIB in São Paulo on August 3 and 4, 2013, to collaboratively draft the final text corresponding to each sub-topic, which was presented to, appraised, discussed and approved in a plenary session that included all 58 participants and aimed to create the final document.
Abstract
Revista Brasileira de Terapia Intensiva. 2013;25(1):3-5
DOI 10.1590/S0103-507X2013000100002
Abstract
Revista Brasileira de Terapia Intensiva. 2012;24(4):322-325