You searched for:"Karla Aparecida Silva"
We found (1) results for your search.-
Special Article
Automated documentation of vital parameters in wards using portable stations – Effect on proper triggering of the rapid response team: a study protocol of a cluster randomized clinical trial
Rev Bras Ter Intensiva. 2022;34(3):319-326
Abstract
Special ArticleAutomated documentation of vital parameters in wards using portable stations – Effect on proper triggering of the rapid response team: a study protocol of a cluster randomized clinical trial
Rev Bras Ter Intensiva. 2022;34(3):319-326
DOI 10.5935/0103-507X.20220101-en
Views2ABSTRACT
Objective:
To evaluate the effectiveness of the Welch Allyn Connex® Spot Monitor/Hillrom Connecta™ solution in activating the rapid response team in a timely manner compared to manual activation.
Methods:
The Hillrom study is a single-center, open-label, superiority, cluster-randomized, parallel-group (1:1 allocation ratio) clinical trial that will be conducted in a tertiary hospital. Two sets of three wards with 28 beds will be included (one as the intervention cluster and the other as the control). The wards will be randomly assigned to use the Welch Allyn Connex® Spot Monitor/Hillrom Connecta™ automated solution (intervention cluster) or to maintain the usual routine (control cluster) regarding rapid response team activation. The primary outcome will be the absolute number of episodes of rapid response team triggering in an appropriate time; as secondary outcomes, clinical features (mortality, cardiac arrest, need for intensive care unit admission and duration of hospitalization) will be assessed according to clusters in an exploratory way. A sample size of 216 rapid response team activations was estimated to identify a possible difference between the groups. The protocol has been approved by the institutional Research Ethics Committee.
Expected results:
The Welch Allyn Connex® Spot Monitor/Hillrom Connecta™ automated solution is expected to be more effective in triggering the nurse call system to activate the rapid response team in a timely and adequate manner compared to manual triggering (usual practice).
ClinicalTrials.gov:
NCT04648579
Keywords:computerizedHealth information interoperabilityHospital rapid response teamMedical records systemsNursing stationsVital signsSee more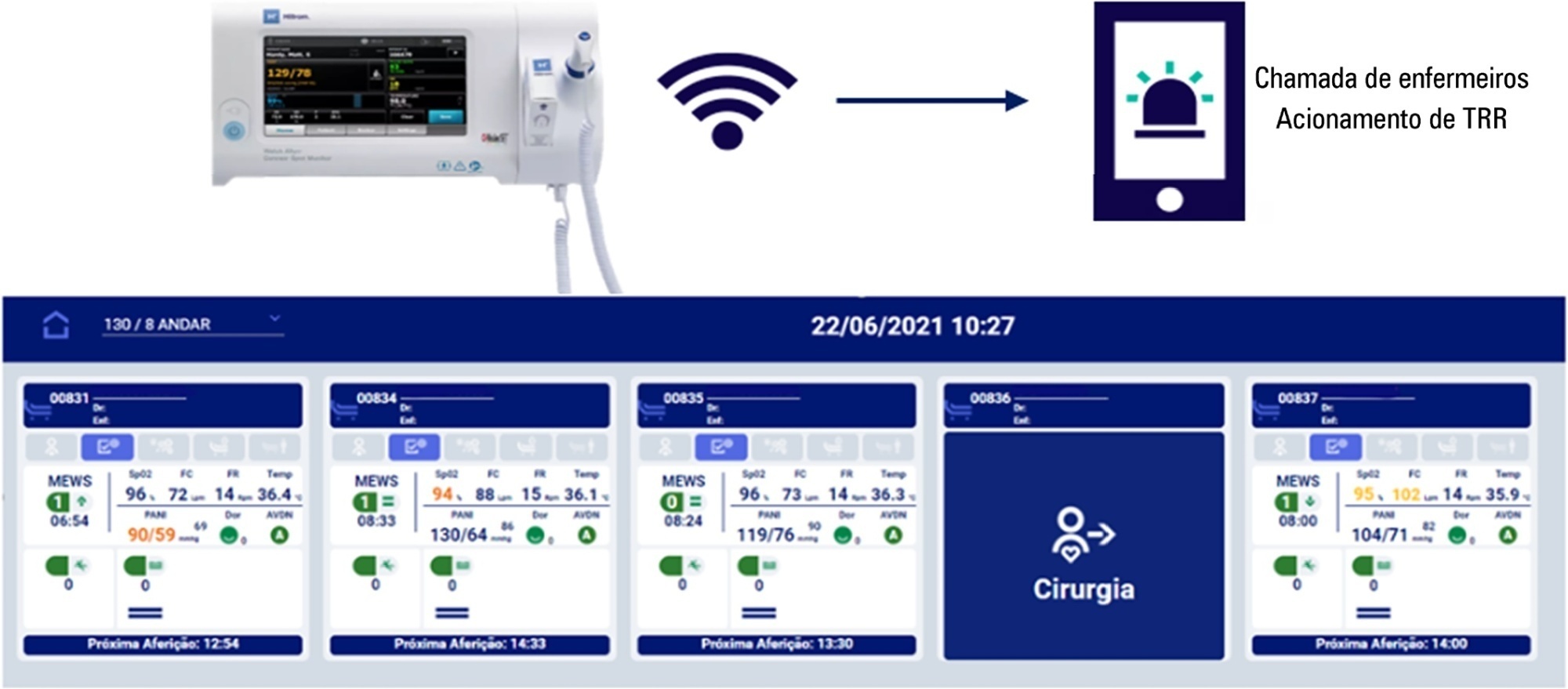
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis




