You searched for:"Nicole Alberti Golin"
We found (2) results for your search.-
Original Article
IMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
- Bruno M Tomazini
 ,
, - Antonio Paulo Nassar Jr
 ,
, - Thiago Costa Lisboa
 ,
, - Luciano César Pontes de Azevedo
 ,
, - Viviane Cordeiro Veiga
 , [ … ],
, [ … ], - Alexandre Biasi Cavalcanti
Abstract
Original ArticleIMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
- Bruno M Tomazini
 ,
, - Antonio Paulo Nassar Jr
 ,
, - Thiago Costa Lisboa
 ,
, - Luciano César Pontes de Azevedo
 ,
, - Viviane Cordeiro Veiga
 ,
, - Daniela Ghidetti Mangas Catarino
 ,
, - Debora Vacaro Fogazzi
 ,
, - Beatriz Arns
 ,
, - Filipe Teixeira Piastrelli
 ,
, - Camila Dietrich
 ,
, - Karina Leal Negrelli,
- Isabella de Andrade Jesuíno,
- Luiz Fernando Lima Reis,
- Renata Rodrigues de Mattos,
- Carla Cristina Gomes Pinheiro,
- Mariane Nascimento Luz,
- Clayse Carla da Silva Spadoni,
- Elisângela Emilene Moro,
- Flávia Regina Bueno,
- Camila Santana Justo Cintra Sampaio,
- Débora Patrício Silva,
- Franca Pellison Baldassare,
- Ana Cecilia Alcantara Silva,
- Thabata Veiga,
- Leticia Barbante,
- Marianne Lambauer,
- Viviane Bezerra Campos,
- Elton Santos,
- Renato Hideo Nakawaga Santos,
- Ligia Nasi Laranjeiras,
- Nanci Valeis,
- Eliana Santucci,
- Tamiris Abait Miranda,
- Ana Cristina Lagoeiro do Patrocínio,
- Andréa de Carvalho,
- Eduvirgens Maria Couto de Sousa,
- Ancelmo Honorato Ferraz de Sousa,
- Daniel Tavares Malheiro,
- Isabella Lott Bezerra,
- Mirian Batista Rodrigues,
- Julliana Chicuta Malicia,
- Sabrina Souza da Silva,
- Bruna dos Passos Gimenes,
- Guilhermo Prates Sesin,
- Alexandre Prehn Zavascki,
- Daniel Sganzerla,
- Gregory Saraiva Medeiros,
- Rosa da Rosa Minho dos Santos,
- Fernanda Kelly Romeiro Silva,
- Maysa Yukari Cheno,
- Carolinne Ferreira Abrahão,
- Haliton Alves de Oliveira Junior,
- Leonardo Lima Rocha,
- Pedro Aniceto Nunes Neto,
- Valéria Chagas Pereira,
- Luis Eduardo Miranda Paciência,
- Elaine Silva Bueno,
- Eliana Bernadete Caser,
- Larissa Zuqui Ribeiro,
- Caio Cesar Ferreira Fernandes,
- Juliana Mazzei Garcia,
- Vanildes de Fátima Fernandes Silva,
- Alisson Junior dos Santos,
- Flávia Ribeiro Machado,
- Maria Aparecida de Souza,
- Bianca Ramos Ferronato,
- Hugo Corrêa de Andrade Urbano,
- Danielle Conceição Aparecida Moreira,
- Vicente Cés de Souza-Dantas,
- Diego Meireles Duarte,
- Juliana Coelho,
- Rodrigo Cruvinel Figueiredo,
- Fernanda Foreque,
- Thiago Gomes Romano,
- Daniel Cubos,
- Vladimir Miguel Spirale,
- Roberta Schiavon Nogueira,
- Israel Silva Maia,
- Cassio Luis Zandonai,
- Wilson José Lovato,
- Rodrigo Barbosa Cerantola,
- Tatiana Gozzi Pancev Toledo,
- Pablo Oscar Tomba,
- Joyce Ramos de Almeida,
- Luciana Coelho Sanches,
- Leticia Pierini,
- Mariana Cunha,
- Michelle Tereza Sousa,
- Bruna Azevedo,
- Felipe Dal-Pizzol,
- Danusa de Castro Damasio,
- Marina Peres Bainy,
- Dagoberta Alves Vieira Beduhn,
- Joana D’Arc Vila Nova Jatobá,
- Maria Tereza Farias de Moura,
- Leila Rezegue de Moraes Rego,
- Adria Vanessa da Silva,
- Luana Pontes Oliveira,
- Eliene Sá Sodré Filho,
- Silvana Soares dos Santos,
- Itallo de Lima Neves,
- Vanessa Cristina de Aquino Leão,
- João Lucidio Lobato Paes,
- Marielle Cristina Mendes Silva,
- Cláudio Dornas de Oliveira,
- Raquel Caldeira Brant Santiago,
- Jorge Luiz da Rocha Paranhos,
- Iany Grinezia da Silva Wiermann,
- Durval Ferreira Fonseca Pedroso,
- Priscilla Yoshiko Sawada,
- Rejane Martins Prestes,
- Glícia Cardoso Nascimento,
- Cintia Magalhães Carvalho Grion,
- Claudia Maria Dantas de Maio Carrilho,
- Roberta Lacerda Almeida de Miranda Dantas,
- Eliane Pereira Silva,
- Antônio Carlos da Silva,
- Sheila Mara Bezerra de Oliveira,
- Nicole Alberti Golin,
- Rogerio Tregnago,
- Valéria Paes Lima,
- Kamilla Grasielle Nunes da Silva,
- Emerson Boschi,
- Viviane Buffon,
- André Sant’Ana Machado,
- Leticia Capeletti,
- Rafael Botelho Foernges,
- Andréia Schubert de Carvalho,
- Lúcio Couto de Oliveira Junior,
- Daniela Cunha de Oliveira,
- Everton Macêdo Silva,
- Julival Ribeiro,
- Francielle Constantino Pereira,
- Fernanda Borges Salgado,
- Caroline Deutschendorf,
- Cristofer Farias da Silva,
- Andre Luiz Nunes Gobatto,
- Carolaine Bomfim de Oliveira,
- Marianna Deway Andrade Dracoulakis,
- Natália Oliveira Santos Alvaia,
- Roberta Machado de Souza,
- Larissa Liz Cardoso de Araújo,
- Rodrigo Morel Vieira de Melo,
- Luiz Carlos Santana Passos,
- Claudia Fernanda de Lacerda Vidal,
- Fernanda Lopes de Albuquerque Rodrigues,
- Pedro Kurtz,
- Cássia Righy Shinotsuka,
- Maria Brandão Tavares,
- Igor das Virgens Santana,
- Luciana Macedo da Silva Gavinho,
- Alaís Brito Nascimento,
- Adriano J Pereira,
- Alexandre Biasi Cavalcanti
Views2ABSTRACT
Objective:
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
Methods:
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
Results:
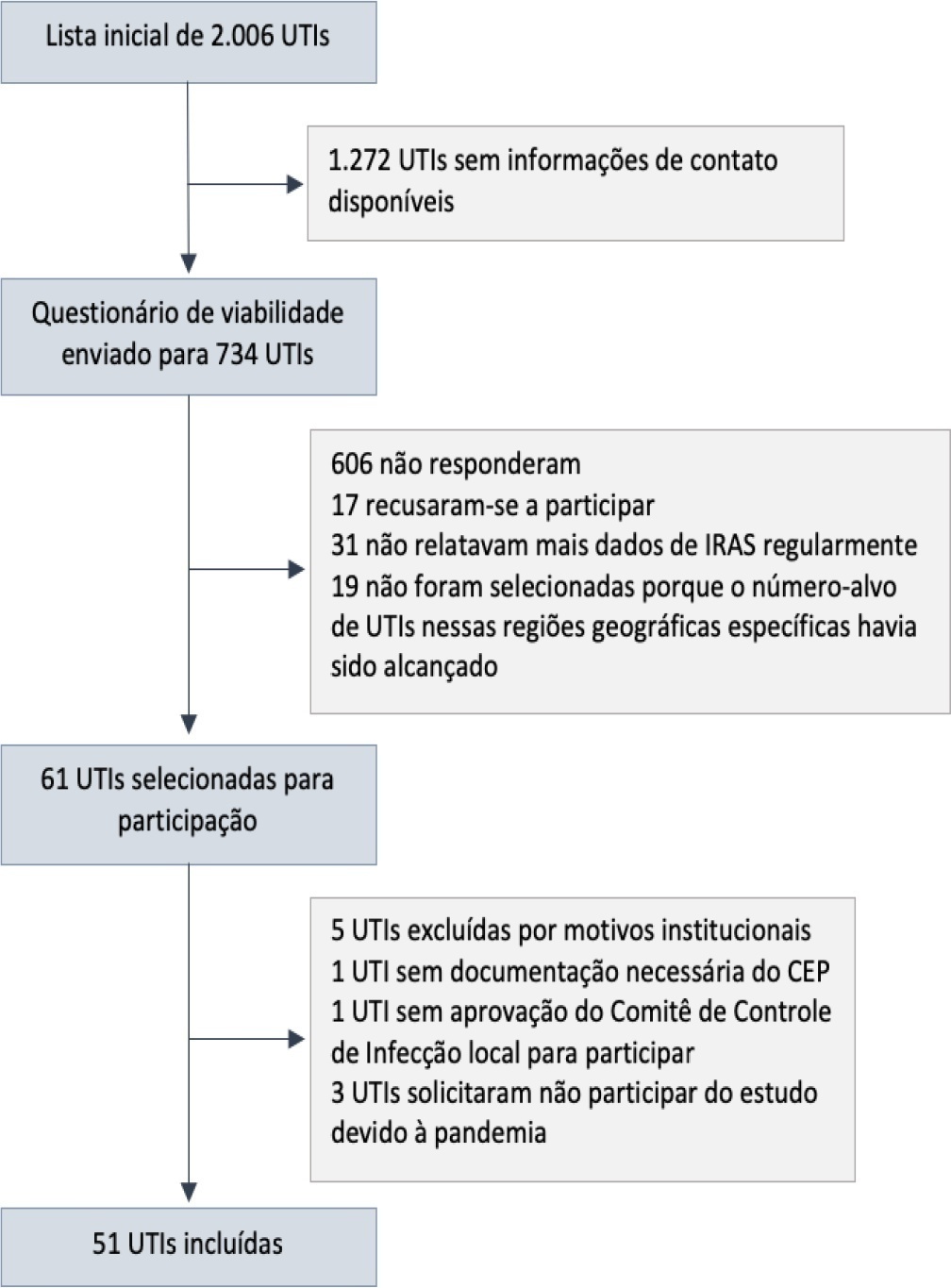
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
Conclusion:
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.
Keywords:bacterialBacterial infectionsDatabaseDatabase management systemsDrug-resistanceIMPACTO-MRIntensive care unitsSoftwareSee moreViews2

Abstract
Original ArticleIMPACTO-MR: a Brazilian nationwide platform study to assess infections and multidrug resistance in intensive care units
Rev Bras Ter Intensiva. 2022;34(4):418-425
DOI 10.5935/0103-507X.20220209-en
- Bruno M Tomazini
 ,
, - Antonio Paulo Nassar Jr
 ,
, - Thiago Costa Lisboa
 ,
, - Luciano César Pontes de Azevedo
 ,
, - Viviane Cordeiro Veiga
 ,
, - Daniela Ghidetti Mangas Catarino
 ,
, - Debora Vacaro Fogazzi
 ,
, - Beatriz Arns
 ,
, - Filipe Teixeira Piastrelli
 ,
, - Camila Dietrich
 ,
, - Karina Leal Negrelli,
- Isabella de Andrade Jesuíno,
- Luiz Fernando Lima Reis,
- Renata Rodrigues de Mattos,
- Carla Cristina Gomes Pinheiro,
- Mariane Nascimento Luz,
- Clayse Carla da Silva Spadoni,
- Elisângela Emilene Moro,
- Flávia Regina Bueno,
- Camila Santana Justo Cintra Sampaio,
- Débora Patrício Silva,
- Franca Pellison Baldassare,
- Ana Cecilia Alcantara Silva,
- Thabata Veiga,
- Leticia Barbante,
- Marianne Lambauer,
- Viviane Bezerra Campos,
- Elton Santos,
- Renato Hideo Nakawaga Santos,
- Ligia Nasi Laranjeiras,
- Nanci Valeis,
- Eliana Santucci,
- Tamiris Abait Miranda,
- Ana Cristina Lagoeiro do Patrocínio,
- Andréa de Carvalho,
- Eduvirgens Maria Couto de Sousa,
- Ancelmo Honorato Ferraz de Sousa,
- Daniel Tavares Malheiro,
- Isabella Lott Bezerra,
- Mirian Batista Rodrigues,
- Julliana Chicuta Malicia,
- Sabrina Souza da Silva,
- Bruna dos Passos Gimenes,
- Guilhermo Prates Sesin,
- Alexandre Prehn Zavascki,
- Daniel Sganzerla,
- Gregory Saraiva Medeiros,
- Rosa da Rosa Minho dos Santos,
- Fernanda Kelly Romeiro Silva,
- Maysa Yukari Cheno,
- Carolinne Ferreira Abrahão,
- Haliton Alves de Oliveira Junior,
- Leonardo Lima Rocha,
- Pedro Aniceto Nunes Neto,
- Valéria Chagas Pereira,
- Luis Eduardo Miranda Paciência,
- Elaine Silva Bueno,
- Eliana Bernadete Caser,
- Larissa Zuqui Ribeiro,
- Caio Cesar Ferreira Fernandes,
- Juliana Mazzei Garcia,
- Vanildes de Fátima Fernandes Silva,
- Alisson Junior dos Santos,
- Flávia Ribeiro Machado,
- Maria Aparecida de Souza,
- Bianca Ramos Ferronato,
- Hugo Corrêa de Andrade Urbano,
- Danielle Conceição Aparecida Moreira,
- Vicente Cés de Souza-Dantas,
- Diego Meireles Duarte,
- Juliana Coelho,
- Rodrigo Cruvinel Figueiredo,
- Fernanda Foreque,
- Thiago Gomes Romano,
- Daniel Cubos,
- Vladimir Miguel Spirale,
- Roberta Schiavon Nogueira,
- Israel Silva Maia,
- Cassio Luis Zandonai,
- Wilson José Lovato,
- Rodrigo Barbosa Cerantola,
- Tatiana Gozzi Pancev Toledo,
- Pablo Oscar Tomba,
- Joyce Ramos de Almeida,
- Luciana Coelho Sanches,
- Leticia Pierini,
- Mariana Cunha,
- Michelle Tereza Sousa,
- Bruna Azevedo,
- Felipe Dal-Pizzol,
- Danusa de Castro Damasio,
- Marina Peres Bainy,
- Dagoberta Alves Vieira Beduhn,
- Joana D’Arc Vila Nova Jatobá,
- Maria Tereza Farias de Moura,
- Leila Rezegue de Moraes Rego,
- Adria Vanessa da Silva,
- Luana Pontes Oliveira,
- Eliene Sá Sodré Filho,
- Silvana Soares dos Santos,
- Itallo de Lima Neves,
- Vanessa Cristina de Aquino Leão,
- João Lucidio Lobato Paes,
- Marielle Cristina Mendes Silva,
- Cláudio Dornas de Oliveira,
- Raquel Caldeira Brant Santiago,
- Jorge Luiz da Rocha Paranhos,
- Iany Grinezia da Silva Wiermann,
- Durval Ferreira Fonseca Pedroso,
- Priscilla Yoshiko Sawada,
- Rejane Martins Prestes,
- Glícia Cardoso Nascimento,
- Cintia Magalhães Carvalho Grion,
- Claudia Maria Dantas de Maio Carrilho,
- Roberta Lacerda Almeida de Miranda Dantas,
- Eliane Pereira Silva,
- Antônio Carlos da Silva,
- Sheila Mara Bezerra de Oliveira,
- Nicole Alberti Golin,
- Rogerio Tregnago,
- Valéria Paes Lima,
- Kamilla Grasielle Nunes da Silva,
- Emerson Boschi,
- Viviane Buffon,
- André Sant’Ana Machado,
- Leticia Capeletti,
- Rafael Botelho Foernges,
- Andréia Schubert de Carvalho,
- Lúcio Couto de Oliveira Junior,
- Daniela Cunha de Oliveira,
- Everton Macêdo Silva,
- Julival Ribeiro,
- Francielle Constantino Pereira,
- Fernanda Borges Salgado,
- Caroline Deutschendorf,
- Cristofer Farias da Silva,
- Andre Luiz Nunes Gobatto,
- Carolaine Bomfim de Oliveira,
- Marianna Deway Andrade Dracoulakis,
- Natália Oliveira Santos Alvaia,
- Roberta Machado de Souza,
- Larissa Liz Cardoso de Araújo,
- Rodrigo Morel Vieira de Melo,
- Luiz Carlos Santana Passos,
- Claudia Fernanda de Lacerda Vidal,
- Fernanda Lopes de Albuquerque Rodrigues,
- Pedro Kurtz,
- Cássia Righy Shinotsuka,
- Maria Brandão Tavares,
- Igor das Virgens Santana,
- Luciana Macedo da Silva Gavinho,
- Alaís Brito Nascimento,
- Adriano J Pereira,
- Alexandre Biasi Cavalcanti
Views2ABSTRACT
Objective:
To describe the IMPACTO-MR, a Brazilian nationwide intensive care unit platform study focused on the impact of health care-associated infections due to multidrug-resistant bacteria.
Methods:
We described the IMPACTO-MR platform, its development, criteria for intensive care unit selection, characterization of core data collection, objectives, and future research projects to be held within the platform.
Results:
The core data were collected using the Epimed Monitor System® and consisted of demographic data, comorbidity data, functional status, clinical scores, admission diagnosis and secondary diagnoses, laboratory, clinical, and microbiological data, and organ support during intensive care unit stay, among others. From October 2019 to December 2020, 33,983 patients from 51 intensive care units were included in the core database.
Conclusion:
The IMPACTO-MR platform is a nationwide Brazilian intensive care unit clinical database focused on researching the impact of health care-associated infections due to multidrug-resistant bacteria. This platform provides data for individual intensive care unit development and research and multicenter observational and prospective trials.
Keywords:bacterialBacterial infectionsDatabaseDatabase management systemsDrug-resistanceIMPACTO-MRIntensive care unitsSoftwareSee more

- Bruno M Tomazini
-
Special Article
Antivirals for adult patients hospitalized with SARS-CoV-2 infection: A randomized, Phase II/III, multicenter, placebo-controlled, adaptive study, with multiple arms and stages. COALITION COVID-19 BRAZIL IX – REVOLUTIOn: protocol and statistical analysis plan
- Israel Silva Maia
 ,
, - Aline Marcadenti
 ,
, - Fernando Godinho Zampieri
 ,
, - Lucas Petri Damiani
 ,
, - Renato Hideo Nakagawa Santos
 , [ … ],
, [ … ], - Alexandre Biasi Cavalcanti

Abstract
Special ArticleAntivirals for adult patients hospitalized with SARS-CoV-2 infection: A randomized, Phase II/III, multicenter, placebo-controlled, adaptive study, with multiple arms and stages. COALITION COVID-19 BRAZIL IX – REVOLUTIOn: protocol and statistical analysis plan
Rev Bras Ter Intensiva. 2022;34(1):44-55
DOI 10.5935/0103-507X.20220002-en
- Israel Silva Maia
 ,
, - Aline Marcadenti
 ,
, - Fernando Godinho Zampieri
 ,
, - Lucas Petri Damiani
 ,
, - Renato Hideo Nakagawa Santos
 ,
, - Karina Leal Negrelli
 ,
, - Samara Pinheiro do Carmo Gomes
 ,
, - Jaqueline Oliveira Gomes
 ,
, - Mariana Barbosa dos Santos Carollo
 ,
, - Tamiris Abait Miranda
 ,
, - Eliana Santucci
 ,
, - Nanci Valeis
 ,
, - Ligia Nasi Laranjeira
 ,
, - Glauco Adrieno Westphal
 ,
, - Jacques Gabriel Alvares Horta
 ,
, - Uri Adrian Prync Flato
 ,
, - Camilo Fernandes
 ,
, - Waldemar Carlos Barros
 ,
, - Renata S Bolan
 ,
, - Otávio Celso Eluf Gebara
 ,
, - Meton Soares de Alencar Filho
 ,
, - Victor Augusto Hamamoto
 ,
, - Mauro Esteves Hernandes
 ,
, - Nicole Alberti Golin
 ,
, - Ronald Torres de Olinda
 ,
, - Flávia Ribeiro Machado
 ,
, - Régis Goulart Rosa
 ,
, - Viviane Cordeiro Veiga
 ,
, - Luciano César Pontes de Azevedo
 ,
, - Alvaro Avezum
 ,
, - Renato Delascio Lopes
 ,
, - Tiago Moreno L Souza
 ,
, - Otávio Berwanger
 ,
, - Alexandre Biasi Cavalcanti

Views2ABSTRACT
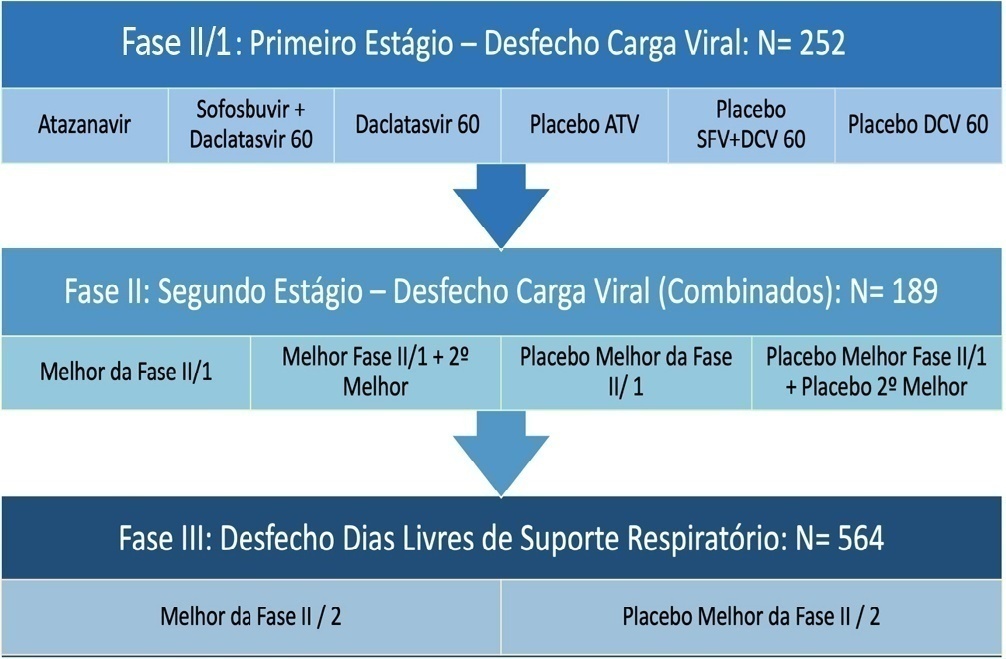
Repurposed drugs are important in resource-limited settings because the interventions are more rapidly available, have already been tested safely in other populations and are inexpensive. Repurposed drugs are an effective solution, especially for emerging diseases such as COVID-19. The REVOLUTIOn trial has the objective of evaluating three repurposed antiviral drugs, atazanavir, daclatasvir and sofosbuvir, already used for HIV- and hepatitis C virus-infected patients in a randomized, placebo-controlled, adaptive, multiarm, multistage study. The drugs will be tested simultaneously in a Phase II trial to first identify whether any of these drugs alone or in combination reduce the viral load. If they do, a Phase III trial will be initiated to investigate if these medications are capable of increasing the number of days free respiratory support. Participants must be hospitalized adults aged ≥ 18 years with initiation of symptoms ≤ 9 days and SpO2 ≤ 94% in room air or a need for supplemental oxygen to maintain an SpO2 > 94%. The expected total sample size ranges from 252 to 1,005 participants, depending on the number of stages that will be completed in the study. Hence, the protocol is described here in detail together with the statistical analysis plan. In conclusion, the REVOLUTIOn trial is designed to provide evidence on whether atazanavir, daclatasvir or sofosbuvir decrease the SARS-CoV-2 load in patients with COVID-19 and increase the number of days patients are free of respiratory support. In this protocol paper, we describe the rationale, design, and status of the trial.
ClinicalTrials.gov identifier:
Keywords:Antiviral agentsCoronavirus infectionsCOVID-19DaclatasvirprotocolRespiratory insufficiencySofosbuvirSee moreViews2

Abstract
Special ArticleAntivirals for adult patients hospitalized with SARS-CoV-2 infection: A randomized, Phase II/III, multicenter, placebo-controlled, adaptive study, with multiple arms and stages. COALITION COVID-19 BRAZIL IX – REVOLUTIOn: protocol and statistical analysis plan
Rev Bras Ter Intensiva. 2022;34(1):44-55
DOI 10.5935/0103-507X.20220002-en
- Israel Silva Maia
 ,
, - Aline Marcadenti
 ,
, - Fernando Godinho Zampieri
 ,
, - Lucas Petri Damiani
 ,
, - Renato Hideo Nakagawa Santos
 ,
, - Karina Leal Negrelli
 ,
, - Samara Pinheiro do Carmo Gomes
 ,
, - Jaqueline Oliveira Gomes
 ,
, - Mariana Barbosa dos Santos Carollo
 ,
, - Tamiris Abait Miranda
 ,
, - Eliana Santucci
 ,
, - Nanci Valeis
 ,
, - Ligia Nasi Laranjeira
 ,
, - Glauco Adrieno Westphal
 ,
, - Jacques Gabriel Alvares Horta
 ,
, - Uri Adrian Prync Flato
 ,
, - Camilo Fernandes
 ,
, - Waldemar Carlos Barros
 ,
, - Renata S Bolan
 ,
, - Otávio Celso Eluf Gebara
 ,
, - Meton Soares de Alencar Filho
 ,
, - Victor Augusto Hamamoto
 ,
, - Mauro Esteves Hernandes
 ,
, - Nicole Alberti Golin
 ,
, - Ronald Torres de Olinda
 ,
, - Flávia Ribeiro Machado
 ,
, - Régis Goulart Rosa
 ,
, - Viviane Cordeiro Veiga
 ,
, - Luciano César Pontes de Azevedo
 ,
, - Alvaro Avezum
 ,
, - Renato Delascio Lopes
 ,
, - Tiago Moreno L Souza
 ,
, - Otávio Berwanger
 ,
, - Alexandre Biasi Cavalcanti

Views2ABSTRACT
Repurposed drugs are important in resource-limited settings because the interventions are more rapidly available, have already been tested safely in other populations and are inexpensive. Repurposed drugs are an effective solution, especially for emerging diseases such as COVID-19. The REVOLUTIOn trial has the objective of evaluating three repurposed antiviral drugs, atazanavir, daclatasvir and sofosbuvir, already used for HIV- and hepatitis C virus-infected patients in a randomized, placebo-controlled, adaptive, multiarm, multistage study. The drugs will be tested simultaneously in a Phase II trial to first identify whether any of these drugs alone or in combination reduce the viral load. If they do, a Phase III trial will be initiated to investigate if these medications are capable of increasing the number of days free respiratory support. Participants must be hospitalized adults aged ≥ 18 years with initiation of symptoms ≤ 9 days and SpO2 ≤ 94% in room air or a need for supplemental oxygen to maintain an SpO2 > 94%. The expected total sample size ranges from 252 to 1,005 participants, depending on the number of stages that will be completed in the study. Hence, the protocol is described here in detail together with the statistical analysis plan. In conclusion, the REVOLUTIOn trial is designed to provide evidence on whether atazanavir, daclatasvir or sofosbuvir decrease the SARS-CoV-2 load in patients with COVID-19 and increase the number of days patients are free of respiratory support. In this protocol paper, we describe the rationale, design, and status of the trial.
ClinicalTrials.gov identifier:
Keywords:Antiviral agentsCoronavirus infectionsCOVID-19DaclatasvirprotocolRespiratory insufficiencySofosbuvirSee more

- Israel Silva Maia
Search
Search in:
KEY WORDS
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis


