Home
Abstract
Rev Bras Ter Intensiva. 2018;30(2):219-225
DOI 10.5935/0103-507X.20180036
To review the evidence on the safety of neuromuscular electrical stimulation when used in the intensive care unit.
A systematic review was conducted; a literature search was performed of the MEDLINE (via PubMed), PEDro, Cochrane CENTRAL and EMBASE databases, and a further manual search was performed among the references cited in randomized studies. Randomized clinical trials that compared neuromuscular electrical stimulation to a control or placebo group in the intensive care unit and reporting on the technique safety in the outcomes were included. Hemodynamic variables and information on adverse effects were considered safety parameters. Articles were independently analyzed by two reviewers, and the data analysis was descriptive.
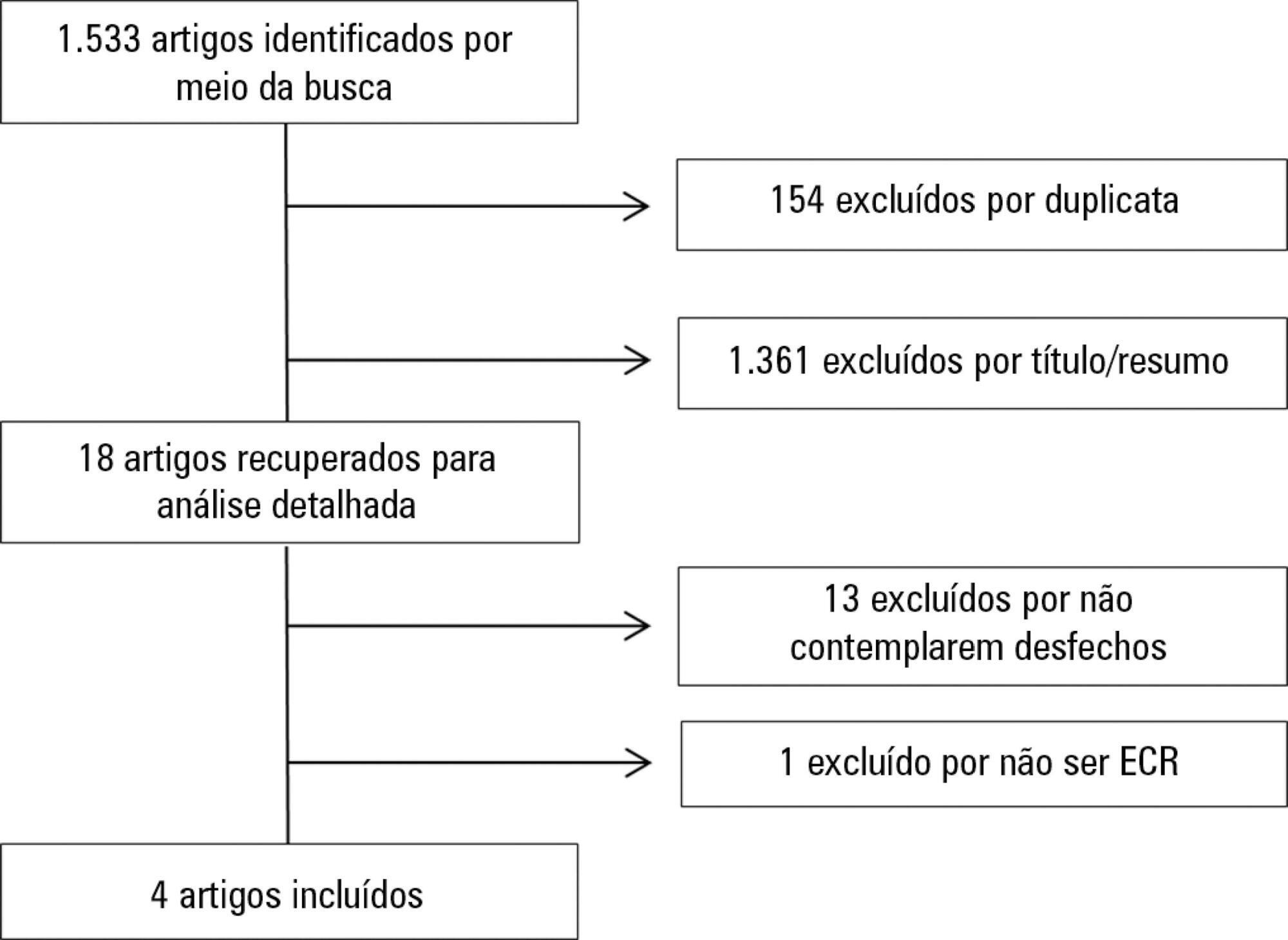
The initial search located 1,533 articles, from which only four randomized clinical trials were included. Two studies assessed safety based on hemodynamic variables, and only one study reported an increase in heart rate, respiratory rate and blood lactate, without clinical relevance. The other two studies assessed safety based on reported adverse effects. In one, 15% of patients described a prickling sensation, without any clinically relevant abnormalities. In the other, one patient suffered a superficial burn due to improper parameter configuration.
Neuromuscular electrical stimulation is safe for critically ill patients; however, it should be applied by duly trained professionals and with proper evidence-based parameters.


Abstract
Rev Bras Ter Intensiva. 2018;30(2):219-225
DOI 10.5935/0103-507X.20180036
To review the evidence on the safety of neuromuscular electrical stimulation when used in the intensive care unit.
A systematic review was conducted; a literature search was performed of the MEDLINE (via PubMed), PEDro, Cochrane CENTRAL and EMBASE databases, and a further manual search was performed among the references cited in randomized studies. Randomized clinical trials that compared neuromuscular electrical stimulation to a control or placebo group in the intensive care unit and reporting on the technique safety in the outcomes were included. Hemodynamic variables and information on adverse effects were considered safety parameters. Articles were independently analyzed by two reviewers, and the data analysis was descriptive.
The initial search located 1,533 articles, from which only four randomized clinical trials were included. Two studies assessed safety based on hemodynamic variables, and only one study reported an increase in heart rate, respiratory rate and blood lactate, without clinical relevance. The other two studies assessed safety based on reported adverse effects. In one, 15% of patients described a prickling sensation, without any clinically relevant abnormalities. In the other, one patient suffered a superficial burn due to improper parameter configuration.
Neuromuscular electrical stimulation is safe for critically ill patients; however, it should be applied by duly trained professionals and with proper evidence-based parameters.


Search
Search in:
Case reports Child Coronavirus infections COVID-19 Critical care Critical illness Extracorporeal membrane oxygenation Infant, newborn Intensive care Intensive care units Intensive care units, pediatric mechanical ventilation Mortality Physical therapy modalities Prognosis Respiration, artificial Respiratory insufficiency risk factors SARS-CoV-2 Sepsis


